Sam Brusco, Associate Editor02.12.24
Mainstay Medical has gained full-body MRI conditional labeling approval from the U.S. Food and Drug Administration (FDA) for its ReActiv8 neurostimulation system.
The approval covers current and future ReActiv8 patients in the U.S. implanted with the current, commercially available 45 cm leads. Patients with ReActiv8 can now consult an FDA MRI Guidelines manual to have 1.5T full-body MRI scans.
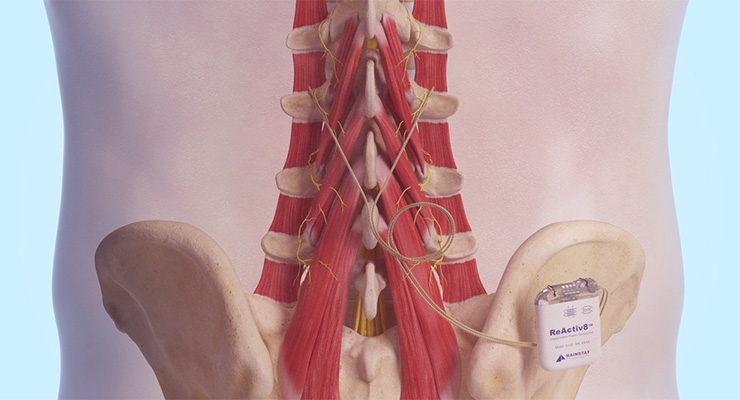
The ReActiv8 implant is used to treat adults with intractable chronic low back pain (CLBP) associated with multifidus muscle dysfunction. Its restorative neurostimulation targets the nerves of the muscle that stabilize the lumbar spine. The neurostimulation overrides underlying multifidus inhibition by eliciting episodic, isolated contractions which, over time, can facilitate recovery from mechanical CLBP, the company says.
Mainstay Medical released ReActiv8 in the U.S. in June 2021.
“This approval expands the existing safety profile of ReActiv8, broadening access to patients who may need (or develop the need) for MRI imaging after implant,” said Mainstay Medical CEO Jason Hannon. “The ReActiv8 MRI labeling is one of the most comprehensive among neurostimulation devices approved for chronic low back pain, with full-body imaging at 1.5T at normal operating mode (maximum specific absorption rate (SAR) of 3.2 W/kg for the head and 2.0 W/kg for the rest of the body). We look forward to building on this as we seek conditional MRI compatibility in Europe and Australia.”
In August, the company completed enrollment in its RESTORE clinical study, which is comparing ReActiv8 restorative neurostimulation therapy to current care paradigms to treat chronic, intractable low back pain.
The approval covers current and future ReActiv8 patients in the U.S. implanted with the current, commercially available 45 cm leads. Patients with ReActiv8 can now consult an FDA MRI Guidelines manual to have 1.5T full-body MRI scans.
The ReActiv8 implant is used to treat adults with intractable chronic low back pain (CLBP) associated with multifidus muscle dysfunction. Its restorative neurostimulation targets the nerves of the muscle that stabilize the lumbar spine. The neurostimulation overrides underlying multifidus inhibition by eliciting episodic, isolated contractions which, over time, can facilitate recovery from mechanical CLBP, the company says.
Mainstay Medical released ReActiv8 in the U.S. in June 2021.
“This approval expands the existing safety profile of ReActiv8, broadening access to patients who may need (or develop the need) for MRI imaging after implant,” said Mainstay Medical CEO Jason Hannon. “The ReActiv8 MRI labeling is one of the most comprehensive among neurostimulation devices approved for chronic low back pain, with full-body imaging at 1.5T at normal operating mode (maximum specific absorption rate (SAR) of 3.2 W/kg for the head and 2.0 W/kg for the rest of the body). We look forward to building on this as we seek conditional MRI compatibility in Europe and Australia.”
In August, the company completed enrollment in its RESTORE clinical study, which is comparing ReActiv8 restorative neurostimulation therapy to current care paradigms to treat chronic, intractable low back pain.