Michael Barbella, Managing Editor02.14.24
Anika Therapeutics Inc. is showcasing recently-launched products in high growth segments of joint preservation and restoration from its Regenerative Solutions, Sports Medicine, and Arthrosurface Joint Solutions businesses during this year's American Academy of Orthopedic Surgeons (AAOS) 2024 Annual Meeting (Feb. 12-16) in San Francisco.
The company is featuring its differentiated shoulder portfolio, including the recently launched Integrity Implant System for rotator cuff repair, RevoMotion Reverse Shoulder Arthroplasty (RSA) System with newly launched AIM (Anika Implant Management) surgical planning software, X-Twist Fixation System with biocomposite suture anchor, OVOMotion with Inlay Glenoid Total Shoulder Arthroplasty (TSA) System.
Anika also announced at the show the limited launch of the X-Twist Fixation System with biocomposite suture anchor. X-Twist’s suture anchors enable fast and strong insertion into bone, requiring only 3.25 turns and offering significant torsional strength. The launch of the X-Twist Fixation System with biocomposite suture anchors now expands the X-Twist family portfolio with both permanent and resorbable material options.
“We’re thrilled to once again participate in the AAOS 2024 Annual Meeting and to showcase our advanced portfolio of regenerative, sports medicine and bone preserving implant solutions designed to meet the evolving needs of surgeons across the orthopedic continuum of care,” Anika President/CEO Cheryl R. Blanchard, Ph.D., said. “Anika is excited to highlight our recently launched Integrity Implant System, our newest regenerative hyaluronic acid (HA)-based scaffold for rotator cuff repair, and our X-Twist Fixation System with biocomposite suture anchor. We have also enhanced our shoulder arthroplasty offerings with the launch of AIM, Anika Implant Management, for 3D preoperative planning which is now available with both the RevoMotion Reverse Shoulder and OVOMotion Total Shoulder arthroplasty systems. These new products, and the continued combination of Anika’s advanced HA-based regenerative technology with our bone preserving implants and sports medicine solutions, provide surgeons with powerful tools and options to support their patients and help them return to active living.”
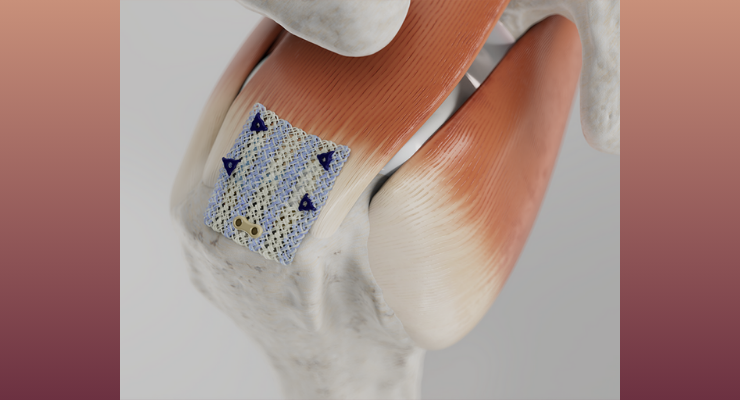
The Integrity Implant System, comprised of a hyaluronic acid-based and PET reinforced scaffold with bone and tendon fixation components and single-use arthroscopic delivery instruments, is designed to protect an injured tendon, promote healing, and support cell infiltration and regenerative healing in rotator cuff repair and other tendon procedures. The Integrity implant is inherently strong both when hydrated and dry and can be confidently manipulated arthroscopically. Integrity offers a differentiated solution for shoulder surgeons to treat rotator cuff tears. The flexible, knitted, porous HA-based scaffold provides greater regenerative capacity compared to first generation collagen patches1 and supports regenerative healing through improved cell infiltration, tissue remodeling, and tendon thickening.1 Early usage during the limited market release indicates high levels of interest and excitement for this product. The implant strength, staple and tissue tack fixation capabilities, unique “lateral first” implant technique, and ability to manipulate the implant are standout features of the Integrity system.
The RevoMotion Reverse Shoulder Arthroplasty System, with the industry’s smallest diameter threaded baseplate, offers surgeons and patients the ability to restore motion and preserve bone. Anika’s newly launched 3D preoperative planning software, AIM allows surgeons to personalize and pre-plan each surgery, including assessing erosion depth and bony defects, determining optimal implant position, and assessing the implant’s impact on range of motion.
AIM, formally launching at AAOS this year and now available in the United States, was developed in partnership with Materialise NV. AIM 3D preoperative planning software enables surgeons to generate powerful insights with both glenoid and humeral planning for Anika’s OVOMotion with Inlay Glenoid TSA and RevoMotion RSA Systems.
“Nearly a year into our experience with RevoMotion RSA, we are consistently seeing significant improvements in patient reported outcome measures and function,” said Christopher Baker, M.D., Florida Orthopaedic Institute, Tampa, Fla. “With the addition of AIM, a three-dimensional preoperative planning program, Anika is now able to offer a cutting-edge planning option for the system. This will allow physicians to more accurately manage difficult anatomic deformities of the glenohumeral joint in reverse shoulder arthroplasty.”
Several new Sports Medicine products will also be available in early 2024, including the ProPass Suture Passer, and other new shoulder instrumentation. The ProPass Suture Passer provides surgeons with an intuitive operation and consistent control that reliably passes suture through thick rotator cuff tissue and is offered with or without an auto-capture window. All are expected to launch in 2024.
AAOS attendees can learn more about Anika’s compelling products at the Anika booth 5145, as well as an Innovation Theater sessions held in booth 4003:
“Integrity: The Next Generation of Rotator Cuff Augmentation,” led by David Porter, M.D., with Progressive Spine & Orthopedics in N.J., will discuss the Integrity Implant System, animal study data and significant histology, patient selection criteria, early case experience, and initial patient outcomes on Feb. 14 at 3:40 p.m.
Anika Therapeutics Inc. is a global joint preservation company that leverages its core expertise in hyaluronic acid and implant solutions to provide minimally invasive products that restore active living for patients. Its focus is on high opportunity spaces within orthopedics, including osteoarthritis pain management, regenerative solutions, sports medicine, and arthrosurface joint solutions, and its products are delivered in key sites of care, including ambulatory surgery centers. Anika’s global operations are headquartered outside of Boston.
Reference
1 Data on file
The company is featuring its differentiated shoulder portfolio, including the recently launched Integrity Implant System for rotator cuff repair, RevoMotion Reverse Shoulder Arthroplasty (RSA) System with newly launched AIM (Anika Implant Management) surgical planning software, X-Twist Fixation System with biocomposite suture anchor, OVOMotion with Inlay Glenoid Total Shoulder Arthroplasty (TSA) System.
Anika also announced at the show the limited launch of the X-Twist Fixation System with biocomposite suture anchor. X-Twist’s suture anchors enable fast and strong insertion into bone, requiring only 3.25 turns and offering significant torsional strength. The launch of the X-Twist Fixation System with biocomposite suture anchors now expands the X-Twist family portfolio with both permanent and resorbable material options.
“We’re thrilled to once again participate in the AAOS 2024 Annual Meeting and to showcase our advanced portfolio of regenerative, sports medicine and bone preserving implant solutions designed to meet the evolving needs of surgeons across the orthopedic continuum of care,” Anika President/CEO Cheryl R. Blanchard, Ph.D., said. “Anika is excited to highlight our recently launched Integrity Implant System, our newest regenerative hyaluronic acid (HA)-based scaffold for rotator cuff repair, and our X-Twist Fixation System with biocomposite suture anchor. We have also enhanced our shoulder arthroplasty offerings with the launch of AIM, Anika Implant Management, for 3D preoperative planning which is now available with both the RevoMotion Reverse Shoulder and OVOMotion Total Shoulder arthroplasty systems. These new products, and the continued combination of Anika’s advanced HA-based regenerative technology with our bone preserving implants and sports medicine solutions, provide surgeons with powerful tools and options to support their patients and help them return to active living.”
The Integrity Implant System, comprised of a hyaluronic acid-based and PET reinforced scaffold with bone and tendon fixation components and single-use arthroscopic delivery instruments, is designed to protect an injured tendon, promote healing, and support cell infiltration and regenerative healing in rotator cuff repair and other tendon procedures. The Integrity implant is inherently strong both when hydrated and dry and can be confidently manipulated arthroscopically. Integrity offers a differentiated solution for shoulder surgeons to treat rotator cuff tears. The flexible, knitted, porous HA-based scaffold provides greater regenerative capacity compared to first generation collagen patches1 and supports regenerative healing through improved cell infiltration, tissue remodeling, and tendon thickening.1 Early usage during the limited market release indicates high levels of interest and excitement for this product. The implant strength, staple and tissue tack fixation capabilities, unique “lateral first” implant technique, and ability to manipulate the implant are standout features of the Integrity system.
The RevoMotion Reverse Shoulder Arthroplasty System, with the industry’s smallest diameter threaded baseplate, offers surgeons and patients the ability to restore motion and preserve bone. Anika’s newly launched 3D preoperative planning software, AIM allows surgeons to personalize and pre-plan each surgery, including assessing erosion depth and bony defects, determining optimal implant position, and assessing the implant’s impact on range of motion.
AIM, formally launching at AAOS this year and now available in the United States, was developed in partnership with Materialise NV. AIM 3D preoperative planning software enables surgeons to generate powerful insights with both glenoid and humeral planning for Anika’s OVOMotion with Inlay Glenoid TSA and RevoMotion RSA Systems.
“Nearly a year into our experience with RevoMotion RSA, we are consistently seeing significant improvements in patient reported outcome measures and function,” said Christopher Baker, M.D., Florida Orthopaedic Institute, Tampa, Fla. “With the addition of AIM, a three-dimensional preoperative planning program, Anika is now able to offer a cutting-edge planning option for the system. This will allow physicians to more accurately manage difficult anatomic deformities of the glenohumeral joint in reverse shoulder arthroplasty.”
Several new Sports Medicine products will also be available in early 2024, including the ProPass Suture Passer, and other new shoulder instrumentation. The ProPass Suture Passer provides surgeons with an intuitive operation and consistent control that reliably passes suture through thick rotator cuff tissue and is offered with or without an auto-capture window. All are expected to launch in 2024.
AAOS attendees can learn more about Anika’s compelling products at the Anika booth 5145, as well as an Innovation Theater sessions held in booth 4003:
“Integrity: The Next Generation of Rotator Cuff Augmentation,” led by David Porter, M.D., with Progressive Spine & Orthopedics in N.J., will discuss the Integrity Implant System, animal study data and significant histology, patient selection criteria, early case experience, and initial patient outcomes on Feb. 14 at 3:40 p.m.
Anika Therapeutics Inc. is a global joint preservation company that leverages its core expertise in hyaluronic acid and implant solutions to provide minimally invasive products that restore active living for patients. Its focus is on high opportunity spaces within orthopedics, including osteoarthritis pain management, regenerative solutions, sports medicine, and arthrosurface joint solutions, and its products are delivered in key sites of care, including ambulatory surgery centers. Anika’s global operations are headquartered outside of Boston.
Reference
1 Data on file