Sam Brusco, Associate Editor06.12.23
Smith+Nephew has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Aetos shoulder system.
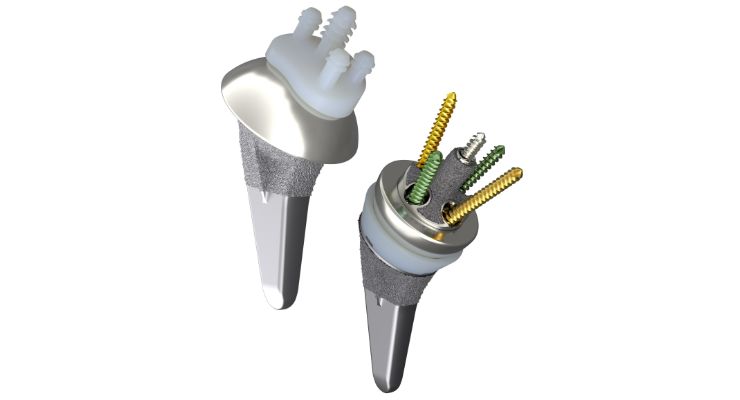
Designed to restore range of motion and minimize arthritic shoulder pain, Aetos feature a Meta Stem that was engineered to maximize stability, preserve bone, and maintain patient anatomy. Its indicated for anatomic and reverse total shoulder arthroplasty.
Aetos also requires fewer steps for conversion and fewer instruments for primary anatomic and reverse surgeries.
Dr. James Kelly, shoulder and elbow specialist at California Pacific Orthopaedics in San Francisco and AETOS System design surgeon, told the press, "The AETOS Shoulder System was designed to be a cutting-edge press fit, bone conserving, convertible humeral stem. The system is designed for surgeons who desire maximum flexibility to reconstruct the humerus and glenoid, using an efficient and intuitive system that helps prioritize patient outcomes."
"Receiving FDA clearance for the AETOS Shoulder System is a major milestone for Smith+Nephew. This platform is the culmination of years of research and development and represents our commitment to providing healthcare professionals with the best possible technology for their patients. We are confident that this platform will further evolve the standard of care for shoulder surgery and are excited to see the impact it has on patient outcomes," added Brad Cannon, president Global Orthopaedics for Smith+Nephew.
In May, Smith+Nephew introduced both RI.KNEE Robotics 2.0 with personalized planning and the RI.INSIGHTS data visualization platform.
Designed to restore range of motion and minimize arthritic shoulder pain, Aetos feature a Meta Stem that was engineered to maximize stability, preserve bone, and maintain patient anatomy. Its indicated for anatomic and reverse total shoulder arthroplasty.
Aetos also requires fewer steps for conversion and fewer instruments for primary anatomic and reverse surgeries.
Dr. James Kelly, shoulder and elbow specialist at California Pacific Orthopaedics in San Francisco and AETOS System design surgeon, told the press, "The AETOS Shoulder System was designed to be a cutting-edge press fit, bone conserving, convertible humeral stem. The system is designed for surgeons who desire maximum flexibility to reconstruct the humerus and glenoid, using an efficient and intuitive system that helps prioritize patient outcomes."
"Receiving FDA clearance for the AETOS Shoulder System is a major milestone for Smith+Nephew. This platform is the culmination of years of research and development and represents our commitment to providing healthcare professionals with the best possible technology for their patients. We are confident that this platform will further evolve the standard of care for shoulder surgery and are excited to see the impact it has on patient outcomes," added Brad Cannon, president Global Orthopaedics for Smith+Nephew.
In May, Smith+Nephew introduced both RI.KNEE Robotics 2.0 with personalized planning and the RI.INSIGHTS data visualization platform.