Sam Brusco, Associate Editor08.08.23
Safe Orthopaedics, a company focused on ready-to-use devices for spine surgery (especially for emergency vertebral fracture) announced publication of a biomechanical study of its Sycamore device and over 160 patients treated with it.
The study was led by professor Jean-Charles Le Huec. It compared the biomechanical behavior of vertebrae treated with “Dowelplasty,” a new surgical technique that uses the Sycamore pedicle anchored device, with a traditional kyphoplasty system.
The study was published in the Journal of Experimental Orthopedics.
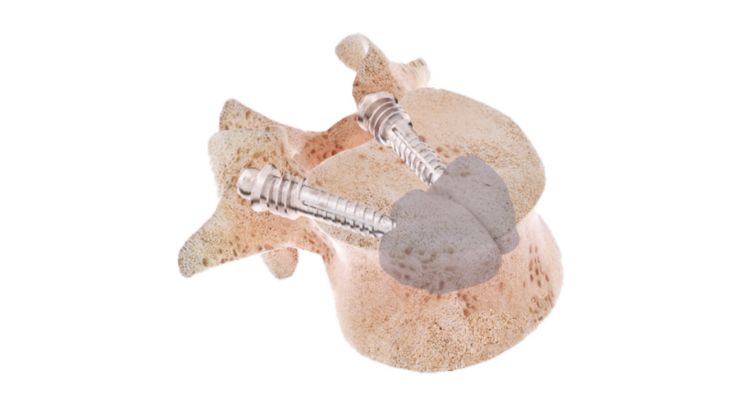
Sycamore is comprised of a cannulated titanium nail and titanium dowel anchored in the pedicle. The cannulated nail is inserted and locked into the dowel, then cement is injected through the nail.
The study showed a “significant increase” of compression strength for patients using Sycamore (373 N ; IC à 95% ; – 331 N versus – 1076 N for Sycamore). It also demonstrated “very significant” increase in resistance to load of the fractures compared to the pre-treatment.
Sycamore also showed greater improvement in load resistance and fracture displacement energy than balloon kyphoplasty and traditional kyphoplasty. The results suggest Sycamore could be a good alternative to treat vertebral compression fractures.
Professor Le Huec confirmed that with a follow-up of two years, losses of corrections don’t exceed 2.1° compared to immediate post-op results.
Pierre Dumouchel, chairman and CEO of Safe Group told the press, “First of all, we would like to thank Professor Jean-Charles Le Huec and all the surgeons who contributed to the development and clinical follow-up of Sycamore, a unique and patented Safe Orthopaedics technology for better management of patients with vertebral fractures. We have treated over 160 patients with our Sycamore technologies and are seeing very promising clinical results. Through recent listings in many French centers and international deployment, we are delighted to be able to benefit a large number of patients from the clinical benefits already observed and aim to accelerate the Safe Orthopaedics sales.”
The study was led by professor Jean-Charles Le Huec. It compared the biomechanical behavior of vertebrae treated with “Dowelplasty,” a new surgical technique that uses the Sycamore pedicle anchored device, with a traditional kyphoplasty system.
The study was published in the Journal of Experimental Orthopedics.
Sycamore is comprised of a cannulated titanium nail and titanium dowel anchored in the pedicle. The cannulated nail is inserted and locked into the dowel, then cement is injected through the nail.
The study showed a “significant increase” of compression strength for patients using Sycamore (373 N ; IC à 95% ; – 331 N versus – 1076 N for Sycamore). It also demonstrated “very significant” increase in resistance to load of the fractures compared to the pre-treatment.
Sycamore also showed greater improvement in load resistance and fracture displacement energy than balloon kyphoplasty and traditional kyphoplasty. The results suggest Sycamore could be a good alternative to treat vertebral compression fractures.
Professor Le Huec confirmed that with a follow-up of two years, losses of corrections don’t exceed 2.1° compared to immediate post-op results.
Pierre Dumouchel, chairman and CEO of Safe Group told the press, “First of all, we would like to thank Professor Jean-Charles Le Huec and all the surgeons who contributed to the development and clinical follow-up of Sycamore, a unique and patented Safe Orthopaedics technology for better management of patients with vertebral fractures. We have treated over 160 patients with our Sycamore technologies and are seeing very promising clinical results. Through recent listings in many French centers and international deployment, we are delighted to be able to benefit a large number of patients from the clinical benefits already observed and aim to accelerate the Safe Orthopaedics sales.”