Sam Brusco, Associate Editor02.03.21
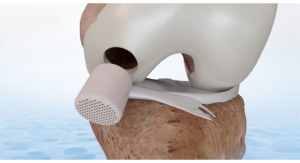
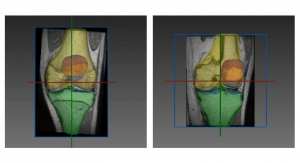
Geistlich Pharma, a Swiss developer of biomaterials for regenerative therapies in orthopedics, has earned Breakthrough Device Designation from the U.S. Food and Drug Administration for its Chondro-Gide articular cartilage cover. Chondro-Gide is a collagen membrane used in a single-step cartilage repair technique.
The highly purified collagen membrane leverages the body’s healing potential. It is used with bone marrow stimulation to treat cartilage defects.
Over 10 years of clinical success in Europe have demonstrated the product as a cost-effective single surgery treatment to repair cartilage lesions, enhancing the effect of established bone marrow stimulation techniques. Cartilage defect and injury repair is key to alleviating or preventing pain and slowing the progression of damage that can lead to osteoarthritis.
“The device is versatile and can be used in minimally invasive procedures where indicated,” Carrie Hartill, Business Unit Director for Geistlich Surgery told the press. “Cost effective, single surgery cartilage repair devices are not currently available in the USA, and Chondro-Gide will fill this unmet clinical need.”
The highly purified collagen membrane leverages the body’s healing potential. It is used with bone marrow stimulation to treat cartilage defects.
Over 10 years of clinical success in Europe have demonstrated the product as a cost-effective single surgery treatment to repair cartilage lesions, enhancing the effect of established bone marrow stimulation techniques. Cartilage defect and injury repair is key to alleviating or preventing pain and slowing the progression of damage that can lead to osteoarthritis.
“The device is versatile and can be used in minimally invasive procedures where indicated,” Carrie Hartill, Business Unit Director for Geistlich Surgery told the press. “Cost effective, single surgery cartilage repair devices are not currently available in the USA, and Chondro-Gide will fill this unmet clinical need.”