Sam Brusco, Associate Editor07.31.23
Axial3D, a firm focused on medical segmentation and 3D solutions, has gained U.S. Food and Drug Administration (FDA) clearance for its automated, artificial intelligence (AI)-driven, cloud-based segmentation platform for orthopedic trauma, orthopedic, maxillofacial, and cardiovascular applications.
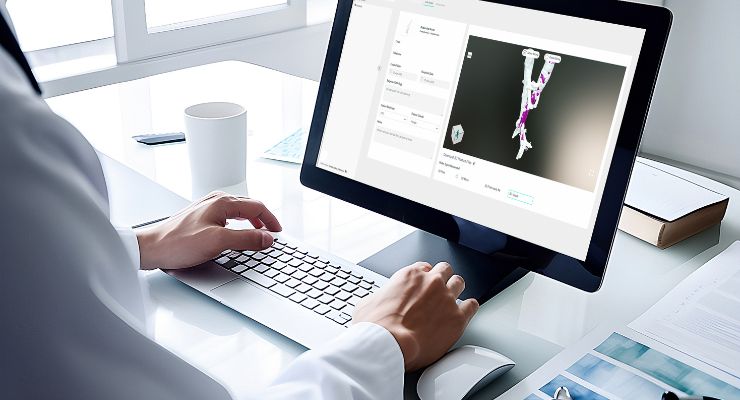
Axial3D INSIGHT automates conversion of 2D DICOM images like CT and MRI scans into accurate 3D visualizations, 3D print-ready files, 3D mesh files, or 3D-printed anatomical models, made with Stratasys 3D printing technology. Automating manual or semi-manual image segmentation, according to the company, enables addressing more applications while saving hours or days per case, and reducing capital expenditures.
"Our FDA clearance for Axial3D INSIGHT is a testament to how far Axial3D has come," Dan Crawford, founder and CSO of Axial3D, told the press. "From our humble beginnings as a startup to now being recognized as a leading medical technology company, this achievement showcases our dedication to pushing the boundaries of innovation in healthcare. We are immensely proud of our team's commitment in delivering exceptional patient care using advanced automation, artificial intelligence, and machine learning technologies."
The 3D data generated can be used to design personalized devices and surgical kits that include surgical plans, models, and surgical guides that can be 3D printed on a variety of printers. Stratasys led an investment round late last year to advance Axial3D’s tech and bring a joint solution to market for healthcare providers and medical device manufacturers.
"Stratasys is proud to partner with Axial3D in driving innovation and in transforming the healthcare landscape with 3D solutions that help make personalized healthcare at scale possible," said Stratasys CEO Dr. Yoav Zeif. "We congratulate Axial3D on achieving FDA clearance for their segmentation platform, leveraging the power of automation and AI as a key business driver for their continued success in the medical sector."
Axial3D INSIGHT automates conversion of 2D DICOM images like CT and MRI scans into accurate 3D visualizations, 3D print-ready files, 3D mesh files, or 3D-printed anatomical models, made with Stratasys 3D printing technology. Automating manual or semi-manual image segmentation, according to the company, enables addressing more applications while saving hours or days per case, and reducing capital expenditures.
"Our FDA clearance for Axial3D INSIGHT is a testament to how far Axial3D has come," Dan Crawford, founder and CSO of Axial3D, told the press. "From our humble beginnings as a startup to now being recognized as a leading medical technology company, this achievement showcases our dedication to pushing the boundaries of innovation in healthcare. We are immensely proud of our team's commitment in delivering exceptional patient care using advanced automation, artificial intelligence, and machine learning technologies."
The 3D data generated can be used to design personalized devices and surgical kits that include surgical plans, models, and surgical guides that can be 3D printed on a variety of printers. Stratasys led an investment round late last year to advance Axial3D’s tech and bring a joint solution to market for healthcare providers and medical device manufacturers.
"Stratasys is proud to partner with Axial3D in driving innovation and in transforming the healthcare landscape with 3D solutions that help make personalized healthcare at scale possible," said Stratasys CEO Dr. Yoav Zeif. "We congratulate Axial3D on achieving FDA clearance for their segmentation platform, leveraging the power of automation and AI as a key business driver for their continued success in the medical sector."