Erik Michalesko, Marketing and Communications Specialist, American Joint Replacement Registry05.18.17
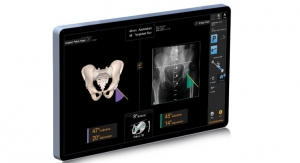
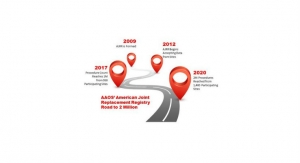
With over one million hip and knee procedures now in its database, the American Joint Replacement Registry (AJRR) is making great strides toward becoming the most comprehensive source of orthopedic outcomes data in the U.S. The Registry previously collected only procedural and patient-reported outcome data, but after the release of its new data specifications in mid-February, the AJRR allows for participating healthcare institutions to collect and report on Level II data, which includes patient risk factors, comorbidities, and complications. The AJRR’s complete dataset will eventually provide its participants with risk adjusted data, which accounts for differences in patient characteristics that may impact health outcomes. The Registry will also be able to use Level II data in analytic efforts to create implant survivorship curves.
AJRR’s selection of appropriate risk variables for Level II included a very detailed process. AJRR conducted a pilot program with a sample of participating hospitals to determine the feasibility of submitting expanded data elements. With those results, AJRR then convened a Risk Adjustment Task Force with members of its Data Management Committee and others with relevant experience. The Task Force identified key variables to include in the Level II system. Subsequently, the American Academy of Orthopaedic Surgeons (AAOS), the American Association of Hip and Knee Surgeons (AAHKS), The Hip Society, The Knee Society, the Centers for Medicare & Medicaid Services (CMS), and AJRR convened a Patient-reported Outcomes Summit for Total Joint Arthroplasty to obtain consensus as to the variables to be included. After the Summit, AJRR decided what Level II data to collect, and now accepts many of the same variables that CMS requests for its quality initiatives such as the Comprehensive Care for Joint Replacement (CJR) model.
“Although the steps toward creating the list of risk variables were a large investment, the results will be well worth it,” said Daniel J. Berry, M.D., AJRR Board of Directors’ chair. “Our Registry will now be able to utilize all of the new data flowing in and provide a deeper, more exact view of arthroplasty outcomes in the United States. Patient risk factors, comorbidities, and complications, the basis of Level II data, will assist several of AJRR’s stakeholders, including our friends in the medical device industry.”
Patient characteristics like conditions and diseases vary from hospital to hospital and may cause arthroplasty outcomes to vary as well. That’s why risk adjustment is so important. Risk adjustment controls for the unique characteristics that impact surgical success that are beyond the control of surgeons or institutions. When it comes to patient comorbidities, risk adjusted data allows for a “leveling” of the playing field.
Complications data is also helpful, as it will allow the AJRR to provide additional metrics on the frequency of complications at the national, network, hospital, and surgeon level. The Registry anticipates that the data will also allow participants to identify root causes of complications that are part of the quality initiatives of patient care and overall costs.
“The ability to analyze patient risk factors is essential to the AJRR’s mission to improve orthopedic care,” continued Dr. Berry. “Level II data introduces many variables that health care providers may consider when planning for surgery. Surgeons will be able to utilize this new data to better inform their decisions, and AJRR’s ability to risk adjust will allow for their findings to not be misconstrued.”
Level II data has other uses besides risk adjustment and identifying the causes of complications; the data will also assist in developing survivorship curves for hip and knee implants. Medical device manufacturers estimate how long a device implant is expected to last, but the Registry will be able to demonstrate the success rate using real-life surgical cases across the country.
“The medical device industry has always been a very important stakeholder group for us,” said Dr. Berry. “They’re represented on our Board of Directors and help guide our decisions as an organization. We would like to be as transparent as possible with our industry partners and inform them when a device isn’t working like it should. If medical device companies are aware of when their products may need improvements, they will be able to develop superior technology and improve patient outcomes.”
The utilization of AJRR’s new data specifications offers new opportunities for healthcare staff and medical device manufacturers alike to use the data presented in the Registry platform in a meaningful way. As the Registry’s procedure count continues to rise past one million, different aspects of joint arthroplasty will continue to be highlighted and different implants will be recorded. AJRR’s data specifications will be updated on an annual basis to keep up with the changing field of orthopedics.
For more information about the AJRR and its data elements, visit www.ajrr.net.
AJRR’s selection of appropriate risk variables for Level II included a very detailed process. AJRR conducted a pilot program with a sample of participating hospitals to determine the feasibility of submitting expanded data elements. With those results, AJRR then convened a Risk Adjustment Task Force with members of its Data Management Committee and others with relevant experience. The Task Force identified key variables to include in the Level II system. Subsequently, the American Academy of Orthopaedic Surgeons (AAOS), the American Association of Hip and Knee Surgeons (AAHKS), The Hip Society, The Knee Society, the Centers for Medicare & Medicaid Services (CMS), and AJRR convened a Patient-reported Outcomes Summit for Total Joint Arthroplasty to obtain consensus as to the variables to be included. After the Summit, AJRR decided what Level II data to collect, and now accepts many of the same variables that CMS requests for its quality initiatives such as the Comprehensive Care for Joint Replacement (CJR) model.
“Although the steps toward creating the list of risk variables were a large investment, the results will be well worth it,” said Daniel J. Berry, M.D., AJRR Board of Directors’ chair. “Our Registry will now be able to utilize all of the new data flowing in and provide a deeper, more exact view of arthroplasty outcomes in the United States. Patient risk factors, comorbidities, and complications, the basis of Level II data, will assist several of AJRR’s stakeholders, including our friends in the medical device industry.”
Patient characteristics like conditions and diseases vary from hospital to hospital and may cause arthroplasty outcomes to vary as well. That’s why risk adjustment is so important. Risk adjustment controls for the unique characteristics that impact surgical success that are beyond the control of surgeons or institutions. When it comes to patient comorbidities, risk adjusted data allows for a “leveling” of the playing field.
Complications data is also helpful, as it will allow the AJRR to provide additional metrics on the frequency of complications at the national, network, hospital, and surgeon level. The Registry anticipates that the data will also allow participants to identify root causes of complications that are part of the quality initiatives of patient care and overall costs.
“The ability to analyze patient risk factors is essential to the AJRR’s mission to improve orthopedic care,” continued Dr. Berry. “Level II data introduces many variables that health care providers may consider when planning for surgery. Surgeons will be able to utilize this new data to better inform their decisions, and AJRR’s ability to risk adjust will allow for their findings to not be misconstrued.”
Level II data has other uses besides risk adjustment and identifying the causes of complications; the data will also assist in developing survivorship curves for hip and knee implants. Medical device manufacturers estimate how long a device implant is expected to last, but the Registry will be able to demonstrate the success rate using real-life surgical cases across the country.
“The medical device industry has always been a very important stakeholder group for us,” said Dr. Berry. “They’re represented on our Board of Directors and help guide our decisions as an organization. We would like to be as transparent as possible with our industry partners and inform them when a device isn’t working like it should. If medical device companies are aware of when their products may need improvements, they will be able to develop superior technology and improve patient outcomes.”
The utilization of AJRR’s new data specifications offers new opportunities for healthcare staff and medical device manufacturers alike to use the data presented in the Registry platform in a meaningful way. As the Registry’s procedure count continues to rise past one million, different aspects of joint arthroplasty will continue to be highlighted and different implants will be recorded. AJRR’s data specifications will be updated on an annual basis to keep up with the changing field of orthopedics.
For more information about the AJRR and its data elements, visit www.ajrr.net.