Globe Newswire01.03.18
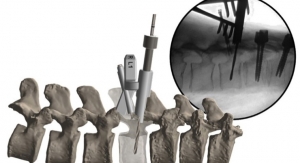
OrthoPediatrics Corp., a company exclusively focused on advancing the field of pediatric orthopedics, has launched Titanium PediPlates, its 24th surgical system. The titanium implant system expands OrthoPediatrics’ physeal tethering offering and includes both O-Plates and I-Plates as well as solid and cannulated low-profile screws. As a market leader in guided growth, OrthoPediatrics’ PediPlates technology harnesses the power of the growth plate through a simple, minimally invasive, and reversible tethering technique.
Joe Hauser, OrthoPediatrics’ vice president of Trauma and Deformity Correction, noted that titanium PediPlates provide an additional correction option to pediatric orthopedic surgeons worldwide. “Adding a titanium offering to OrthoPediatrics’ PediPlates physeal tethering franchise expands physicians’ treatment options, especially in patients who have a nickel allergy. The launch of our 24th surgical system strengthens our strategy to provide pediatric orthopedic surgeons with superior options for treating children.”
The PediPlates system, which includes the I-Plate, O-Plate, and Delta Plate, provides a physeal tethering technique that does not disrupt the integrity of the physis, or growth plate. It features simple plate and screw constructs that span the growth center and restrain the physis by inhibiting growth where the plates and screws are applied. The system also features stainless steel plates and screws, which provide excellent strength, resistance to breakage, and ease of removal.
Founded in 2006, OrthoPediatrics is focused exclusively on providing a product offering to the pediatric orthopedic market to improve the lives of children with orthopedic conditions. OrthoPediatrics currently markets 24 surgical systems that serve three of the largest categories within the pediatric orthopedic market. This offering spans trauma and deformity, complex spine, and ACL reconstruction procedures. OrthoPediatrics’ global sales organization is focused exclusively on pediatric orthopedics and distributes its products in the United States and 35 countries outside the United States. The firm is based in Warsaw, Ind.
Joe Hauser, OrthoPediatrics’ vice president of Trauma and Deformity Correction, noted that titanium PediPlates provide an additional correction option to pediatric orthopedic surgeons worldwide. “Adding a titanium offering to OrthoPediatrics’ PediPlates physeal tethering franchise expands physicians’ treatment options, especially in patients who have a nickel allergy. The launch of our 24th surgical system strengthens our strategy to provide pediatric orthopedic surgeons with superior options for treating children.”
The PediPlates system, which includes the I-Plate, O-Plate, and Delta Plate, provides a physeal tethering technique that does not disrupt the integrity of the physis, or growth plate. It features simple plate and screw constructs that span the growth center and restrain the physis by inhibiting growth where the plates and screws are applied. The system also features stainless steel plates and screws, which provide excellent strength, resistance to breakage, and ease of removal.
Founded in 2006, OrthoPediatrics is focused exclusively on providing a product offering to the pediatric orthopedic market to improve the lives of children with orthopedic conditions. OrthoPediatrics currently markets 24 surgical systems that serve three of the largest categories within the pediatric orthopedic market. This offering spans trauma and deformity, complex spine, and ACL reconstruction procedures. OrthoPediatrics’ global sales organization is focused exclusively on pediatric orthopedics and distributes its products in the United States and 35 countries outside the United States. The firm is based in Warsaw, Ind.