Sam Brusco, Associate Editor02.13.24
Smith+Nephew announced full commercial availability of its Aetos shoulder system in the U.S., along with U.S. Food and Drug Administration (FDA) clearance for use with its AtlasPlan 3D planning software and patient-specific instrumentation for total shoulder arthroplasty.
The company made the announcement at this year’s American Academy of Orthopaedic Surgeons (AAOS) meeting. The Aetos shoulder earned FDA clearance in June 2023.
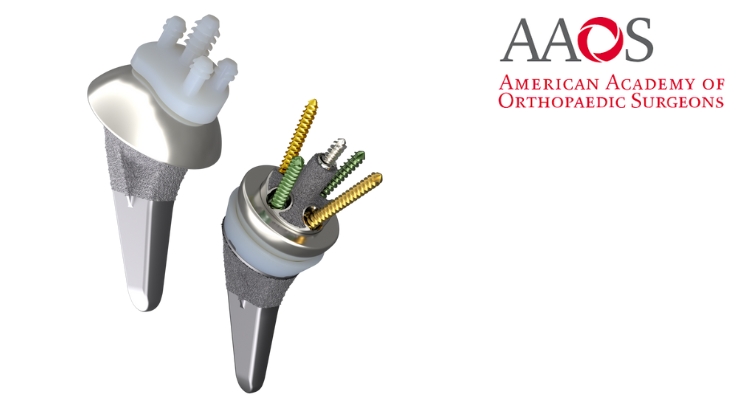
Aetos features the Meta stem, which the company said was designed for stability with metaphyseal fixation and an inlay collar, bone preservation, and to maintain patient anatomy. It’s indicated for both anatomic and reverse shoulder arthroplasty.
According to the company, it has fewer steps for conversion and fewer instruments for primary anatomic and reverse shoulder replacement. Designed to simplify operating room workflow, it can be of particular value in an ambulatory service center (ASC).
AtlasPlan allows pre-operative case planning and was developing in partnership with Materialise. It features an online, web-based planner that can be accessed from a computer or tablet and fast turnaround from image upload to planning. It also has an optional 3D-printed glenoid guide, with a patented coracoid clip for stability.
“After using the system for the past several months, I can say that the AETOS Shoulder System is a game changer,” said Dr. Charles Jobin, Orthopaedic Surgeon at Columbia University Irving Medical Center. “With the stability of its Meta Stem, the flexibility the system provides and streamlined instrumentation, it is a great solution not only benefitting patients but surgeons alike.”
“I’m thrilled with the stem fixation of the implant that the AETOS Shoulder System provides and believe it will be a noticeable, positive improvement for my patients,” added Dr. Matthew Ramsey, Shoulder and Elbow Specialist at Rothman Orthopaedics. “I’m also very pleased with the instrumentation—especially the glenoid reamer being so low profile and easy to use.”
The company made the announcement at this year’s American Academy of Orthopaedic Surgeons (AAOS) meeting. The Aetos shoulder earned FDA clearance in June 2023.
Aetos features the Meta stem, which the company said was designed for stability with metaphyseal fixation and an inlay collar, bone preservation, and to maintain patient anatomy. It’s indicated for both anatomic and reverse shoulder arthroplasty.
According to the company, it has fewer steps for conversion and fewer instruments for primary anatomic and reverse shoulder replacement. Designed to simplify operating room workflow, it can be of particular value in an ambulatory service center (ASC).
AtlasPlan allows pre-operative case planning and was developing in partnership with Materialise. It features an online, web-based planner that can be accessed from a computer or tablet and fast turnaround from image upload to planning. It also has an optional 3D-printed glenoid guide, with a patented coracoid clip for stability.
“After using the system for the past several months, I can say that the AETOS Shoulder System is a game changer,” said Dr. Charles Jobin, Orthopaedic Surgeon at Columbia University Irving Medical Center. “With the stability of its Meta Stem, the flexibility the system provides and streamlined instrumentation, it is a great solution not only benefitting patients but surgeons alike.”
“I’m thrilled with the stem fixation of the implant that the AETOS Shoulder System provides and believe it will be a noticeable, positive improvement for my patients,” added Dr. Matthew Ramsey, Shoulder and Elbow Specialist at Rothman Orthopaedics. “I’m also very pleased with the instrumentation—especially the glenoid reamer being so low profile and easy to use.”