Sam Brusco, Associate Editor02.03.22
Exactech has received U.S. Food and Drug Administration (FDA) 510(k) clearance for version two of its Equinoxe Planning App for shoulder surgery.
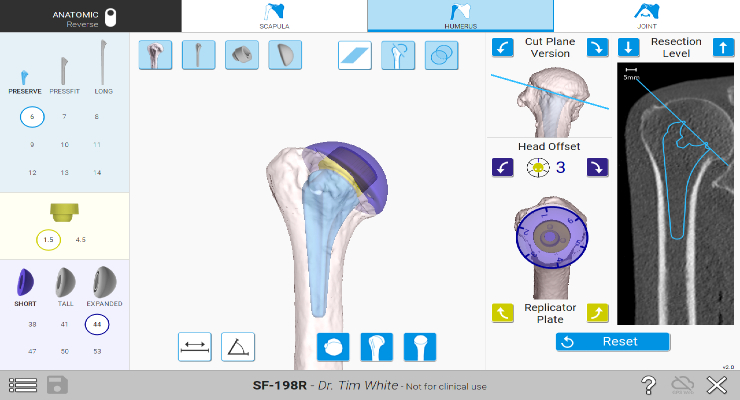
The latest version allows pre-operative planning of humeral implant options to model best fit for unique anatomies. Shoulder range of motion and impingement can be assessed using the tool, and it offers visual enhancements to boost clarity and ease of use.
“Exactech is driven to create new solutions that enhance our surgeons' surgical experiences,” Exactech senior VP of Extremities Chris Roche told the press. “The Equinoxe Planning App's newest update helps surgeons plan and select various implant types and sizes for the scapula and humerus based on a patient's specific boney morphology to facilitate planning of impingement-free movement."
The new smart shoulder technology is planned to be available in the U.S. in this year’s second quarter, and international market clearance is targeted for the third quarter.
The latest version allows pre-operative planning of humeral implant options to model best fit for unique anatomies. Shoulder range of motion and impingement can be assessed using the tool, and it offers visual enhancements to boost clarity and ease of use.
“Exactech is driven to create new solutions that enhance our surgeons' surgical experiences,” Exactech senior VP of Extremities Chris Roche told the press. “The Equinoxe Planning App's newest update helps surgeons plan and select various implant types and sizes for the scapula and humerus based on a patient's specific boney morphology to facilitate planning of impingement-free movement."
The new smart shoulder technology is planned to be available in the U.S. in this year’s second quarter, and international market clearance is targeted for the third quarter.