Business Wire05.13.19
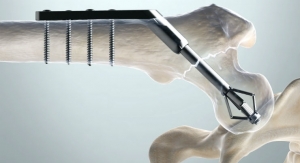
Bone Biologics Corp., a developer of orthobiologic products for domestic and international spine fusion markets, has received Human Research Ethics Committee (HREC) approval for the first center of a multicenter pilot clinical trial to evaluate NB1 (NELL-1/DBX) in 30 patients in Australia. The pilot study will evaluate the safety and effectiveness of NB1 in adult subjects with degenerative disc disease (DDD) at one level from L2-S1, who may also have up to Grade 1 spondylolisthesis or Grade 1 retrolisthesis at the involved level who undergo transforaminal lumbar interbody fusion (TLIF).
The study design has been previously reviewed by the U.S. Food and Drug Administration’s (FDA’s) Division of Orthopedic Devices in a pre-submission and is intended to support progression to a pivotal clinical study in the United States. The Therapeutic Goods Administration (TGA) in Australia will be notified of the conduct of the study through the Clinical Trial Notification Scheme. “This marks a significant milestone in the advancement of NELL-1/DBX,” remarked company Chief Operating Officer Jeffrey Frelick.
Lumbar DDD is one of the most common causes of low back pain. DDD also leads to substantial disability, with many patients suffering from decreased ability to walk, sit, stand, and/or sleep. For some people, DDD is part of the natural process of growing older and is a significant medical issue that is increasing as the global population ages.
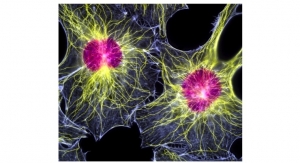
Bone Biologics was founded to pursue regenerative medicine for bone. Bone Biologics Corporation is undertaking groundbreaking work and building on unprecedented research on the NELL-1 molecule that has produced a significant number of studies and publications in peer-reviewed scientific literature. Bone Biologics is currently focusing its development efforts for its bone graft substitute product on bone regeneration in spinal fusion. NELL-1 is a recombinant human protein growth factor that is essential for normal bone development.
The study design has been previously reviewed by the U.S. Food and Drug Administration’s (FDA’s) Division of Orthopedic Devices in a pre-submission and is intended to support progression to a pivotal clinical study in the United States. The Therapeutic Goods Administration (TGA) in Australia will be notified of the conduct of the study through the Clinical Trial Notification Scheme. “This marks a significant milestone in the advancement of NELL-1/DBX,” remarked company Chief Operating Officer Jeffrey Frelick.
Lumbar DDD is one of the most common causes of low back pain. DDD also leads to substantial disability, with many patients suffering from decreased ability to walk, sit, stand, and/or sleep. For some people, DDD is part of the natural process of growing older and is a significant medical issue that is increasing as the global population ages.
Bone Biologics was founded to pursue regenerative medicine for bone. Bone Biologics Corporation is undertaking groundbreaking work and building on unprecedented research on the NELL-1 molecule that has produced a significant number of studies and publications in peer-reviewed scientific literature. Bone Biologics is currently focusing its development efforts for its bone graft substitute product on bone regeneration in spinal fusion. NELL-1 is a recombinant human protein growth factor that is essential for normal bone development.