K2M Group Holdings Inc. 11.08.17
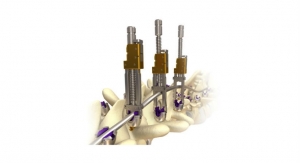
K2M Group Holdings Inc., a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance, has acquired from Cardinal Spine, a privately held medical device company, the PALO ALTO Cervical Static Corpectomy Cage System. PALO ALTO, a cervical vertebral body replacement device, is the first and only static corpectomy cage in the world to receive a cervical 510(k) clearance from the U.S. Food and Drug Administration (FDA). In addition to PALO ALTO, K2M has also acquired the associated intellectual property and product inventory.
The PALO ALTO Cervical Static Corpectomy Cage System is indicated for use in the cervical spine in skeletally mature patients to replace diseased or damaged vertebral bodies, or for reconstruction to achieve decompression of the spinal cord and neural tissues in cervical degenerative disorders. It is intended for use with autograft or allogenic bone graft comprising cancellous and/or corticocancellous bone graft, as an adjunct to fusion.
“K2M has impressed me with their technologies. I’m elated about PALO ALTO; the long-term clinical data showed a 96 percent fusion rate,” said Frank P. Castro Jr., M.D., an orthopedic surgeon at Baptist Health Floyd in New Albany, Ind., and founder of Cardinal Spine. “The trapezoidal design helps increase the graft-host surface contact area, while providing resistance to posterior cage migration.”
“We are excited to expand our complex cervical offerings by adding the first and only FDA-cleared static cervical vertebral body replacement device,” K2M Chairman, President, and CEO Eric Major said. “We look forward to fully integrating this clinically supported technology into our product portfolio, which when complemented by our Balance ACS platform, allows surgeons to address the full range of spinal pathologies, and ultimately, to facilitate Total Body Balance in their patients.”
K2M Group Holdings Inc. is a global developer of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance. Since its inception, K2M has designed, developed, and commercialized complex spine and minimally invasive spine technologies and techniques used by spine surgeons to treat some of the most complicated spinal pathologies. K2M has leveraged these core competencies into Balance ACS, a platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes, with the goal of supporting the full continuum of care to facilitate quality patient outcomes. The company is based in Leesburg, Va.
The PALO ALTO Cervical Static Corpectomy Cage System is indicated for use in the cervical spine in skeletally mature patients to replace diseased or damaged vertebral bodies, or for reconstruction to achieve decompression of the spinal cord and neural tissues in cervical degenerative disorders. It is intended for use with autograft or allogenic bone graft comprising cancellous and/or corticocancellous bone graft, as an adjunct to fusion.
“K2M has impressed me with their technologies. I’m elated about PALO ALTO; the long-term clinical data showed a 96 percent fusion rate,” said Frank P. Castro Jr., M.D., an orthopedic surgeon at Baptist Health Floyd in New Albany, Ind., and founder of Cardinal Spine. “The trapezoidal design helps increase the graft-host surface contact area, while providing resistance to posterior cage migration.”
“We are excited to expand our complex cervical offerings by adding the first and only FDA-cleared static cervical vertebral body replacement device,” K2M Chairman, President, and CEO Eric Major said. “We look forward to fully integrating this clinically supported technology into our product portfolio, which when complemented by our Balance ACS platform, allows surgeons to address the full range of spinal pathologies, and ultimately, to facilitate Total Body Balance in their patients.”
K2M Group Holdings Inc. is a global developer of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance. Since its inception, K2M has designed, developed, and commercialized complex spine and minimally invasive spine technologies and techniques used by spine surgeons to treat some of the most complicated spinal pathologies. K2M has leveraged these core competencies into Balance ACS, a platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes, with the goal of supporting the full continuum of care to facilitate quality patient outcomes. The company is based in Leesburg, Va.