GlobeNewswire09.27.18
Alphatec Holdings Inc., a provider of spine surgery solutions with a mission to improve patient lives through the relentless pursuit of superior outcomes, today announced that it has received 510(k) clearance from the U.S. Food & Drug Administration (FDA) for its OsseoScrew System, making it available for clinical use for the first time in the United States.
"I am exceptionally pleased that OsseoScrew has been cleared for the U.S. market,” said Pat Miles, Chairman and CEO. “The system will create new market opportunities for ATEC by uniquely addressing an unmet need for an underserved patient population. We are building an organic innovation machine and today’s announcement represents another significant step toward creating a clinically distinguished portfolio.”
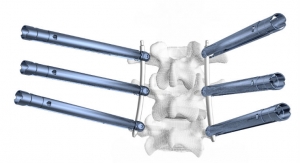
OsseoScrew is a next-generation expandable pedicle screw system, intended to restore the integrity of the spinal column in elderly patients with advanced stage tumors involving the thoracic and lumbar spine. It is designed to be implanted into the pedicle, then expanded after implantation to achieve increased screw fixation and reduced post-operative pullout in bone with poor density. OsseoScrew has been clinically proven to increase pullout and holding strength, improving fixation in the bone-implant interface by 29 percent, as compared to conventional pedicle screws. It performs comparably to cemented fenestrated screws without the risk associated with cement leakage.
Alphatec will display OsseoScrew at the North American Spine Society (NASS) 2018 Annual Meeting, which is being held from September 26-29 at the Los Angeles Convention Center.
"I am exceptionally pleased that OsseoScrew has been cleared for the U.S. market,” said Pat Miles, Chairman and CEO. “The system will create new market opportunities for ATEC by uniquely addressing an unmet need for an underserved patient population. We are building an organic innovation machine and today’s announcement represents another significant step toward creating a clinically distinguished portfolio.”
OsseoScrew is a next-generation expandable pedicle screw system, intended to restore the integrity of the spinal column in elderly patients with advanced stage tumors involving the thoracic and lumbar spine. It is designed to be implanted into the pedicle, then expanded after implantation to achieve increased screw fixation and reduced post-operative pullout in bone with poor density. OsseoScrew has been clinically proven to increase pullout and holding strength, improving fixation in the bone-implant interface by 29 percent, as compared to conventional pedicle screws. It performs comparably to cemented fenestrated screws without the risk associated with cement leakage.
Alphatec will display OsseoScrew at the North American Spine Society (NASS) 2018 Annual Meeting, which is being held from September 26-29 at the Los Angeles Convention Center.