Sam Brusco, Associate Editor04.24.24
Blue Arbor Technologies has received breakthrough status from the U.S. Food and Drug Administration (FDA) for its RESTORE neuromuscular interface system for people with upper limb loss. The system was also accepted into the FDA’s Total Product Lifecycle (TPLC) Advisory Program (TAP) Pilot.
The RESTORE system integrates the peripheral nervous system with commercially available robotic prosthetics to restore naturalistic hand and arm function. The company said the platform may help patients move upper limb prosthetic devices with unprecedented dexterity, speed, and reliability.
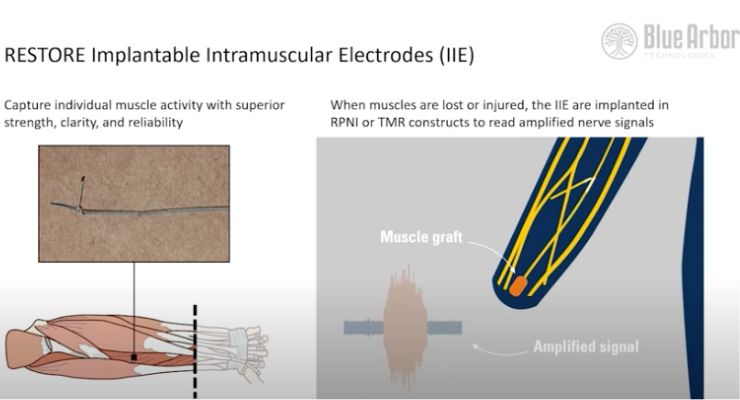
The system provides a direct connection to the patient’s residual muscles and peripheral nerves for reliable, voluntary movement control signals. This is a contrast to current interface technologies, which depend of surface skin electrodes.
Further, RESTORE allows simultaneous and independent movement of finger, wrist, and elbow joints. The system was found to consistently capture motor signals for over five years in early feasibility human trials.
RESTORE can be implanted at the time of amputation or any upper limb reconstructive operation to treat neuroma pain and phantom limb pain, or to revise the residual limb’s shape.
The system consists of:
“Blue Arbor Technologies is dedicated to developing solutions that address the unmet need for improved robotic prosthetic control options for people with limb loss so they can more easily integrate a prothesis in their daily lives and return to normality,” said Paul Cederna, MD, president of Blue Arbor Technologies. “The Breakthrough Designation and TAP enrollment is a valuable step in the pathway to FDA market clearance. We look forward to working closely with the agency to make the RESTORE System available to people with upper limb loss to hopefully increase prosthesis adoption and use.”
The RESTORE system integrates the peripheral nervous system with commercially available robotic prosthetics to restore naturalistic hand and arm function. The company said the platform may help patients move upper limb prosthetic devices with unprecedented dexterity, speed, and reliability.
The system provides a direct connection to the patient’s residual muscles and peripheral nerves for reliable, voluntary movement control signals. This is a contrast to current interface technologies, which depend of surface skin electrodes.
Further, RESTORE allows simultaneous and independent movement of finger, wrist, and elbow joints. The system was found to consistently capture motor signals for over five years in early feasibility human trials.
RESTORE can be implanted at the time of amputation or any upper limb reconstructive operation to treat neuroma pain and phantom limb pain, or to revise the residual limb’s shape.

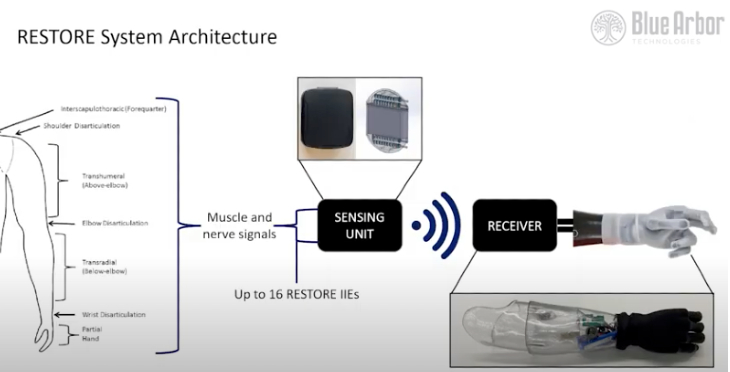
The system consists of:
- Implantable intramuscular electrodes
- A sensing unit to filter, condition, and process patient-generated electromyographic (EMG) signals and wirelessly transmit them to a socket-mounted receiver
- A socket-mounted receiver the decodes the signals into movement commands for the prosthesis
- A software package that controls the entire system
“Blue Arbor Technologies is dedicated to developing solutions that address the unmet need for improved robotic prosthetic control options for people with limb loss so they can more easily integrate a prothesis in their daily lives and return to normality,” said Paul Cederna, MD, president of Blue Arbor Technologies. “The Breakthrough Designation and TAP enrollment is a valuable step in the pathway to FDA market clearance. We look forward to working closely with the agency to make the RESTORE System available to people with upper limb loss to hopefully increase prosthesis adoption and use.”