Sam Brusco, Associate Editor04.09.24
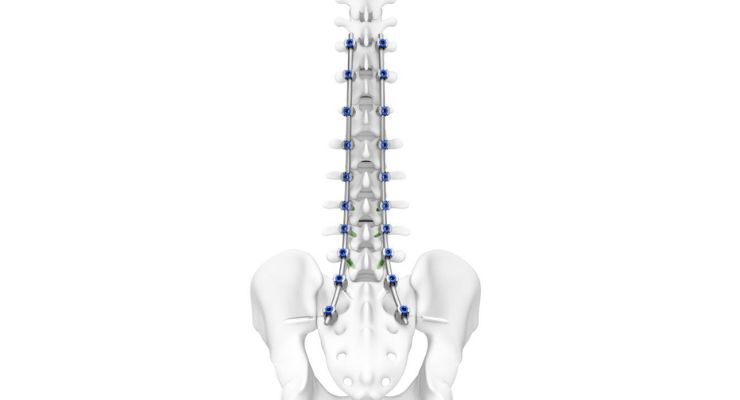
DePuy Synthes announced it will highlight new technological advancements in its spine portfolio, including its latest innovation—the TriALTIS spine system—this week at the 31st International Meeting on Advance Spine Techniques (IMAST) 2024.
The company revealed that TriALTIS earned U.S. Food and Drug Administration (FDA) clearance in October 2023.
The next-gen posterior thoracolumbar pedicle screw system features a comprehensive implant portfolio and instrumentation designed to be integrated with enabling technology, according to DePuy Synthes. Combined with navigation-enabled instruments, TriALTIS aims to address unmet clinical needs across degenerative, tumor, trauma and deformity pathologies in the spine.
Both the TriALTIS spine system and TriALTIS navigation-enabled instruments are now available in the U.S.
DePuy Synthes said TriALTIS was designed around three pillars: tech that enables integration with power and navigation, reliability rooted in the legacy of DePuy Synthes spine implants, and innovation that offers a new development platform and consistent user experience across all indicated pathologies.
“We are thrilled to officially launch the TriALTIS Spine System,” said Russell Powers, Worldwide President, Spine, DePuy Synthes. “Our dedication to improving patient outcomes and optimizing the surgeon experience is evident in every aspect of this system—from its seamless integration with power and navigation, to its precision in implant placement to drive more efficiency in the operating room. The TriALTIS Spine System marks a significant stride in the future of surgical excellence, and we’re excited to continue to pioneer innovation that helps to advance the treatment of complex spine conditions for surgeons and their patients.”
The company revealed that TriALTIS earned U.S. Food and Drug Administration (FDA) clearance in October 2023.
The next-gen posterior thoracolumbar pedicle screw system features a comprehensive implant portfolio and instrumentation designed to be integrated with enabling technology, according to DePuy Synthes. Combined with navigation-enabled instruments, TriALTIS aims to address unmet clinical needs across degenerative, tumor, trauma and deformity pathologies in the spine.
Both the TriALTIS spine system and TriALTIS navigation-enabled instruments are now available in the U.S.
DePuy Synthes said TriALTIS was designed around three pillars: tech that enables integration with power and navigation, reliability rooted in the legacy of DePuy Synthes spine implants, and innovation that offers a new development platform and consistent user experience across all indicated pathologies.
“We are thrilled to officially launch the TriALTIS Spine System,” said Russell Powers, Worldwide President, Spine, DePuy Synthes. “Our dedication to improving patient outcomes and optimizing the surgeon experience is evident in every aspect of this system—from its seamless integration with power and navigation, to its precision in implant placement to drive more efficiency in the operating room. The TriALTIS Spine System marks a significant stride in the future of surgical excellence, and we’re excited to continue to pioneer innovation that helps to advance the treatment of complex spine conditions for surgeons and their patients.”