Sam Brusco, Associate Editor09.05.23
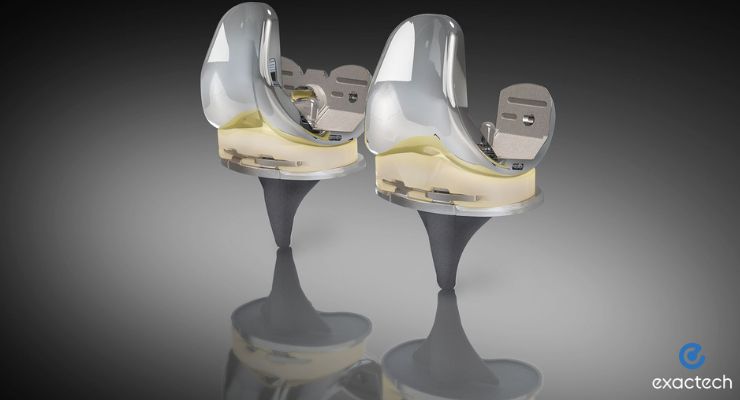
Exactech announced the first cases finished using its TriVerse primary knee, which was designed to address the demands of today’s active knee population.
Orthopedic surgeons Chris Christensen, M. of Bluegrass Orthopaedics in Lexington, Ky. and David Taunton, Jr., MD, of Texas Orthopedic Specialists in Bedford, Texas, were the first to perform TriVerse knee surgeries. TriVerse gained U.S. Food and Drug Administration (FDA) 510(k) clearance in November 2022.
TriVerse has anterior stabilized and posterior stabilized constructs and is exclusively available with highly crosslinked vitamin E polyethylene. The implants will launch alongside a set of mechanical instruments that aim to boost the efficiency needed in same-day surgical environments.
“As a high-volume surgeon in the hospital and surgery center settings, efficiency and flexibility are of paramount importance to me and my O.R. staff,” Dr. Christensen told the press. “Exactech’s TriVerse Knee system has been designed with a streamlined, simplified approach while offering a comprehensive array of primary knee implants that give me the best of both worlds.”
“For more than three decades, Exactech has continued to push forward total knee replacement innovations that are built on time-tested results,” added Adam Hayden, CMO and SVP, Large Joints Business Unit at Exactech. “TriVerse adds to the company’s advanced portfolio of knee solutions, including the award-winning Truliant primary and revision systems and integrated Newton ligament balancing platform, providing yet another tried-and-true system that helps address the challenging health landscape.”
Exactech said TriVerse has limited availability at present but has full launch slated for 2024.
Orthopedic surgeons Chris Christensen, M. of Bluegrass Orthopaedics in Lexington, Ky. and David Taunton, Jr., MD, of Texas Orthopedic Specialists in Bedford, Texas, were the first to perform TriVerse knee surgeries. TriVerse gained U.S. Food and Drug Administration (FDA) 510(k) clearance in November 2022.
TriVerse has anterior stabilized and posterior stabilized constructs and is exclusively available with highly crosslinked vitamin E polyethylene. The implants will launch alongside a set of mechanical instruments that aim to boost the efficiency needed in same-day surgical environments.
“As a high-volume surgeon in the hospital and surgery center settings, efficiency and flexibility are of paramount importance to me and my O.R. staff,” Dr. Christensen told the press. “Exactech’s TriVerse Knee system has been designed with a streamlined, simplified approach while offering a comprehensive array of primary knee implants that give me the best of both worlds.”
“For more than three decades, Exactech has continued to push forward total knee replacement innovations that are built on time-tested results,” added Adam Hayden, CMO and SVP, Large Joints Business Unit at Exactech. “TriVerse adds to the company’s advanced portfolio of knee solutions, including the award-winning Truliant primary and revision systems and integrated Newton ligament balancing platform, providing yet another tried-and-true system that helps address the challenging health landscape.”
Exactech said TriVerse has limited availability at present but has full launch slated for 2024.