Globe Newswire02.02.18
Advanced Biomedical Technologies Inc., a developer and manufacturer of orthopedic internal fixation devices, has been issued a Chinese patent titled, “Bone Fracture Plate Made of High Polymer Materials.”
The company’s subsidiary Shenzhen Changhua Biomedical Engineering Company Limited is entitled for the new patent, which strengthens the firm’s position in the manufacturing process and related controls using polyamide materials (PA).
Advanced Biomedical CEO Wang Hui said, “This new patent strengthens our IP rights and enables us to develop new products. The combination of fiber-reinforced technology and bionic structure design, allows the medical devices to contain similar mechanical properties to human bone. The elastic fixation does not produce stress shielding, resulting in good fixation effect and biocompatibility. The devices can stimulate bone tissues to achieve effective biological integration, which will benefit bone regeneration. Our formulated materials can be widely used in the field of orthopedic internal fixation in an ever-growing and prospective market which is currently dominated by existing materials such as metal and polylactic acid-based devices.”
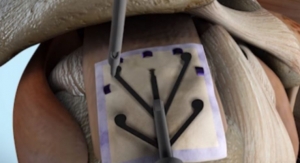
Advanced Biomedical Technologies Inc.’s primary product line includes internal fixation devices consisting of proprietary high grade polymers (polyamide). Its products are used in a variety of applications including orthopedic trauma, sports related medical treatment, or cartilage injuries, and reconstructive dental procedures. During the healing process, the products stimulate new bone growth which replaces the degrading device, leaving newer, stronger bone in the exact location of the injury; thus making the site of the injury stronger and more resistant to recurring damage.
These products provide an alternative to metal implants and overcome the limitations of other re-absorbable fixation devices. The material is utilized in producing human body implant screws, binding wires, rods and related implantation products. With over 15 years of meticulous in clinical trials, the company has developed the internal fixation devices to be clinically effective and safe.
The products and materials that the company has created differ from competing bio-degradable and metal based products currently being marketed by:
The company’s subsidiary Shenzhen Changhua Biomedical Engineering Company Limited is entitled for the new patent, which strengthens the firm’s position in the manufacturing process and related controls using polyamide materials (PA).
Advanced Biomedical CEO Wang Hui said, “This new patent strengthens our IP rights and enables us to develop new products. The combination of fiber-reinforced technology and bionic structure design, allows the medical devices to contain similar mechanical properties to human bone. The elastic fixation does not produce stress shielding, resulting in good fixation effect and biocompatibility. The devices can stimulate bone tissues to achieve effective biological integration, which will benefit bone regeneration. Our formulated materials can be widely used in the field of orthopedic internal fixation in an ever-growing and prospective market which is currently dominated by existing materials such as metal and polylactic acid-based devices.”
Advanced Biomedical Technologies Inc.’s primary product line includes internal fixation devices consisting of proprietary high grade polymers (polyamide). Its products are used in a variety of applications including orthopedic trauma, sports related medical treatment, or cartilage injuries, and reconstructive dental procedures. During the healing process, the products stimulate new bone growth which replaces the degrading device, leaving newer, stronger bone in the exact location of the injury; thus making the site of the injury stronger and more resistant to recurring damage.
These products provide an alternative to metal implants and overcome the limitations of other re-absorbable fixation devices. The material is utilized in producing human body implant screws, binding wires, rods and related implantation products. With over 15 years of meticulous in clinical trials, the company has developed the internal fixation devices to be clinically effective and safe.
The products and materials that the company has created differ from competing bio-degradable and metal based products currently being marketed by:
- The ability to control the speed that the device degrades; therefore improving upon healing time.
- Eliminating the need for a second surgery to replace device due to infection or other post-operative complications.
- The capability of being evenly absorbed from outer layer inwards, so it gives enough restoration time for bone healing and re-growth.