08.14.19
AT A GLANCE
Rank: #1 (Last year: #1)
$13.60 Billion
Prior Fiscal: $12.44 Billion
Percentage Change: +9.3%
No. of Employees: 36,000
Global Headquarters: Kalamazoo, Mich.
KEY EXECUTIVES:
Kevin A. Lobo, Chairman and CEO
Glenn S. Boehnlein, VP and CFO
Timothy J. Scannell, President and COO
Andy Pierce, Group President, MedSurg
Spencer Stiles, Group President, Neurotechnology, Instruments and Spine
Xavier M.C. Berling, President, Trauma & Extremities
Dylan B. Crotty, President, Instruments
Brent W. Ladd, President, Endoscopy
Eric D. Major, President, Spine
Mark H. Paul, President, Neurovascular
Bradford L. Saar, President, Medical
Stuart F. Simpson, President, Joint Replacement
Stryker’s epic run of tuck-in acquisitions shows no sign of slowing down, and its largest buy last year really showed some backbone.
The orthopedic giant proclaimed its intent to acquire Virginia-based spinal device maker K2M Group Holdings for $1.4 billion last August. “This acquisition underscores our commitment to the spinal market, which is the largest segment of orthopedics with significant unmet needs,” Chairman and CEO Kevin Lobo stated to the press at the time of announcement.
K2M is known for its Balance ACS platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes. The firm’s portfolio also includes minimally invasive implants like the adjustable MOJAVE 3D printed spinal support implant, biologics, and complex spine injury treatments like the MESA 2 Deformity Spinal System.
“K2M’s main focus is complex spine, which we believe is an attractive segment since the procedures are typically performed by high-volume surgeons and generate significant revenue per case,” Mike Matson, an analyst with Needham & Co., wrote in a research note. “K2M faces only two key competitors in complex spine (Johnson & Johnson and Medtronic) and its innovative products including its MESA 2 system have enabled it to gain significant share in this segment.”
Analysts have said Stryker’s Spine unit has struggled as of late. K2M raked in nearly $300 million in 2017 thanks to a strong position in the complex spine markets, an attractive minimally invasive surgery portfolio, and additive manufacturing capabilities.
“Spine has been a problem child for Stryker,” Evercore analyst Vijay Kumar said in a report to clients. “We think K2M provides visibility in spine turning into a low-single-digit to mid-single-digit growth segment once the deal annualizes.”
The deal, which closed in November, also reaffirmed Stryker’s acquisition strategy. Early last summer, chatter suggested the firm may have been in the market to buy Boston Scientific. Stryker later squashed that rumor.
“There was some nervousness around whether Stryker was contemplating a departure from its tuck-in strategy,” Kumar told Investor’s Business Daily. “The K2M deal should provide confidence in Stryker sticking to its well-oiled strategy of tuck-in acquisitions that are accretive to (the top line) and margins.”
This acquisition strategy helped land Stryker well in the black once again. The company recorded its 39th consecutive year of sales growth with last year’s $13.6 billion, a healthy 9.3 percent jump from the year prior. Higher shipments of medical, instruments, endoscopy, neurotechnology, knees, and trauma and extremities products were the main cause for the near-double-digit revenue hike.
ANALYST INSIGHTS: Stryker continues to be aggressive with both organic and inorganic growth opportunities. Look for Stryker to continue to be opportunistic in M&A across its market segments while also looking for M&A to expand its outside U.S. footprint.
Neurotechnology and Spine’s performance last year easily outstripped the rest even without K2M’s help, skyrocketing 18 percent to rest at $2.6 billion in sales. Not much was offered in the way of reasoning for this momentous increase apart from higher shipments of neurotechnology products and the addition of new neurovascular products last year. Neurotechnology claimed the lion’s share of the earnings with $1.7 billion—a whopping 22.1 percent over the prior year. Spine sales grew 10.3 percent, accruing $828 million (though this segment is likely to blossom next year as K2M becomes fully integrated).
Stryker and 3D Systems began a distribution partnership for craniomaxillofacial Virtual Surgical Planning (VSP) and anatomical models last January. VSP is a service-based approach to personalized surgery, combining medical imaging, surgical simulation, and 3D printing. To use VSP, the surgeon brings clinical knowledge and a desired surgical plan to an online meeting with a 3D Systems biomedical engineer to simulate and plan the procedure. The result is a digital plan transferred to the operating room via accurate 3D printed patient-specific models, guides, and templates.
The company closed the $664 million Entellus Medical buy initiated in December 2017 last February. With this purchase, Stryker gained a family of minimally invasive balloon device products, including Entellus’ flagship Xpress device, which treats blocked sinuses.
Stryker’s Trevo Retriever acquired an expanded indication last February as a front-line treatment for acute ischemic stroke up to 24 hours from symptom onset, increasing the treatment window by 18 hours. The Trevo Retriever, which is the first and only thrombectomy device FDA cleared to significantly reduce disability up to 24 hours from symptom onset, received the expanded indication thanks to last year’s updated treatment guidelines from the American Heart Association and American Stroke Association.
The company crossed the 95 percent thresholds in the VEXIM acquisition it had begun in October 2017 last February. In the weeks following, Stryker filed a proposed public buy-out offer for all outstanding shares. VEXIM specializes in minimally invasive treatment of vertebral fractures. Its SpineJack Implantable Fracture Reduction System, which reduces painful osteoporotic vertebral compression fractures, obtained FDA clearance last September. SpineJack has been proven to be superior to balloon kyphoplasty in terms of freedom from adjacent level fracture and midline vertebral height restoration, pain reduction, and improvement in function.
Last March saw FDA clearance for the Tritanium TL Curved Posterior Lumbar Cage, a 3D-printed interbody fusion cage for lumbar fixation. The hollow implant features a configuration of both solid and porous structures built using Stryker’s proprietary AMagine manufacturing process. The Tritanium TL Cage is composed of Tritanium Technology, a highly porous titanium material inspired by the microstructure of cancellous bone. Central graft and lateral windows help reduce the cage’s stiffness, aid in fusion visualization, and allow for bone graft containment. Its multidirectional teeth facilitate multidirectional fixation so surgeons can steer and rotate the cage to their desired placement.
The Surpass Streamline Flow Diverter attained FDA pre-market approval last July to treat unruptured large and giant wide neck intracranial aneurysms. It is the first flow diverter for large and giant posterior communicating artery aneurysms, which are more challenging to treat due to their location and surrounding anatomy. The stent reliably opens and provides consistent mesh density across the neck of the aneurysm to help with occlusion while maintaining perforator artery patency. It can also be resheathed, repositioned, and recaptured without losing distal wire position.
Stryker acquired HyperBranch Medical Technology for $220 million last October. HyperBranch develops medical devices based on its proprietary polymers and cross-linked hydrogels, and its Adherus AutoSpray Dural Sealant is one of two FDA-approved dural sealants on the market. The PMA approved and CE marked hydrogel is an adjunct to standard dural repair methods, creating a watertight closure of the dura. The Adherus product complements Stryker’s craniomaxillofacial portfolio.
ANALYST INSIGHTS: Kevin Lobo continues to do his John Brown imitation by delivering predictable high performance shareholder return. This is built on the back of thoughtful, well executed organic and inorganic growth strategies that span all divisions and, as usual, best in industry leadership and management throughout the organization.
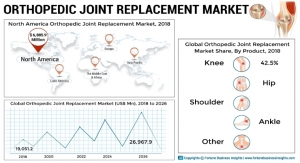
Orthopaedics division revenue swelled 5.9 percent last year to reach $5 billion. Unit volume expanded thanks to higher shipments of knees and trauma and extremity products, partially offset by lower prices. With $1.7 billion of proceeds last year, knee sales grew 6.6 percent. Hips sales widened 2.5 percent to collect $1.3 billion. Trauma and Extremities gathered $1.6 billion for a 6.9 percent jump, and “other” orthopedic products blossomed 11 percent with $374 million in revenue.
The company launched the Trident II Acetabular System at last year’s American Academy of Orthopaedic Surgeons meeting. The system is composed of Trident II Tritanium, which combines AMagine additive manufactured Tritanium In-Growth Technology with the precision of Mako Robotic-Arm Assisted Surgery. Trident II Tritanium’s slim wall allows for large femoral size options and optimal poly thickness to aid in greater range of motion, joint stability, and lower dislocation risk. The Trident II Tritanium Acetabular System gained FDA clearance in October 2016. The full release offers five shell options—three in Tritanium, and three in hydroxyapatite.
That same day saw the launch of Pivot Guardian, the industry’s first post-free hip distraction system. Pivot Guardian helps to mitigate groin complications and heel slip linked to hip arthroscopy. The post-free design lets surgeons create adequate force for distraction without applying pressure to the groin while alleviating heel slip and excessive pressure applied to the foot.
The next day witnessed another market first with the unveiling of Cobra, a new reusable suture passing technology for rotator cuff repair. The first-of-its-kind technology features novel needle manufacturing and innovative jaw design to produce the first reusable needle technology. Cobra’s improved needle stiffness helps prevent intraoperative misfires and distal needle tip breakages. The suture passer utilizes technology acquired from Arthrogenx in 2017.
MedSurg franchise revenue last year advanced 8.8 percent to $6 billion. Higher shipments of medical, instruments, and endoscopy products all contributed to this rise, which was somewhat offset by lower prices. Within the division, Instruments proceeds grew 8.6 percent to $1.8 billion, Endoscopy sales rose 11.7 percent to $1.8 billion, Medical revenue jumped 7.6 percent to $2.1 billion, and Sustainability earnings flattened at $259 million.
Building on a partnership to reprocess single-use devices (SUDs) that began in 2015, Hygia Health Services entered Stryker’s fold last May. With this buy, Stryker hopes to help hospitals and health systems increase the value their SUD reprocessing programs deliver. Hygia’s product lineup includes air transfer mattresses, fall alarms, ECG leads, cerebral-somatic sensors, and infusor bags. In 2017, Stryker helped its almost 3,000 SUD reprocessing customers collective save $326 million in supply costs and divert 13.4 million pounds of waste from landfills.
Stryker nabbed Swiss surgical smoke evacuation solutions firm SafeAir AG last June. Highly complementary to the Instruments division, the company’s smoke evacuation products help reduce exposure to hazards associated with surgical smoke. The acquisition closed in the fourth quarter of last year.
The string of acquisitions continued last September when Stryker bought San Francisco-based Invuity Inc. for $190 million. The deal added advanced photonics and single-use, lighted instruments that offer enhanced visualization during orthopedic and spine surgery, general surgery, and women’s health procedures to the firm’s Instruments business. The acquisition was completed a month and a half later.
The Stryker F1 Small Bone Power System was introduced last March. The cordless, balanced, lightweight powered instrument boosts the efficiency of distal extremity procedures. Surgeons can choose from two gripping styles—either pencil or pistol-style. The device features five motors and three attachments that are interchangeable between the SmartGRIP Control Module to let surgeons choose their specific approach. The F1 System also connects to an online data module that helps optimize management of Stryker’s smart devices through device-derived, actionable data.
Rank: #1 (Last year: #1)
$13.60 Billion
Prior Fiscal: $12.44 Billion
Percentage Change: +9.3%
No. of Employees: 36,000
Global Headquarters: Kalamazoo, Mich.
KEY EXECUTIVES:
Kevin A. Lobo, Chairman and CEO
Glenn S. Boehnlein, VP and CFO
Timothy J. Scannell, President and COO
Andy Pierce, Group President, MedSurg
Spencer Stiles, Group President, Neurotechnology, Instruments and Spine
Xavier M.C. Berling, President, Trauma & Extremities
Dylan B. Crotty, President, Instruments
Brent W. Ladd, President, Endoscopy
Eric D. Major, President, Spine
Mark H. Paul, President, Neurovascular
Bradford L. Saar, President, Medical
Stuart F. Simpson, President, Joint Replacement
Stryker’s epic run of tuck-in acquisitions shows no sign of slowing down, and its largest buy last year really showed some backbone.
The orthopedic giant proclaimed its intent to acquire Virginia-based spinal device maker K2M Group Holdings for $1.4 billion last August. “This acquisition underscores our commitment to the spinal market, which is the largest segment of orthopedics with significant unmet needs,” Chairman and CEO Kevin Lobo stated to the press at the time of announcement.
K2M is known for its Balance ACS platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes. The firm’s portfolio also includes minimally invasive implants like the adjustable MOJAVE 3D printed spinal support implant, biologics, and complex spine injury treatments like the MESA 2 Deformity Spinal System.
“K2M’s main focus is complex spine, which we believe is an attractive segment since the procedures are typically performed by high-volume surgeons and generate significant revenue per case,” Mike Matson, an analyst with Needham & Co., wrote in a research note. “K2M faces only two key competitors in complex spine (Johnson & Johnson and Medtronic) and its innovative products including its MESA 2 system have enabled it to gain significant share in this segment.”
Analysts have said Stryker’s Spine unit has struggled as of late. K2M raked in nearly $300 million in 2017 thanks to a strong position in the complex spine markets, an attractive minimally invasive surgery portfolio, and additive manufacturing capabilities.
“Spine has been a problem child for Stryker,” Evercore analyst Vijay Kumar said in a report to clients. “We think K2M provides visibility in spine turning into a low-single-digit to mid-single-digit growth segment once the deal annualizes.”
The deal, which closed in November, also reaffirmed Stryker’s acquisition strategy. Early last summer, chatter suggested the firm may have been in the market to buy Boston Scientific. Stryker later squashed that rumor.
“There was some nervousness around whether Stryker was contemplating a departure from its tuck-in strategy,” Kumar told Investor’s Business Daily. “The K2M deal should provide confidence in Stryker sticking to its well-oiled strategy of tuck-in acquisitions that are accretive to (the top line) and margins.”
This acquisition strategy helped land Stryker well in the black once again. The company recorded its 39th consecutive year of sales growth with last year’s $13.6 billion, a healthy 9.3 percent jump from the year prior. Higher shipments of medical, instruments, endoscopy, neurotechnology, knees, and trauma and extremities products were the main cause for the near-double-digit revenue hike.
ANALYST INSIGHTS: Stryker continues to be aggressive with both organic and inorganic growth opportunities. Look for Stryker to continue to be opportunistic in M&A across its market segments while also looking for M&A to expand its outside U.S. footprint.
—Dave Sheppard, Co-Founder and Managing Director, MedWorld Advisors
Neurotechnology and Spine’s performance last year easily outstripped the rest even without K2M’s help, skyrocketing 18 percent to rest at $2.6 billion in sales. Not much was offered in the way of reasoning for this momentous increase apart from higher shipments of neurotechnology products and the addition of new neurovascular products last year. Neurotechnology claimed the lion’s share of the earnings with $1.7 billion—a whopping 22.1 percent over the prior year. Spine sales grew 10.3 percent, accruing $828 million (though this segment is likely to blossom next year as K2M becomes fully integrated).
Stryker and 3D Systems began a distribution partnership for craniomaxillofacial Virtual Surgical Planning (VSP) and anatomical models last January. VSP is a service-based approach to personalized surgery, combining medical imaging, surgical simulation, and 3D printing. To use VSP, the surgeon brings clinical knowledge and a desired surgical plan to an online meeting with a 3D Systems biomedical engineer to simulate and plan the procedure. The result is a digital plan transferred to the operating room via accurate 3D printed patient-specific models, guides, and templates.
The company closed the $664 million Entellus Medical buy initiated in December 2017 last February. With this purchase, Stryker gained a family of minimally invasive balloon device products, including Entellus’ flagship Xpress device, which treats blocked sinuses.
Stryker’s Trevo Retriever acquired an expanded indication last February as a front-line treatment for acute ischemic stroke up to 24 hours from symptom onset, increasing the treatment window by 18 hours. The Trevo Retriever, which is the first and only thrombectomy device FDA cleared to significantly reduce disability up to 24 hours from symptom onset, received the expanded indication thanks to last year’s updated treatment guidelines from the American Heart Association and American Stroke Association.
The company crossed the 95 percent thresholds in the VEXIM acquisition it had begun in October 2017 last February. In the weeks following, Stryker filed a proposed public buy-out offer for all outstanding shares. VEXIM specializes in minimally invasive treatment of vertebral fractures. Its SpineJack Implantable Fracture Reduction System, which reduces painful osteoporotic vertebral compression fractures, obtained FDA clearance last September. SpineJack has been proven to be superior to balloon kyphoplasty in terms of freedom from adjacent level fracture and midline vertebral height restoration, pain reduction, and improvement in function.
Last March saw FDA clearance for the Tritanium TL Curved Posterior Lumbar Cage, a 3D-printed interbody fusion cage for lumbar fixation. The hollow implant features a configuration of both solid and porous structures built using Stryker’s proprietary AMagine manufacturing process. The Tritanium TL Cage is composed of Tritanium Technology, a highly porous titanium material inspired by the microstructure of cancellous bone. Central graft and lateral windows help reduce the cage’s stiffness, aid in fusion visualization, and allow for bone graft containment. Its multidirectional teeth facilitate multidirectional fixation so surgeons can steer and rotate the cage to their desired placement.
The Surpass Streamline Flow Diverter attained FDA pre-market approval last July to treat unruptured large and giant wide neck intracranial aneurysms. It is the first flow diverter for large and giant posterior communicating artery aneurysms, which are more challenging to treat due to their location and surrounding anatomy. The stent reliably opens and provides consistent mesh density across the neck of the aneurysm to help with occlusion while maintaining perforator artery patency. It can also be resheathed, repositioned, and recaptured without losing distal wire position.
Stryker acquired HyperBranch Medical Technology for $220 million last October. HyperBranch develops medical devices based on its proprietary polymers and cross-linked hydrogels, and its Adherus AutoSpray Dural Sealant is one of two FDA-approved dural sealants on the market. The PMA approved and CE marked hydrogel is an adjunct to standard dural repair methods, creating a watertight closure of the dura. The Adherus product complements Stryker’s craniomaxillofacial portfolio.
ANALYST INSIGHTS: Kevin Lobo continues to do his John Brown imitation by delivering predictable high performance shareholder return. This is built on the back of thoughtful, well executed organic and inorganic growth strategies that span all divisions and, as usual, best in industry leadership and management throughout the organization.
—Patrick West, Partner, Mirus Capital Advisors
Orthopaedics division revenue swelled 5.9 percent last year to reach $5 billion. Unit volume expanded thanks to higher shipments of knees and trauma and extremity products, partially offset by lower prices. With $1.7 billion of proceeds last year, knee sales grew 6.6 percent. Hips sales widened 2.5 percent to collect $1.3 billion. Trauma and Extremities gathered $1.6 billion for a 6.9 percent jump, and “other” orthopedic products blossomed 11 percent with $374 million in revenue.
The company launched the Trident II Acetabular System at last year’s American Academy of Orthopaedic Surgeons meeting. The system is composed of Trident II Tritanium, which combines AMagine additive manufactured Tritanium In-Growth Technology with the precision of Mako Robotic-Arm Assisted Surgery. Trident II Tritanium’s slim wall allows for large femoral size options and optimal poly thickness to aid in greater range of motion, joint stability, and lower dislocation risk. The Trident II Tritanium Acetabular System gained FDA clearance in October 2016. The full release offers five shell options—three in Tritanium, and three in hydroxyapatite.
That same day saw the launch of Pivot Guardian, the industry’s first post-free hip distraction system. Pivot Guardian helps to mitigate groin complications and heel slip linked to hip arthroscopy. The post-free design lets surgeons create adequate force for distraction without applying pressure to the groin while alleviating heel slip and excessive pressure applied to the foot.
The next day witnessed another market first with the unveiling of Cobra, a new reusable suture passing technology for rotator cuff repair. The first-of-its-kind technology features novel needle manufacturing and innovative jaw design to produce the first reusable needle technology. Cobra’s improved needle stiffness helps prevent intraoperative misfires and distal needle tip breakages. The suture passer utilizes technology acquired from Arthrogenx in 2017.
MedSurg franchise revenue last year advanced 8.8 percent to $6 billion. Higher shipments of medical, instruments, and endoscopy products all contributed to this rise, which was somewhat offset by lower prices. Within the division, Instruments proceeds grew 8.6 percent to $1.8 billion, Endoscopy sales rose 11.7 percent to $1.8 billion, Medical revenue jumped 7.6 percent to $2.1 billion, and Sustainability earnings flattened at $259 million.
Building on a partnership to reprocess single-use devices (SUDs) that began in 2015, Hygia Health Services entered Stryker’s fold last May. With this buy, Stryker hopes to help hospitals and health systems increase the value their SUD reprocessing programs deliver. Hygia’s product lineup includes air transfer mattresses, fall alarms, ECG leads, cerebral-somatic sensors, and infusor bags. In 2017, Stryker helped its almost 3,000 SUD reprocessing customers collective save $326 million in supply costs and divert 13.4 million pounds of waste from landfills.
Stryker nabbed Swiss surgical smoke evacuation solutions firm SafeAir AG last June. Highly complementary to the Instruments division, the company’s smoke evacuation products help reduce exposure to hazards associated with surgical smoke. The acquisition closed in the fourth quarter of last year.
The string of acquisitions continued last September when Stryker bought San Francisco-based Invuity Inc. for $190 million. The deal added advanced photonics and single-use, lighted instruments that offer enhanced visualization during orthopedic and spine surgery, general surgery, and women’s health procedures to the firm’s Instruments business. The acquisition was completed a month and a half later.
The Stryker F1 Small Bone Power System was introduced last March. The cordless, balanced, lightweight powered instrument boosts the efficiency of distal extremity procedures. Surgeons can choose from two gripping styles—either pencil or pistol-style. The device features five motors and three attachments that are interchangeable between the SmartGRIP Control Module to let surgeons choose their specific approach. The F1 System also connects to an online data module that helps optimize management of Stryker’s smart devices through device-derived, actionable data.