Sam Brusco, Associate Editor02.17.23
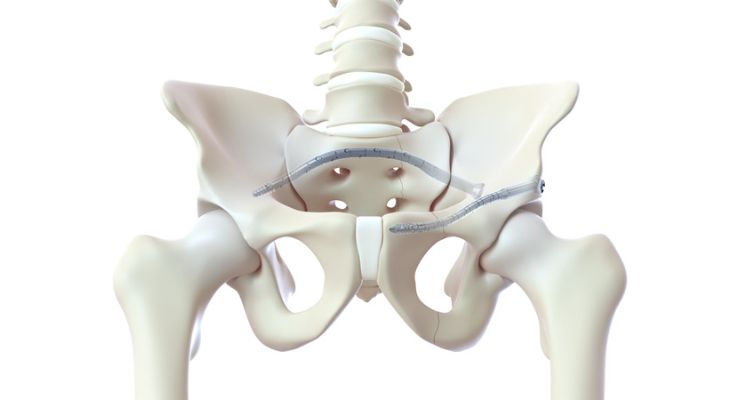
CurvaFix has launched its smaller diameter, 7.5 mm CurvaFix IM (intramedullary) implant to simplify surgery and provide strong, stable fixation in small-boned patients.
The company will showcase the new 7.5 mm intramedullary device, of which over two dozen have already been implanted in patients, at the American Academy of Orthopedic Surgeons (AAOS) 2023 Annual Meeting in Las Vegas, March 7-11.
“I’ve been pleasantly surprised with the flexibility of the CurvaFix devices, and I believe they will continue to play a role in pelvic fixation, especially in osteoporotic patients,” Brett Crist, MD, Director of Orthopaedic Trauma Service and Orthopaedic Trauma Fellowship at the University of Missouri in Columbia, Mo., told the press. “We recently treated a 63-year-old morbidly obese female (BMI 46.5) patient who sustained a low energy fall two years prior, resulting in pelvic fragility fractures. Her treatment had been non-operative management, leaving her wheelchair-dependent before eventually being referred to me. Based on her pre-operative CT scan, sacral dysmorphism, and bone quality, I thought the 7.5mm CurvaFix Implant would work well and that the maneuverable ball-tipped guide wire would aid us in being able to align the rami for fixation. Pleased with the stability that the 9.5mm CurvaFix device provides in osteoporotic patients, I wanted to use the smaller device to be able to place it in the smaller corridor.
“Overall, I was very pleased to be able to use the new 7.5mm CurvaFix devices for the left and right rami nonunions and the second sacral corridor while also being very satisfied with the smoothness of the anterior ring fixation,” continued Dr. Crist. “Following two years of being dependent on a wheelchair, this patient was discharged from the hospital with a walker within 24 hours of her CurvaFix procedure. Moreover, her visual analog scale for pain (VAS) was between an 8 and 9 prior to her surgery, and within 12 hours of the procedure, her VAS pain score was down to a 3.”
According to the company, over 175 patients have undergone the CurvaFix procedure to date. The patient population demonstrated surgical utility and potential benefits in a variety of pelvic injuries and conditions, including polytrauma patients with multiple complex injuries, FFP patients with weak bone, patients with dysmorphic bony anatomy, oncology patients with pelvic fractures, revision surgery for failed pelvic fracture fixation, and patients with impeding total hip or lumbosacral spinal hardware.
“The new 7.5mm device is designed to simplify surgery and provide strong, stable, curved fixation in smaller patients,” said Steve Dimmer, CEO of CurvaFix. “Additionally, our novel device has been shown to offer many geriatric patients immediate pain relief and early mobility, which is critically important in older patients where mobility is such a key to life.”
The company will showcase the new 7.5 mm intramedullary device, of which over two dozen have already been implanted in patients, at the American Academy of Orthopedic Surgeons (AAOS) 2023 Annual Meeting in Las Vegas, March 7-11.
“I’ve been pleasantly surprised with the flexibility of the CurvaFix devices, and I believe they will continue to play a role in pelvic fixation, especially in osteoporotic patients,” Brett Crist, MD, Director of Orthopaedic Trauma Service and Orthopaedic Trauma Fellowship at the University of Missouri in Columbia, Mo., told the press. “We recently treated a 63-year-old morbidly obese female (BMI 46.5) patient who sustained a low energy fall two years prior, resulting in pelvic fragility fractures. Her treatment had been non-operative management, leaving her wheelchair-dependent before eventually being referred to me. Based on her pre-operative CT scan, sacral dysmorphism, and bone quality, I thought the 7.5mm CurvaFix Implant would work well and that the maneuverable ball-tipped guide wire would aid us in being able to align the rami for fixation. Pleased with the stability that the 9.5mm CurvaFix device provides in osteoporotic patients, I wanted to use the smaller device to be able to place it in the smaller corridor.
“Overall, I was very pleased to be able to use the new 7.5mm CurvaFix devices for the left and right rami nonunions and the second sacral corridor while also being very satisfied with the smoothness of the anterior ring fixation,” continued Dr. Crist. “Following two years of being dependent on a wheelchair, this patient was discharged from the hospital with a walker within 24 hours of her CurvaFix procedure. Moreover, her visual analog scale for pain (VAS) was between an 8 and 9 prior to her surgery, and within 12 hours of the procedure, her VAS pain score was down to a 3.”
According to the company, over 175 patients have undergone the CurvaFix procedure to date. The patient population demonstrated surgical utility and potential benefits in a variety of pelvic injuries and conditions, including polytrauma patients with multiple complex injuries, FFP patients with weak bone, patients with dysmorphic bony anatomy, oncology patients with pelvic fractures, revision surgery for failed pelvic fracture fixation, and patients with impeding total hip or lumbosacral spinal hardware.
“The new 7.5mm device is designed to simplify surgery and provide strong, stable, curved fixation in smaller patients,” said Steve Dimmer, CEO of CurvaFix. “Additionally, our novel device has been shown to offer many geriatric patients immediate pain relief and early mobility, which is critically important in older patients where mobility is such a key to life.”