Michael Barbella, Managing Editor12.12.23
ZimVie Inc. has received U.S. Food and Drug Administration (FDA) clearance for its Vital Spinal Fixation System, including instruments for use with Brainlab Spine & Trauma Navigation. The clearance follows execution of the companies’ Development Cooperation Agreement, announced in March 2023, to achieve compatibility between ZimVie’s Vital and Virage systems and Brainlab Spine & Trauma Navigation, which helps surgeons plan and execute spinal procedures, accurately place pedicle screws, and minimize radiation exposure.
ZimVie plans to co-market Brainlab Spine & Trauma Navigation alongside its Vital and Virage lines, with first Vital sets expected to be launched in the United States early next y ear. Having secured that clearance, the company plans to submit a 510(k) application to the FDA for Virage in 2024 as well.
“This is a positive milestone for both teams and the first of what we hope will be many FDA clearances for compatibility. The strong collaboration between our organizations resulted in an expedited project timeline,” ZimVie Spine Global President Rebecca Whitney said. “We have been focused on expanding our portfolio with enabling technologies to drive greater adoption, and I am excited to see the team advance toward the launch.”
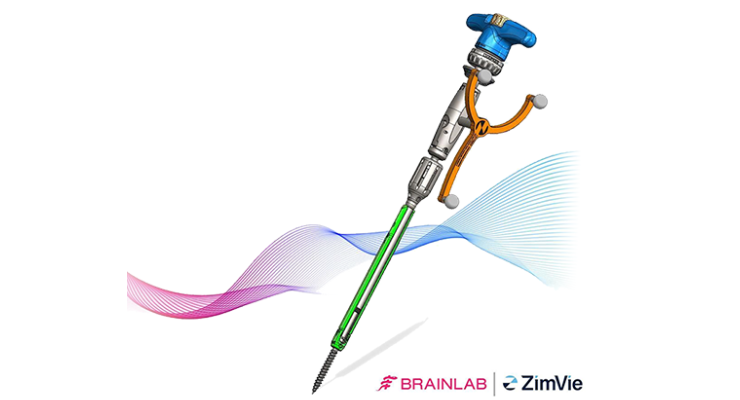
ZimVie's Vital Spinal Fixation System includes an optimized instrument and implant kit configuration for complex and degenerative thoracolumbar procedures designed to provide surgeons with the flexibility to utilize instrumentation based on their personal technique, preference and specific patient needs. A series of instruments and reference arrays designed to be compatible with Brainlab spine and trauma navigation technologies allows navigation during bone preparation and pedicle screw placement during spinal surgery to assist the surgeon in precisely locating anatomical structures in either open or minimally invasive procedures.
Brainlab digitizes medical workflows, from diagnosis to therapy, to offer clinicians and patients better treatment possibilities. Its digital ecosystem forms the basis for modern healthcare technology in 6,300 hospitals in 120 countries. Munich-based Brainlab employs around 2,200 people with expertise across the entire healthcare value chain in 25 locations worldwide.
ZimVie develops, manufactures, and delivers products and solutions designed to treat various spine pathologies and support dental tooth replacement and restoration procedures. The company was founded in March 2022 as an independent, publicly traded spin-off of the Dental and Spine business units of Zimmer Biomet to breathe new life, dedicated energy, and strategic focus to its portfolio of trusted brands and products. From its headquarters in Westminster, Colo., and additional facilities worldwide, the company serves customers in more than 70 countries worldwide with dental and spine solutions including differentiated product platforms supported by extensive clinical evidence.
ZimVie plans to co-market Brainlab Spine & Trauma Navigation alongside its Vital and Virage lines, with first Vital sets expected to be launched in the United States early next y ear. Having secured that clearance, the company plans to submit a 510(k) application to the FDA for Virage in 2024 as well.
“This is a positive milestone for both teams and the first of what we hope will be many FDA clearances for compatibility. The strong collaboration between our organizations resulted in an expedited project timeline,” ZimVie Spine Global President Rebecca Whitney said. “We have been focused on expanding our portfolio with enabling technologies to drive greater adoption, and I am excited to see the team advance toward the launch.”
ZimVie's Vital Spinal Fixation System includes an optimized instrument and implant kit configuration for complex and degenerative thoracolumbar procedures designed to provide surgeons with the flexibility to utilize instrumentation based on their personal technique, preference and specific patient needs. A series of instruments and reference arrays designed to be compatible with Brainlab spine and trauma navigation technologies allows navigation during bone preparation and pedicle screw placement during spinal surgery to assist the surgeon in precisely locating anatomical structures in either open or minimally invasive procedures.
Brainlab digitizes medical workflows, from diagnosis to therapy, to offer clinicians and patients better treatment possibilities. Its digital ecosystem forms the basis for modern healthcare technology in 6,300 hospitals in 120 countries. Munich-based Brainlab employs around 2,200 people with expertise across the entire healthcare value chain in 25 locations worldwide.
ZimVie develops, manufactures, and delivers products and solutions designed to treat various spine pathologies and support dental tooth replacement and restoration procedures. The company was founded in March 2022 as an independent, publicly traded spin-off of the Dental and Spine business units of Zimmer Biomet to breathe new life, dedicated energy, and strategic focus to its portfolio of trusted brands and products. From its headquarters in Westminster, Colo., and additional facilities worldwide, the company serves customers in more than 70 countries worldwide with dental and spine solutions including differentiated product platforms supported by extensive clinical evidence.