Business Wire02.11.19
Orthofix Medical Inc., a global medical device company focused on musculoskeletal products and therapies, has received U.S. Food and Drug Administration (FDA) approval of the M6-C artificial cervical disc for patients suffering from cervical disc degeneration. The M6-C artificial cervical disc was developed by Spinal Kinetics, a company acquired by Orthofix in April 2018.
“With the approval of the M6-C artificial cervical disc, Orthofix now has the industry’s most comprehensive portfolio of cervical spine products to best serve our surgeon customers and patients,” said Orthofix President and CEO Brad Mason. “In addition to a full line of anterior, posterior and interbody fusion cervical implants, we offer the CervicalStimdevice, the only FDA-approved cervical bone growth stimulation therapy, and the Trinity ELITE allograft, an allograft developed in partnership with MTF Biologics. The launch of the M6-C artificial cervical disc in the U.S. will complement this existing spine portfolio and represents a significant milestone in the execution of our strategy to accelerate topline growth.”
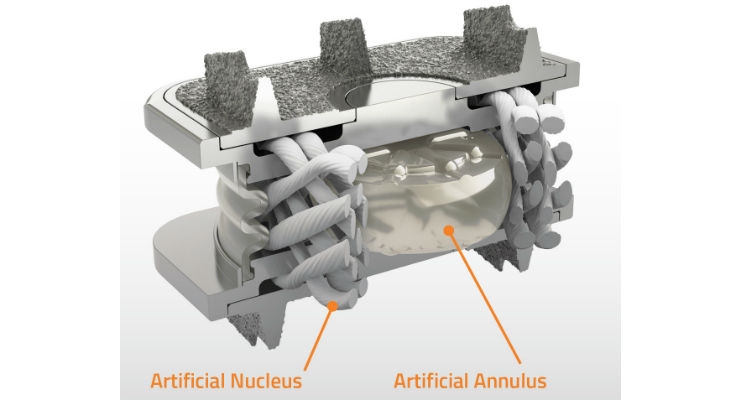
The M6-C artificial cervical disc is a next-generation artificial disc developed to replace an intervertebral disc damaged by cervical disc degeneration. Designed to restore physiologic motion to the spine, the M6-C disc is indicated as an alternative to cervical fusion. The M6-C artificial cervical disc preserves motion by restoring biomechanical function at the treated level after native disc removal and potentially reduces subsequent degeneration of adjacent vertebral segments. The M6-C device is the only artificial cervical disc that mimics the anatomic structure of a natural disc by incorporating an artificial visco-elastic nucleus and fiber annulus into its design. Like a natural disc, this unique construct allows for shock absorption at the implanted level, as well as provides a controlled range of motion when the spine transitions in its combined complex movements.
Pre-market approval was based on clinical data from a U.S. Investigational Device Exemption (IDE) study that evaluated the safety and effectiveness of the M6-C artificial cervical disc compared to anterior cervical discectomy and fusion (ACDF) for the treatment of symptomatic cervical radiculopathy with or without cord compression. Patients in the study presented with degenerative cervical radiculopathy requiring surgical intervention, and confirmed clinically and radiographically at one vertebral level from C3 to C7.
Study results at 24 months include the following clinical patient outcomes:
The prospective, non-randomized, concurrently controlled, multi-center clinical trial was conducted at 23 sites in the United States.
“Cervical disc degeneration is a common condition that can cause pain in the neck, shoulders, arms and hands and lead to numbness and weakness in the arm or hands. Often this condition is the result of a vertebral disc that has deteriorated or been damaged due to the natural aging process or an injury,” said Dr. Rick Sasso, orthopedic spine surgeon, professor and chief of spine surgery at the Indiana University School of Medicine in Indianapolis and an investigator in the M6-C artificial cervical disc IDE study. “For many of these patients, the M6-C artificial cervical disc will give us the ability to replace the patient’s damaged disc with one that closely mimics their natural disc, enabling them to regain motion and return to their normal activities.”
Orthofix expects to release the M6-C artificial cervical disc in 2019 through a controlled, limited market launch in the United States accompanied by an extensive training and education curriculum for surgeons. The M6-C artificial cervical disc received CE Mark approval for distribution in the European Union and other international geographies in 2006 and there have been more than 45,000 implants of the M6-C artificial cervical disc outside of the United States to date.
Orthofix Medical Inc. is a global medical device company focused on musculoskeletal products and therapies. The company’s mission is to improve patients’ lives by providing superior reconstruction and regenerative musculoskeletal solutions to physicians worldwide. Headquartered in Lewisville, Texas, Orthofix’s spine and orthopedic extremities products are distributed in more than seventy countries via the company’s sales representatives and distributors.
“With the approval of the M6-C artificial cervical disc, Orthofix now has the industry’s most comprehensive portfolio of cervical spine products to best serve our surgeon customers and patients,” said Orthofix President and CEO Brad Mason. “In addition to a full line of anterior, posterior and interbody fusion cervical implants, we offer the CervicalStimdevice, the only FDA-approved cervical bone growth stimulation therapy, and the Trinity ELITE allograft, an allograft developed in partnership with MTF Biologics. The launch of the M6-C artificial cervical disc in the U.S. will complement this existing spine portfolio and represents a significant milestone in the execution of our strategy to accelerate topline growth.”
The M6-C artificial cervical disc is a next-generation artificial disc developed to replace an intervertebral disc damaged by cervical disc degeneration. Designed to restore physiologic motion to the spine, the M6-C disc is indicated as an alternative to cervical fusion. The M6-C artificial cervical disc preserves motion by restoring biomechanical function at the treated level after native disc removal and potentially reduces subsequent degeneration of adjacent vertebral segments. The M6-C device is the only artificial cervical disc that mimics the anatomic structure of a natural disc by incorporating an artificial visco-elastic nucleus and fiber annulus into its design. Like a natural disc, this unique construct allows for shock absorption at the implanted level, as well as provides a controlled range of motion when the spine transitions in its combined complex movements.
Pre-market approval was based on clinical data from a U.S. Investigational Device Exemption (IDE) study that evaluated the safety and effectiveness of the M6-C artificial cervical disc compared to anterior cervical discectomy and fusion (ACDF) for the treatment of symptomatic cervical radiculopathy with or without cord compression. Patients in the study presented with degenerative cervical radiculopathy requiring surgical intervention, and confirmed clinically and radiographically at one vertebral level from C3 to C7.
Study results at 24 months include the following clinical patient outcomes:
- Nearly 91 percent of patients who received the M6-C disc demonstrated a meaningful clinical improvement in the Neck Disability Index.
- A meaningful clinical improvement in arm pain score was seen in 91.2 percent of the M6-C disc patients compared to 77.9 percent in ACDF patients. In addition, this same meaningful improvement was seen in neck pain scores with 90.5 percent of patients who received the M6-C disc compared to 79.9 percent in patients who underwent the ACDF procedure.
- The M6-C disc preserved range of motion in flexion-extension and lateral bending in the study group.
- Prior to surgery, 80.6 percent of the M6-C disc patients and 85.7 percent of the ACDF patients were taking some type of pain medication for the treatment of their cervical spine condition. At 24 months, 14 percent of the M6-C disc patients were still taking some type of pain medication compared to 38.2 percent of the ACDF patients.
- Of the patients still taking pain medication there was a seven times higher rate of opioid use with the ACDF patients than with patients who received the M6-C disc.
- Additional surgery at the treated level was needed in 4.8 percent of the ACDF patients compared to 1.9 percent of the M6-C disc patients.
The prospective, non-randomized, concurrently controlled, multi-center clinical trial was conducted at 23 sites in the United States.
“Cervical disc degeneration is a common condition that can cause pain in the neck, shoulders, arms and hands and lead to numbness and weakness in the arm or hands. Often this condition is the result of a vertebral disc that has deteriorated or been damaged due to the natural aging process or an injury,” said Dr. Rick Sasso, orthopedic spine surgeon, professor and chief of spine surgery at the Indiana University School of Medicine in Indianapolis and an investigator in the M6-C artificial cervical disc IDE study. “For many of these patients, the M6-C artificial cervical disc will give us the ability to replace the patient’s damaged disc with one that closely mimics their natural disc, enabling them to regain motion and return to their normal activities.”
Orthofix expects to release the M6-C artificial cervical disc in 2019 through a controlled, limited market launch in the United States accompanied by an extensive training and education curriculum for surgeons. The M6-C artificial cervical disc received CE Mark approval for distribution in the European Union and other international geographies in 2006 and there have been more than 45,000 implants of the M6-C artificial cervical disc outside of the United States to date.
Orthofix Medical Inc. is a global medical device company focused on musculoskeletal products and therapies. The company’s mission is to improve patients’ lives by providing superior reconstruction and regenerative musculoskeletal solutions to physicians worldwide. Headquartered in Lewisville, Texas, Orthofix’s spine and orthopedic extremities products are distributed in more than seventy countries via the company’s sales representatives and distributors.