Sam Brusco, Associate Editor10.09.23
DePuy Synthes has secured U.S. Food and Drug Administration (FDA) 510(k) clearances for its TriALTIS spine system and TriALTIS navigation-enabled instruments.
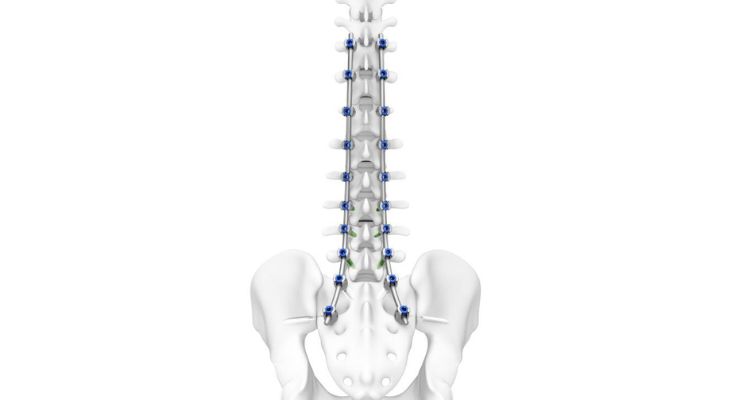
The TriALTIS spine system is a next-gen posterior thoracolumbar pedicle screw system with a comprehensive implant portfolio and advanced instrumentation designed to be integrated with enabling technology. The TriALTIS navigation-enabled instruments include drills, taps, and screwdrivers that can either be operated manually or under power for navigated and non-navigated use.
DePuy Synthes said the system aims to address unmet clinical needs and help achieve more consistent outcomes in treating complex spine conditions—degenerative, tumor, trauma, and deformity pathologies included.
"The TriALTIS Spine System will be our premier pedicle screw platform designed to work seamlessly across our entire DePuy Synthes portfolio of enabling technology solutions," DePuy Synthes’ worldwide president of Spine Russell Powers told the press. “Johnson & Johnson MedTech is proud to deliver this exciting innovation that can help facilitate the surgical treatment of spinal degeneration and deformity with the goal to improve fixation, drive better patient outcomes and potentially reduce the clinical and economic burden associated with the current standard of care. DePuy Synthes has been a leader in the complex spine space for over 35 years, and the launch of the TriALTIS Spine System will mark a critical milestone towards our vision to continue this legacy long into the future.”
The company said it aims to launch the TriALTIS spine system in the U.S. in 2024.
The TriALTIS spine system is a next-gen posterior thoracolumbar pedicle screw system with a comprehensive implant portfolio and advanced instrumentation designed to be integrated with enabling technology. The TriALTIS navigation-enabled instruments include drills, taps, and screwdrivers that can either be operated manually or under power for navigated and non-navigated use.
DePuy Synthes said the system aims to address unmet clinical needs and help achieve more consistent outcomes in treating complex spine conditions—degenerative, tumor, trauma, and deformity pathologies included.
"The TriALTIS Spine System will be our premier pedicle screw platform designed to work seamlessly across our entire DePuy Synthes portfolio of enabling technology solutions," DePuy Synthes’ worldwide president of Spine Russell Powers told the press. “Johnson & Johnson MedTech is proud to deliver this exciting innovation that can help facilitate the surgical treatment of spinal degeneration and deformity with the goal to improve fixation, drive better patient outcomes and potentially reduce the clinical and economic burden associated with the current standard of care. DePuy Synthes has been a leader in the complex spine space for over 35 years, and the launch of the TriALTIS Spine System will mark a critical milestone towards our vision to continue this legacy long into the future.”
The company said it aims to launch the TriALTIS spine system in the U.S. in 2024.