Michael Barbella, Managing Editor06.12.23
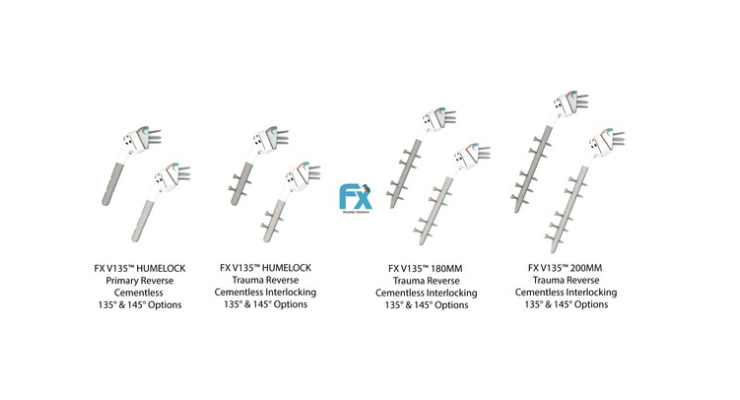
FX has received U.S. Food and Drug Administration 510(k) clearance to launch more distal interlocking stem options. FX adds the lengths 120mm, 180mm and 200mm cementless interlocking stems to further complement the previously cleared 70mm (not interlocking) FX V135 mini.
FX now offers a range of sizes for its interlocking humeral stems with intraoperative options for the surgeon to decide between 135 degree and 145 degree neck shaft angle in the reverse construct. Surgeons have this choice—to change the neck shaft angle, even at the last minute before reducing the joint—thanks to FX's net shape molded humeral cups. FX's previously cleared Humelock Reversed and Humelock II shoulder systems are supported by published data to demonstrate the efficacy and advantage these interlocking cementless stems provide.
"We anticipate these additions to our portfolio will generate a lot of excitement as we bring additional interlocking cementless humeral stems to a complex and challenging corner of the shoulder market," FX CEO Baptiste Martin said. "When you add to that the intraoperative option to decide between 135 degrees and 145 degrees by simply changing the one-piece FX humeral component, it is truly a shoulder system like no other on the market. We continue to stay true to our identity as shoulder specialists focused exclusively and solely on shoulder arthroplasty. This additional 510(k) clearance is really a game changer for the shoulder market and for the surgeon to be able to treat their patients differently."
The 120mm FX V135 Humelock allows surgeons to use 135 degree or 145 degree neck shaft angle on primary reverse procedures as well as three to four parts fractures with the same stem. This interlocking stem is completely unique in the shoulder arthroplasty market as no other company can provide this option.
The 180 and 200mm FX V135 interlocking humeral long stems are the first-to-market and expand the FX portfolio to provide cementless stems for complex fractures. Likewise, these stems may be used in a revision setting providing a novel shoulder solution to surgeons.
With the FX V135 shoulder system, the surgeon may trial on the broach as well as the definitive stem at both 135 degree and 145 degree neck shaft angles.
The additional FX V135 stems feature polished suture holes proximally for soft tissue repair.
The FX glenoid reverse system remains the same along the portfolio with a total of 12 baseplate options and six different glenosphere options (sizes 32mm, 36mm, and 40mm in centered or eccentric styles).
The FX V135 shoulder system offers wideranging options to create a solution that adapts the system to the patient's anatomy rather than making the patient conform to the components.
FX operates a U.S. headquarters in Dallas, Texas, and an outside-U.S. headquarters in Viriat, France.
FX now offers a range of sizes for its interlocking humeral stems with intraoperative options for the surgeon to decide between 135 degree and 145 degree neck shaft angle in the reverse construct. Surgeons have this choice—to change the neck shaft angle, even at the last minute before reducing the joint—thanks to FX's net shape molded humeral cups. FX's previously cleared Humelock Reversed and Humelock II shoulder systems are supported by published data to demonstrate the efficacy and advantage these interlocking cementless stems provide.
"We anticipate these additions to our portfolio will generate a lot of excitement as we bring additional interlocking cementless humeral stems to a complex and challenging corner of the shoulder market," FX CEO Baptiste Martin said. "When you add to that the intraoperative option to decide between 135 degrees and 145 degrees by simply changing the one-piece FX humeral component, it is truly a shoulder system like no other on the market. We continue to stay true to our identity as shoulder specialists focused exclusively and solely on shoulder arthroplasty. This additional 510(k) clearance is really a game changer for the shoulder market and for the surgeon to be able to treat their patients differently."
The 120mm FX V135 Humelock allows surgeons to use 135 degree or 145 degree neck shaft angle on primary reverse procedures as well as three to four parts fractures with the same stem. This interlocking stem is completely unique in the shoulder arthroplasty market as no other company can provide this option.
The 180 and 200mm FX V135 interlocking humeral long stems are the first-to-market and expand the FX portfolio to provide cementless stems for complex fractures. Likewise, these stems may be used in a revision setting providing a novel shoulder solution to surgeons.
With the FX V135 shoulder system, the surgeon may trial on the broach as well as the definitive stem at both 135 degree and 145 degree neck shaft angles.
The additional FX V135 stems feature polished suture holes proximally for soft tissue repair.
The FX glenoid reverse system remains the same along the portfolio with a total of 12 baseplate options and six different glenosphere options (sizes 32mm, 36mm, and 40mm in centered or eccentric styles).
The FX V135 shoulder system offers wideranging options to create a solution that adapts the system to the patient's anatomy rather than making the patient conform to the components.
FX operates a U.S. headquarters in Dallas, Texas, and an outside-U.S. headquarters in Viriat, France.