James A. Dunning, QPC Services08.09.16
I grew up in Roseville, Mich., at a time when manufacturing was central to the local economy. Back then, heavy industrial manufacturing was king. But over the years, the art of mass production has changed significantly. From automation and robotics to computer-controlled systems, the manufacturing environment looks radically different today than it did in decades past.
Manufacturing remains critically important to both the developing and the advanced world. But the way in which products are made is undergoing a radical transformation, thanks largely to 3D printing technology, otherwise known as additive manufacturing. Experts predict significant growth in 3D printing over the next five years, with the market for the technology swelling to $5.2 billion by 2020. “The 3D printing industry is expected to change nearly every industry it touches, completely disrupting the traditional manufacturing process,” the website On 3D Printing states.
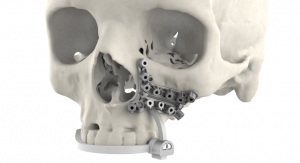
No industry arguably will be transformed by 3D printing as much as healthcare. There already are countless examples of the technology being used to print items like prosthetic devices, surgical models, and even living tissue. And while there are ethical issues to consider and customer education initiatives to develop, the potential for revolutionary change truly exists.
I am certainly not an expert on 3D printing and neither are the folks at the U.S. Food and Drug Administration (FDA), but the agency is attempting to educate itself and establish some guidelines to regulate the additive manufacturing of medical devices. The FDA is currently highlighting 3D printing on its website (http://bit.ly/1sv1qox).
The FDA has long had an interest in 3D printing. In August, 2013, the agency’s blog, “FDA Voice,” included an entry by Steven K. Pollack, Ph.D., and James Coburn, M.S, entitled “FDA Goes 3D.” The authors discussed ways the agency is using the technology to better evaluate FDA-regulated products, noting, “Here at FDA, we’re using it to expand our research efforts and expand our capabilities to review innovative medical products. In fact, 3D printing is fast becoming a focus in our practice of regulatory science—that is, the science of developing new tools, standards, and approaches to assess the safety, effectiveness, quality, and performance of FDA-regulated products.”
The blog even gives examples of the FDA’s use of 3D technology: “At our Functional Performance and Device Use Laboratory, we’ve developed and adapted computer-modeling methods to help us determine the effect of design changes on the safety and performance of devices when used in different patient populations. The 3D technology enables us to tweak the design in ways large and small, and to see precisely how those tweaks will change both fit and functionality. In an era of increasingly personalized medicine, which involves the development of treatments that are tailored to an individual patient or a group that shares certain characteristics, including anatomical features, it helps us to fine-tune our evaluation of patient-fitted products.”
Pharmaceutical products are now being produced through 3D printing as well. Last year, the FDA approved the first 3D-printed drug, an epilepsy pill called Spritam. A news release from Ohio-based Aprecia Pharmaceuticals claims the drug is made using a 3D printing method called ZipDose technology, which produces a porous pill that rapidly disintegrates with a sip of liquid. Although 3D-printed drug development is still in its infancy, experts believe the technique could eventually enable companies to make custom-ordered pharmaceuticals based on specific patient needs, thus replacing the traditional “one-product-fits-all” approach.
3D Printing Guidance for Medical Devices
The FDA has cleared more than 85 applications for 3D-printed medical devices but none of those submissions have involved high-risk products requiring premarket approval. The “leap-frog” draft guidance released by the FDA in May is designed to provide manufacturers with the agency’s outlook on the technical considerations involved in making 3D-printed products as well as its thoughts on characterizing and validating such devices.
Specifically, the guidance covers considerations for two major areas of medical device development: design and manufacturing; and device testing, which includes characterization, validation, and verification. The guidance, however, does not address 3D-printed products containing biologics, cells, or human tissues. “Biological, cellular, or tissue-based products manufactured using AM [additive manufacturing] technology may necessitate additional regulatory and manufacturing process considerations and/or different regulatory pathways,” the proposed regulations state.
In its 28-page guidance document, the FDA lists several advantages of 3D printing over traditional manufacturing; namely, creating anatomically-matched devices and surgical instruments using a patient’s own medical imaging. Other benefits include the simplicity in fabricating complex geometric structures, and the development of engineered porous structures, tortuous internal channels, and internal support structures.
Yet the agency acknowledges the technology is not without its shortcomings. “The unique aspects of the AM process, such as the layer-wise fabrication process, and the relative lack of medical device history of devices manufactured using AM techniques, pose challenges in determining optimal characterization and assessment methods for the final finished device, as well as optimal process validation and acceptance methods for these devices,” the FDA guidance states.
The proposed regulations are based on feedback the FDA received from a 2014 public workshop on 3D printing challenges. Among the major takeaways from that workshop include the importance of material control, the impact of the 3D printer and post-printing processes on final device performance, and the need for a “robust” process validation and acceptance protocol.
In its guidance, the FDA says it expects 3D-printed products to adhere to quality systems requirements to ensure the devices perform as intended. Also, device makers must “clearly identify each step in the printing process…from initial device design to the post-processing of the final” product due to the fact that various additive manufacturing technologies, materials, and printers exist.
The FDA’s testing directive for 3D-printed devices doesn’t stray too far from its regulations for traditionally manufactured products: Required application data must be based on “intended use, risk profile, and classification,” and will most likely vary, depending on whether the device is implantable or customized. In addition, the agency requires that patient-specific products specify a range of dimensions that encompass all possible sizes for production. Essentially, 3D-printed devices should be tested for the same performance characteristics as products made using conventional methods, the guidance notes.
Modernizing Manufacturing
Advanced manufacturing methodologies are radically impacting regulated industry. For example, the FDA has been encouraging advanced manufacturing methods in the pharmaceutical sector for several years now, in large part due to critical drug shortages. In a formal statement to a congressional subcommittee nearly three years ago, former FDA Commissioner Janet Woodcock, M.D., detailed the agency’s more than 10-year effort to modernize drug manufacturing, noting that the future of drug manufacturing lies in high-technology, computer-controlled production facilities that can rapidly respond to changes in demand and are capable of seamlessly producing various dosages and dosage forms.
“FDA has been working to stimulate development of novel manufacturing technologies in collaboration with academic and industry experts. The new technologies enable forms of ‘continuous manufacturing,’ wherein the finished drug product is produced in a continuous stream, as opposed to traditional methods that involve a series of so-called ‘unit operations,’such as milling, mixing, granulation, and so forth,” Woodcock said. “In examples of advanced novel manufacturing, production is continuous from chemical synthesis of the active ingredient through production of the tablets or other dosage form. This type of manufacturing is on the verge of entering commercial production. There are a multitude of advantages of this type of production, when done well. Product quality can be precisely controlled. Production scale-up issues, which frequently bedevil drug development, will likely be much less of an issue. Increases in capacity can be handled in a straightforward manner. A range of strengths or doses may be prepared more easily, which may be important for personalized medicine. However, other key advantages do not relate to the specific drug product being made. For example, continuous manufacturing plants require a smaller footprint and can be located closer to markets, thus reducing the need for transcontinental shipping of components.”
Sterilization Methodologies
The FDA recently approved a guidance document that had been in draft form for seven years. This guidance document communicated FDA’s current thinking on the supporting data needed in a 510(k) application to support various sterilization methods. The new proposal shows that newer, usually more advanced sterilization methods are lesser known to the FDA, and therefore require more supporting information to be included in the submission.
While the FDA clearly supports innovative sterilization methods, the agency requires supporting data for the processes due to the critical role it plays in product development. I believe this to be a prudent regulatory approach.
Other Factors Driving Advanced Manufacturing
FDA-regulated products are becoming more complicated. Combination products that incorporate drugs/biologics and medical devices benefit from advanced manufacturing processes because they combine entities. Micro devices often require advanced manufacturing methods because of their size, and high-cost materials necessitate such production processes to reduce waste.
Summary
Regulatory oversight and advanced manufacturing processes each impact the other in profound ways. In general, regulatory agencies like the FDA move slower than industry would like in order to properly perform their mission; manufacturers, though, benefit from regulators’ due diligence because these gatekeepers help keep the industry in check. Ultimately, the interaction between regulatory agencies and medtech manufacturers is mutually beneficial. The public, however, is the true beneficiary, served by the joint effort between regulators and manufacturers to improve patient health and safety.
James A. “Jim” Dunning’s consulting career began in 2001. He has provided quality and regulatory consulting services for various companies ranging from Fortune 500 medical device firms to startups. Dunning’s passion, however, lies with startups and small companies, especially those in regulatory distress. He has amassed significant experience in preparing 510(k) applications, developing complete Quality Management Systems, providing Quality System Training, and advising on quality, business, and leadership issues. Dunning is a senior member of the American Society for Quality (ASQ) and a member of the Regulatory Affairs Professional Society (RAPS). He can be reached at jdunning@qpcservices.com.
Manufacturing remains critically important to both the developing and the advanced world. But the way in which products are made is undergoing a radical transformation, thanks largely to 3D printing technology, otherwise known as additive manufacturing. Experts predict significant growth in 3D printing over the next five years, with the market for the technology swelling to $5.2 billion by 2020. “The 3D printing industry is expected to change nearly every industry it touches, completely disrupting the traditional manufacturing process,” the website On 3D Printing states.
No industry arguably will be transformed by 3D printing as much as healthcare. There already are countless examples of the technology being used to print items like prosthetic devices, surgical models, and even living tissue. And while there are ethical issues to consider and customer education initiatives to develop, the potential for revolutionary change truly exists.
I am certainly not an expert on 3D printing and neither are the folks at the U.S. Food and Drug Administration (FDA), but the agency is attempting to educate itself and establish some guidelines to regulate the additive manufacturing of medical devices. The FDA is currently highlighting 3D printing on its website (http://bit.ly/1sv1qox).
The FDA has long had an interest in 3D printing. In August, 2013, the agency’s blog, “FDA Voice,” included an entry by Steven K. Pollack, Ph.D., and James Coburn, M.S, entitled “FDA Goes 3D.” The authors discussed ways the agency is using the technology to better evaluate FDA-regulated products, noting, “Here at FDA, we’re using it to expand our research efforts and expand our capabilities to review innovative medical products. In fact, 3D printing is fast becoming a focus in our practice of regulatory science—that is, the science of developing new tools, standards, and approaches to assess the safety, effectiveness, quality, and performance of FDA-regulated products.”
The blog even gives examples of the FDA’s use of 3D technology: “At our Functional Performance and Device Use Laboratory, we’ve developed and adapted computer-modeling methods to help us determine the effect of design changes on the safety and performance of devices when used in different patient populations. The 3D technology enables us to tweak the design in ways large and small, and to see precisely how those tweaks will change both fit and functionality. In an era of increasingly personalized medicine, which involves the development of treatments that are tailored to an individual patient or a group that shares certain characteristics, including anatomical features, it helps us to fine-tune our evaluation of patient-fitted products.”
Pharmaceutical products are now being produced through 3D printing as well. Last year, the FDA approved the first 3D-printed drug, an epilepsy pill called Spritam. A news release from Ohio-based Aprecia Pharmaceuticals claims the drug is made using a 3D printing method called ZipDose technology, which produces a porous pill that rapidly disintegrates with a sip of liquid. Although 3D-printed drug development is still in its infancy, experts believe the technique could eventually enable companies to make custom-ordered pharmaceuticals based on specific patient needs, thus replacing the traditional “one-product-fits-all” approach.
3D Printing Guidance for Medical Devices
The FDA has cleared more than 85 applications for 3D-printed medical devices but none of those submissions have involved high-risk products requiring premarket approval. The “leap-frog” draft guidance released by the FDA in May is designed to provide manufacturers with the agency’s outlook on the technical considerations involved in making 3D-printed products as well as its thoughts on characterizing and validating such devices.
Specifically, the guidance covers considerations for two major areas of medical device development: design and manufacturing; and device testing, which includes characterization, validation, and verification. The guidance, however, does not address 3D-printed products containing biologics, cells, or human tissues. “Biological, cellular, or tissue-based products manufactured using AM [additive manufacturing] technology may necessitate additional regulatory and manufacturing process considerations and/or different regulatory pathways,” the proposed regulations state.
In its 28-page guidance document, the FDA lists several advantages of 3D printing over traditional manufacturing; namely, creating anatomically-matched devices and surgical instruments using a patient’s own medical imaging. Other benefits include the simplicity in fabricating complex geometric structures, and the development of engineered porous structures, tortuous internal channels, and internal support structures.
Yet the agency acknowledges the technology is not without its shortcomings. “The unique aspects of the AM process, such as the layer-wise fabrication process, and the relative lack of medical device history of devices manufactured using AM techniques, pose challenges in determining optimal characterization and assessment methods for the final finished device, as well as optimal process validation and acceptance methods for these devices,” the FDA guidance states.
The proposed regulations are based on feedback the FDA received from a 2014 public workshop on 3D printing challenges. Among the major takeaways from that workshop include the importance of material control, the impact of the 3D printer and post-printing processes on final device performance, and the need for a “robust” process validation and acceptance protocol.
In its guidance, the FDA says it expects 3D-printed products to adhere to quality systems requirements to ensure the devices perform as intended. Also, device makers must “clearly identify each step in the printing process…from initial device design to the post-processing of the final” product due to the fact that various additive manufacturing technologies, materials, and printers exist.
The FDA’s testing directive for 3D-printed devices doesn’t stray too far from its regulations for traditionally manufactured products: Required application data must be based on “intended use, risk profile, and classification,” and will most likely vary, depending on whether the device is implantable or customized. In addition, the agency requires that patient-specific products specify a range of dimensions that encompass all possible sizes for production. Essentially, 3D-printed devices should be tested for the same performance characteristics as products made using conventional methods, the guidance notes.
Modernizing Manufacturing
Advanced manufacturing methodologies are radically impacting regulated industry. For example, the FDA has been encouraging advanced manufacturing methods in the pharmaceutical sector for several years now, in large part due to critical drug shortages. In a formal statement to a congressional subcommittee nearly three years ago, former FDA Commissioner Janet Woodcock, M.D., detailed the agency’s more than 10-year effort to modernize drug manufacturing, noting that the future of drug manufacturing lies in high-technology, computer-controlled production facilities that can rapidly respond to changes in demand and are capable of seamlessly producing various dosages and dosage forms.
“FDA has been working to stimulate development of novel manufacturing technologies in collaboration with academic and industry experts. The new technologies enable forms of ‘continuous manufacturing,’ wherein the finished drug product is produced in a continuous stream, as opposed to traditional methods that involve a series of so-called ‘unit operations,’such as milling, mixing, granulation, and so forth,” Woodcock said. “In examples of advanced novel manufacturing, production is continuous from chemical synthesis of the active ingredient through production of the tablets or other dosage form. This type of manufacturing is on the verge of entering commercial production. There are a multitude of advantages of this type of production, when done well. Product quality can be precisely controlled. Production scale-up issues, which frequently bedevil drug development, will likely be much less of an issue. Increases in capacity can be handled in a straightforward manner. A range of strengths or doses may be prepared more easily, which may be important for personalized medicine. However, other key advantages do not relate to the specific drug product being made. For example, continuous manufacturing plants require a smaller footprint and can be located closer to markets, thus reducing the need for transcontinental shipping of components.”
Sterilization Methodologies
The FDA recently approved a guidance document that had been in draft form for seven years. This guidance document communicated FDA’s current thinking on the supporting data needed in a 510(k) application to support various sterilization methods. The new proposal shows that newer, usually more advanced sterilization methods are lesser known to the FDA, and therefore require more supporting information to be included in the submission.
While the FDA clearly supports innovative sterilization methods, the agency requires supporting data for the processes due to the critical role it plays in product development. I believe this to be a prudent regulatory approach.
Other Factors Driving Advanced Manufacturing
FDA-regulated products are becoming more complicated. Combination products that incorporate drugs/biologics and medical devices benefit from advanced manufacturing processes because they combine entities. Micro devices often require advanced manufacturing methods because of their size, and high-cost materials necessitate such production processes to reduce waste.
Summary
Regulatory oversight and advanced manufacturing processes each impact the other in profound ways. In general, regulatory agencies like the FDA move slower than industry would like in order to properly perform their mission; manufacturers, though, benefit from regulators’ due diligence because these gatekeepers help keep the industry in check. Ultimately, the interaction between regulatory agencies and medtech manufacturers is mutually beneficial. The public, however, is the true beneficiary, served by the joint effort between regulators and manufacturers to improve patient health and safety.
James A. “Jim” Dunning’s consulting career began in 2001. He has provided quality and regulatory consulting services for various companies ranging from Fortune 500 medical device firms to startups. Dunning’s passion, however, lies with startups and small companies, especially those in regulatory distress. He has amassed significant experience in preparing 510(k) applications, developing complete Quality Management Systems, providing Quality System Training, and advising on quality, business, and leadership issues. Dunning is a senior member of the American Society for Quality (ASQ) and a member of the Regulatory Affairs Professional Society (RAPS). He can be reached at jdunning@qpcservices.com.