Sam Brusco, Associate Editor10.05.23
Biotronik released results from its BENEFIT-02 trial to clinically evaluate its RESONANCE multiphase stimulation, which is used in its Prospera spinal cord stimulation (SCS) system to treat chronic pain patients.
Results of the study were published in the journal Neuromodulation.
The 65-patient feasibility study included patients with chronic low back and/or leg pain and baseline numerical rating scale (NRS) for overall pain intensity ≥6. After a commercial SCS trial, subjects were randomized to multiphase SCS therapy (A: about 600-1,500 Hz or B: about 300-600 Hz). After washout an investigational external pulse generator and existing leads were used to delivery therapy during an 11-12 day period.
Biotronik reported “no statistically significant difference” in average NRS reduction or percent pain relief between the two multiphase therapies. In the home setting, 63.9% of participants reported greater pain relief with multiphase than commercial SCS therapy, in addition to increased average sleep quality and physical activity.
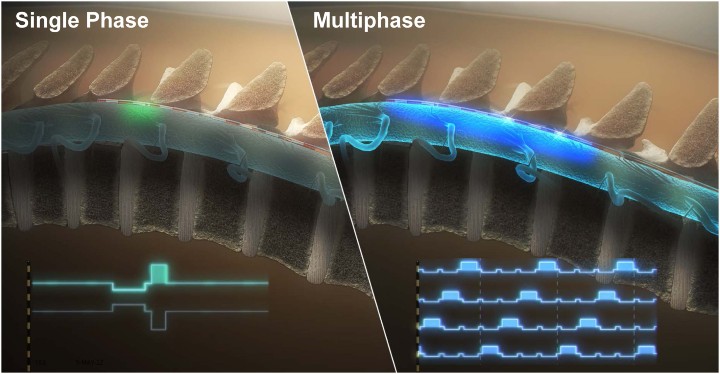
Single phase commercial stimulation vs. multiphase stimulation.
The Prospera SCS received U.S. Food and Drug Administration (FDA) approval in March 2023. In addition to RESONANCE multiphase stimulation, it includes the company’s Embrace One model for automatic, objective, daily remote monitoring.
The company’s BENEFIT-03 study is evaluating long-term Prospera SCS with RESONANCE stimulation use, as well as its automatic, daily, and objective device monitoring.
“As we await additional data from our BENEFIT-03 study, we're excited to see these initial data on the long-term effects of our SCS system,” BIOTRONIK Neuro’s president Todd Langevin told the press. “We developed Prospera to offer patients sustainable pain relief – and we believe its proactive care model will have a clinically meaningful impact on lowering long-term failure rates and reducing the service burden of SCS.”
Results of the study were published in the journal Neuromodulation.
The 65-patient feasibility study included patients with chronic low back and/or leg pain and baseline numerical rating scale (NRS) for overall pain intensity ≥6. After a commercial SCS trial, subjects were randomized to multiphase SCS therapy (A: about 600-1,500 Hz or B: about 300-600 Hz). After washout an investigational external pulse generator and existing leads were used to delivery therapy during an 11-12 day period.
Biotronik reported “no statistically significant difference” in average NRS reduction or percent pain relief between the two multiphase therapies. In the home setting, 63.9% of participants reported greater pain relief with multiphase than commercial SCS therapy, in addition to increased average sleep quality and physical activity.

Single phase commercial stimulation vs. multiphase stimulation.
The Prospera SCS received U.S. Food and Drug Administration (FDA) approval in March 2023. In addition to RESONANCE multiphase stimulation, it includes the company’s Embrace One model for automatic, objective, daily remote monitoring.
The company’s BENEFIT-03 study is evaluating long-term Prospera SCS with RESONANCE stimulation use, as well as its automatic, daily, and objective device monitoring.
“As we await additional data from our BENEFIT-03 study, we're excited to see these initial data on the long-term effects of our SCS system,” BIOTRONIK Neuro’s president Todd Langevin told the press. “We developed Prospera to offer patients sustainable pain relief – and we believe its proactive care model will have a clinically meaningful impact on lowering long-term failure rates and reducing the service burden of SCS.”













