Michael Barbella, Managing Editor05.19.17
The caller sounded desperate. Yet there was also a bit of boldness to the voice summoning Steven S. Shin, M.D., to duty that day—an odd mix of despondence and daredevilry, tinged with just the slightest bit of angst. It was a paradoxical phone call of sorts, familiar in structure but almost surreal in tone, as if the caller was somehow directed to Shin.
He wasn’t, of course, though it certainly seemed that way. The caller, a highly competitive amateur athlete, had fractured his thumb and wanted a quick fix to get back in the game. He contacted Shin, an Ivy League-educated orthopedic surgeon and hand consultant to five Los Angeles-area professional sports teams (the doctor deems retired L.A. Lakers shooting guard Kobe Bryant a “very down to earth and pleasant patient”).
The athlete’s injury was fairly routine: He had sustained a Bennett fracture—i.e., a break and dislocation of his right (dominant) thumb metacarpal base. Common in hard-hitting contact sports such as football, boxing, and rugby, these highly unstable fractures typically extend into the joint between the metacarpal and wrist bone.
Treatment is relatively straightforward, depending on the injury’s severity. Bennett fractures involving dislocations are usually surgically repaired using wires, plates, pins, or screws, while uncomplicated breaks are simply casted.
Shin is an ardent hardware proponent, having fixed countless Bennett breaks with pins and casts during his 18-year medical career. He would have chosen that option for his recreational athlete, too, had the able-bodied man not begged for an expedited alternative to the traditional six-week healing process.
Shin found that surrogate in the Mini TightRope product line from Naples, Fla.-based Arthrex Inc. Developed to treat carpometacarpal (CMC) arthritis, the Mini TightRope procedure stabilizes the thumb with a solid suture and small buttons after the trapezium is removed (this bone is usually extracted to relieve “grinding” pain). A FiberWire suture construct is passed through the first and second metacarpal bones and secured with an oblong stainless steel button on each side. The suture provides immediate support for the thumb, thereby sparing the patient’s own tendon.
The product is considered a game-changer by many surgeons for its biomechanical stability (comparable to K-wire fixation), faster rehabilitation, and shorter recovery period. Patients undergoing the CMC Mini Tightrope procedure typically begin therapy in five to seven days rather than the conventional four weeks.
“I’ve become very familiar with the Mini Tightrope, which I’ve used for my revision thumb CMC joint arthroplasties,” Shin wrote in a 2016 blog posted on Arthrex’s website. “The Mini Tightrope does such a good job of suspending the thumb metacarpal after excision of the trapezium, so why not use this [technique] for cases of thumb CMC joint instability?”
Good point. Why not?
Shin posed this question to his strapping patient-athlete, who “jumped” at the option without really knowing anything about the procedure. Shin used the same surgical approach for thumb CMC joint arthritis on the athlete’s Bennett fracture, first passing a FiberWire suture around the thumb metacarpal base to lasso a bone fragment into place. He tied the knot dorsally and buried it in the thenar muscles, then placed the Mini Tightrope across the bases of the thumb and index metacarpals to stabilize the thumb CMC joint.
Within days of the surgery, the athlete was moving his injured thumb.
Versatile products like the Mini Tightrope are likely to become more common in the coming decades with the growing prevalence of both traumatic and sports-related injuries as well as obesity, diabetes, and geriatric-exclusive conditions like arthritis, osteoporosis, and osteomyelitis. The global trauma and extremities market is expected to reach $15.9 billion by 2024,1 with internal fixation devices—particularly plates and screws—driving much of the growth due to their clinical benefits (shorter procedure times, expedited recovery, early functionality) and multifaceted design.
“With extremities, the intricacy of the anatomy is very different so we need to develop plates, screws, and solutions that may be appropriate for the average patient’s anatomy, but it also needs to be customizable,” noted Emad Abdelnaby, senior director, Trauma and CMF (craniomaxillofacial) at DePuy Synthes Companies. “Traditional plates and screws won’t necessarily work in such a complex part of the anatomy.”
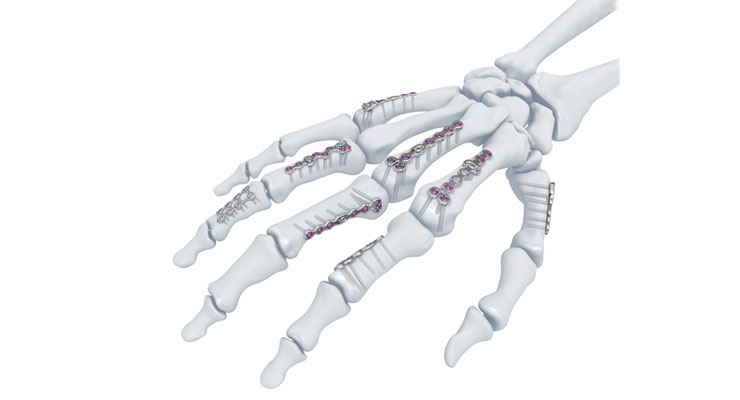
Indeed, tradition is often forsaken in the extremities space to accommodate significantly smaller anatomies and more challenging repair jobs. Consider DePuy’s Variable Angle Locking Hand System, for example: Launched last summer, the device is comprised of 40 basic and specific plates for various types of hand fractures and hand sizes, thus enabling surgeons to adjust the insertion specific to the patient’s bone anatomy to improve stability.
The first to feature 1.3 mm locking screws for hand plating, DePuy’s Variable Angle Locking Hand System also includes 1.5 and 2 mm variable angle locking plates. The thin plates have anatomic contours and smooth surfaces to minimize soft tissue irritation during contact with tendons. Recessed screw heads also reduce the potential for soft tissue irritation. The plates are available in both stainless steel and titanium, and intended for both lateral and direct dorsal application.
“...We manufacture products for spine, for trauma, for craniomaxillofacial, for both small and large joints. But customers need to be served very differently in each of these segments, and it’s important to recognize that the anatomy is very different, therefore the products must be different,” Abdelnaby told Orthopedic Design & Technology. “There is core trauma, which typically encompasses larger bone structures and then there are extremities. The bone structure in extremities is different than in a tibia or a hip.”
Markedly different, actually. The tibia and hip have relatively simple architecture—the tibia (shin bone) forms the knee joint with the femur and the ankle joint with the fibula and tarsus, while the hip joint connects the os coxal (hip) and femur, the latter being the longest, heaviest, and strongest bone in the body.
The hand, conversely, is comprised of 27 distinct bones, each of which is a fraction of the size of a major joint connector. The scaphoid, for instance—the large carpal bone buttressing the radius below the thumb—is roughly the size and shape of a medium cashew.
Such discrepancies in size and structure require creative yet comprehensive solutions that integrate fracture pattern, location, bone quality, and implant placement. One comparable elixir is offered by Addison, Texas-based OsteoMed, which manufactures a hand plating system that gives surgeons locking, non-locking, and cannulated repair options. The solution encompasses five screw types, three K-Wires, 53 plates, and angled locking technology up to 22 degrees; the low-profile titanium plates come in four sizes (1.2-2.4 mm), and are anatomically contoured with a polished surface for biocompatibility with bone and soft tissue. Moreover, the system’s headless screws feature narrow trailing diameters, making them ideal remedies for distal interphalangeal joint injuries and scaphoid fractures.
Structural diversity is also inherent in Acumed LLC’s Acu-Loc 2 Wrist Plating System, a solution designed to treat multiple fracture patterns of the distal radius and distal ulna. Offering surgeons 48 different wrist plating options, the Acu-Loc 2 features various anatomically pre-contoured plates that help restore original bone geometry and mend both intra-articular or extra-articular fractures. The system improves upon its predecessor (the original Acu-Loc) with a two-part cannulated compression screw that captures and stabilizes dorsal and volar fragments, and 2.3 mm locking variable angle screws designed to help capture specific bone fragments. Additionally, the compression screw—dubbed Frag-Loc—can be inserted through a small incision, making it a more attractive option for patients and surgeons riding the minimally invasive bandwagon.
“The natural evolution of the orthopedic market from largely open, less instrumented fusions to less invasive, highly reproducible, motion-preserving approaches has, for the most part, already occurred in other areas of orthopedics such as large joint and spine,” explained Ryan D. Schlotterback, chief commercial officer for Nextremity Solutions Inc., a privately held musculoskeletal product development company based in Warsaw, Ind. “Those of us in the extremity space are challenged to address these same market needs and bring solutions that help make our surgeons better while allowing our patients to get back to daily life sooner with less pain. We still have some work to do to bring our clinical outcomes up to par with some of the other areas of orthopedics.”
Nextremity is doing its part to raise the clinical bar with its Re+Line Bunion Correction System and Nextra Hammertoe Correction System.
The Re+Line device is a low-profile tension-band compression plate designed to help surgeons perform precise, repeatable bunion correction procedures. The technology, developed specifically for small and large correction Chevron/Austin osteotomies, allows for medial plate positioning, and resists deforming forces while providing compression across the osteotomy site. The Re+Line system has been associated with faster healing, earlier weight-bearing and less postoperative pain compared with other surgical techniques.
Some of those same clinical benefits are possible with the Nextra Hammertoe device, a system touted by the company as “the industry’s only adjustable two-piece hammertoe implant.” Completely reversible and fully adjustable in-situ, the product features a natural 10-degree angulation and a design that enhances precision, adjustability, bone-to- bone apposition, and stability.
Studies have shown the Hammertoe device to be clinically superior to traditional Kirschner Wires (K-Wires), achieving a bone fusion rate more than five times higher than its conventional counterpart (84 percent vs. 16 percent). Patient satisfaction levels are better too, as recipients have reported greater pain relief, higher activity levels, and fewer related disabilities with the Nextra implant.
“We believe we’ll continue to see less invasive, more robust and reproducible solutions come to the extremity space,” Schlotterback said. “Not many patients enjoy, nor can they afford, being off their feet for six to eight weeks. Our surgeons believe there are better ways to approach these pathologies. Biologics and some of the newer resorbable materials will continue to play a larger role.”
So will carbon fiber technology and nickel-titanium alloys.
Carbon fiber is a relative newcomer to the orthopedic arena, having been worshipped for years by the automotive and aerospace industries for its incredible strength-to-weight ratio. But it is quickly gaining favor with implant manufacturers for its high heat tolerance, elasticity, corrosion resistance, and radiolucency.
Helping spearhead the carbon fiber revolution is CarboFix Orthopedics Ltd. of Herzeliya, Israel. The company has developed continuous carbon fiber-reinforced PEEK (polyether ether ketone) implants that are stronger, more pliable, and more durable than traditional metal. In comparative fatigue experiments of Ø8.5 mm nails, CarboFix nails lasted more than nine times longer than stainless steel rods and 100 times longer than titanium rods, according to company data (4 million cycles vs. 440,000 and 40,000 cycles, respectively).
The material’s radiolucency renders CarboFix implants nearly invisible on imaging scans, thereby allowing physicians to better gauge the healing process. The products, in fact, have contrived such an effective vanishing act that surgeons have dubbed the company’s carbon fiber nails the “invisible nail.” To keep them from being completely hidden, CarboFix implants are marked with faint radio-opaque markers, and some of the products have predrilled holes so they can be attached with standard metal screws.
“There are all sorts of new technologies emerging in the orthopedic medical device world,” declared Dave Neal, president and founder of STAT Design LLC, a Morris Plains, N.J.-based outsourcing partner for the orthopedic industry. “Special CAD software, such as that by nTopology, adds porous structure to designs assisting with the bone in-growth feature in implants. Materials such as PEEK are being used more, along with the addition of titanium coatings applied on this material.”
Indeed, titanium has long been medicine’s metal of choice due to its corrosion resistance, flexibility, strength, and biocompatibility—attributes that are nearly identical to carbon fiber polymers. The most common types of medical-grade titanium are 6AL4V and 6AL4V ELI, alloys comprised of 6 percent aluminum and
4 percent vanadium.
When combined with nickel, however, titanium becomes a radically different alloy, transforming itself into a virtual self-forming elastic. The amalgamated material, known as nitinol, essentially can change and recover its shape even in the face of opposing forces. It can recover up to 10 times more strain and possesses more deformability compared with traditional metals, enabling it to respond better to loading changes.
A staple in the cardiovascular sector for at least two decades, nitinol is now finding more widespread use in orthopedics as a material for guidewires, staples, anchors, and internal fixation products. In bone fusion and fracture fixation devices, nitinol “provides both the capacity for sustained compression during postoperative healing and the opportunity to minimize device stiffness and mitigate stress shielding compared to rigid titanium implants,” according to an American Society for Metals paper.
One of the first nitinol implant manufacturers was BioMedical Enterprises Inc. (BME), a privately held firm purchased by DePuy Synthes last summer for an undisclosed sum. The San Antonio, Texas-based company developed musculoskeletal implants to treat bunions, hammertoes, and other foot and ankle deformities as well as hand and wrist deformities; its products were sold ready-to-use on a sterilized, pre-loaded, fully disposable insertion tool. The deal boosted DePuy’s portfolio of foot, ankle, hand, and wrist products.
“Small companies have the best chance of success by making their extremity devices so attractive that the larger companies are seeking them out for an acquisition,” Neal said. “In the past, the big orthopedic companies tended to ignore the [extremities] market, opting to concentrate on the larger hip and knee markets. Now they are adding this segment to their portfolio either through in-house product development or through an acquisition.”
Or, in the case of Zimmer Holdings Inc., both. The company significantly expanded its reach into the global extremity market with the $13.4 billion purchase of former rival Biomet Inc. in 2015. The combined firm’s sports medicine, extremities, and trauma (SET) division is its third-largest unit, responsible for 21 percent of total company revenue in 2016, up from 20.2 percent the previous year and 16.3 percent in 2012.
Zimmer Biomet also augmented its SET portfolio with the April 2016 purchase of Cayenne Medical Inc., a designer, developer, and seller of soft tissue repair and reconstruction products for knees, shoulders, and extremities; and its global distribution agreement with Nextremity Solutions. The October 2016 deal positions Zimmer Biomet to distribute Nextremity Solutions’ Nextra hammertoe correction system, MSP metatarsal shortening system, Re+Line bunion correction system and Arcus stable system.
“To stay innovative, Zimmer Biomet believes it is important to focus on unmet needs for the patient, for the surgeon, and for the hospital. We believe that it is imperative to consider each of these stakeholders when contemplating new solution strategies,” observed Steve Vankoski, Zimmer Biomet’s senior director of global marketing, Extremities. “Zimmer Biomet also contends that it must continue to focus on those underserved patients who may benefit from orthopedic innovation that is simply not available to them yet. And lastly, to not pigeonhole one’s thinking and look for assets and opportunities outside of the extremities market that may serve as solutions in the extremities space. Innovation can be achieved through organic growth or acquisitions.”
Organic growth at Zimmer Biomet stems partly from the company’s foray into patient-specific instrumentation and reverse shoulder arthroplasties. Debuting in the United States in 2003, reverse shoulder arthroplasties have become more prevalent in the last decade, thanks to a rise in sports-related and traumatic injuries (particularly rotator cuff tears), age-related conditions (arthritis), and better fracture management. The global market for this procedure is estimated to swell more than 12 percent over the next three years, with stemless designs fueling much of the growth.
In tapping this previously underserved sector, Zimmer Biomet developed the Trabecular Metal Reverse Shoulder System, a solution featuring Trabecular metal base plates and 60-degree humeral components designed to reduce the likelihood of scapular notching in patients with rotator cuff deficiencies. Two years ago, the company began offering the system with Vivacit-E (vitamin E) ultra-high molecular weight polyethylene liners, an option that gives surgeons a high-tech bearing surface for better oxidative stability and low wear. The device has been used in more than 32,000 procedures worldwide since 2006.
Zimmer Biomet’s most recent shoulder market entry is the Comprehensive Vault Reconstruction System (VRS), a patient-matched glenoid implant for victims of severely deficient rotator cuffs and/or extensive bone loss. The system uses computed tomography imaging and advanced three-dimensional reconstruction techniques to create an implant model that helps surgeons plan for the placement, size, and trajectory of the central and peripheral screws, ensuring the best possible fixation.
The model is used to create customized implants that fill bone voids and can accommodate a bone loss envelope of approximately 50 by 50 by 35 mm based on pre-surgical evaluation. When enough bone exists to enhance fixation in the deficient glenoid vault, surgeons may add a custom boss to the implant along with a matching custom guide. The implant model, bone model, and custom boss reaming guide are all made from a sterilizable material and are intended to be single-use disposable instruments as well as tools for the initial plan review.
“With the VRS, we can develop any shape glenoid as long as the implant falls within a certain volumetric space and utilizes a specific number of screws, ensuring the implant fits like a glove,” Vankoski explained. “Additionally, there is no need to remove any more bone than the patient has already lost. More than 60 VRS cases have been performed since September [2016]. While this volume is not tremendously high, the technology is valuable because it offers customization, helping patients who may not have any other solution.”
While such trump cards are becoming a fundamental part of most large-joint solutions, they have yet to really infiltrate the extremity sector. Zimmer Biomet’s PSI Shoulder and Signature Glenoid system are among the few offerings available for small bone specialists; other options are provided by Wright Medical Technologies Inc., which markets the PROPHECY patient-specific total ankle guides, and Additive Orthopaedic LLC’s customized 3D printed titanium digital fusion implant. The latter product received U.S. Food and Drug Administration (FDA) 510(k) clearance last summer.
3D printing is giving patients further recourse against hard-to-treat conditions as well. Last year, Zimmer Biomet introduced a 3D-printed technology for joint fusion therapy in the foot and ankle, and Chinese doctors used 3D-printed grafts in two ankle joint deformity surgeries. 4WEB Medical has also targeted the planet’s $3.82 billion foot and ankle device market with its line of 74 osteotomy implants.
A lower extremity focus is bound to serve these companies well, as the worldwide foot and ankle sector is forecast to grow 7.2 percent annually over the next three years to reach $5.41 billion by 2020. Research firm Markets and Markets attributes the surge in market value to an increase in sports-related injuries, diabetes, and arthritis cases; an influx of new products; and a greater number of injured baby boomers.
Smith & Nephew plc is taking aim at this lucrative market through the toe. The United Kingdom-based orthopedic behemoth developed a hammertoe solution comprised of three separate repair options. Its HAT-TRICK Lesser Toe Repair System features products for metatarsophalangeal ligament repair and reconstruction, a metatarsal osteotomy guide, and a revisable, all-PEEK implant for proximal Inter-phalanges fusion.
The company is also fighting for market share with the help of its BST-CarGel, a first-line cartilage repair product it acquired in early 2016 from Montreal-based Piramal Healthcare Limited. Approved for use in various countries, including Australia, Canada and most of Europe, BST-CarGel is a biopolymer-based solution that is mixed with a patient’s blood and implanted into the joint after a microfracture procedure. The solution can be delivered arthroscopically to treat damaged synovial joint cartilage in the knee, hip, ankle, and shoulder. Once implanted, it acts as a scaffold, adhering to the cartilage surface to stabilize the blood clot while new cartilage is regenerated.
“The extremity market is ready for continued innovation to improve mobility, reduce pain, and increase patient satisfaction,” reflected John Clausen, vice president of trauma and extremities marketing at Smith and Nephew. “This can be seen with such products as the Smith & Nephew HAT-TRICK and VLP Mini-Mod. We are just starting to see the large joint companies switch their engineering focus to extremity solutions. This competition will spur the smaller companies to increase their innovation strength as well. With this much focus on small joints, we are entering into a very exciting time to see some unique product innovations which will benefit patients.”
References
To read the participants' full responses, please view the following links:
More of a Niche Market - Dave Neal, president and founder of STAT Design LLC
Innovation With Speed - Ryan D. Schlotterback, chief commercial officer of Nextremity Solutions Inc.
Big Trends Towards Small Bone Plating - John Clausen, vice president of trauma and extremities marketing for Smith & Nephew plc
Extremities Market is Still Maturing - Steve Vankoski, senior director of global marketing for Zimmer Biomet
The Intricacy of Extremity Anatomy - Emad Abdelnaby, senior director, Trauma and CMF for DePuy Synthes
He wasn’t, of course, though it certainly seemed that way. The caller, a highly competitive amateur athlete, had fractured his thumb and wanted a quick fix to get back in the game. He contacted Shin, an Ivy League-educated orthopedic surgeon and hand consultant to five Los Angeles-area professional sports teams (the doctor deems retired L.A. Lakers shooting guard Kobe Bryant a “very down to earth and pleasant patient”).
The athlete’s injury was fairly routine: He had sustained a Bennett fracture—i.e., a break and dislocation of his right (dominant) thumb metacarpal base. Common in hard-hitting contact sports such as football, boxing, and rugby, these highly unstable fractures typically extend into the joint between the metacarpal and wrist bone.
Treatment is relatively straightforward, depending on the injury’s severity. Bennett fractures involving dislocations are usually surgically repaired using wires, plates, pins, or screws, while uncomplicated breaks are simply casted.
Shin is an ardent hardware proponent, having fixed countless Bennett breaks with pins and casts during his 18-year medical career. He would have chosen that option for his recreational athlete, too, had the able-bodied man not begged for an expedited alternative to the traditional six-week healing process.
Shin found that surrogate in the Mini TightRope product line from Naples, Fla.-based Arthrex Inc. Developed to treat carpometacarpal (CMC) arthritis, the Mini TightRope procedure stabilizes the thumb with a solid suture and small buttons after the trapezium is removed (this bone is usually extracted to relieve “grinding” pain). A FiberWire suture construct is passed through the first and second metacarpal bones and secured with an oblong stainless steel button on each side. The suture provides immediate support for the thumb, thereby sparing the patient’s own tendon.
The product is considered a game-changer by many surgeons for its biomechanical stability (comparable to K-wire fixation), faster rehabilitation, and shorter recovery period. Patients undergoing the CMC Mini Tightrope procedure typically begin therapy in five to seven days rather than the conventional four weeks.
“I’ve become very familiar with the Mini Tightrope, which I’ve used for my revision thumb CMC joint arthroplasties,” Shin wrote in a 2016 blog posted on Arthrex’s website. “The Mini Tightrope does such a good job of suspending the thumb metacarpal after excision of the trapezium, so why not use this [technique] for cases of thumb CMC joint instability?”
Good point. Why not?
Shin posed this question to his strapping patient-athlete, who “jumped” at the option without really knowing anything about the procedure. Shin used the same surgical approach for thumb CMC joint arthritis on the athlete’s Bennett fracture, first passing a FiberWire suture around the thumb metacarpal base to lasso a bone fragment into place. He tied the knot dorsally and buried it in the thenar muscles, then placed the Mini Tightrope across the bases of the thumb and index metacarpals to stabilize the thumb CMC joint.
Within days of the surgery, the athlete was moving his injured thumb.
Versatile products like the Mini Tightrope are likely to become more common in the coming decades with the growing prevalence of both traumatic and sports-related injuries as well as obesity, diabetes, and geriatric-exclusive conditions like arthritis, osteoporosis, and osteomyelitis. The global trauma and extremities market is expected to reach $15.9 billion by 2024,1 with internal fixation devices—particularly plates and screws—driving much of the growth due to their clinical benefits (shorter procedure times, expedited recovery, early functionality) and multifaceted design.
“With extremities, the intricacy of the anatomy is very different so we need to develop plates, screws, and solutions that may be appropriate for the average patient’s anatomy, but it also needs to be customizable,” noted Emad Abdelnaby, senior director, Trauma and CMF (craniomaxillofacial) at DePuy Synthes Companies. “Traditional plates and screws won’t necessarily work in such a complex part of the anatomy.”
Indeed, tradition is often forsaken in the extremities space to accommodate significantly smaller anatomies and more challenging repair jobs. Consider DePuy’s Variable Angle Locking Hand System, for example: Launched last summer, the device is comprised of 40 basic and specific plates for various types of hand fractures and hand sizes, thus enabling surgeons to adjust the insertion specific to the patient’s bone anatomy to improve stability.
The first to feature 1.3 mm locking screws for hand plating, DePuy’s Variable Angle Locking Hand System also includes 1.5 and 2 mm variable angle locking plates. The thin plates have anatomic contours and smooth surfaces to minimize soft tissue irritation during contact with tendons. Recessed screw heads also reduce the potential for soft tissue irritation. The plates are available in both stainless steel and titanium, and intended for both lateral and direct dorsal application.
“...We manufacture products for spine, for trauma, for craniomaxillofacial, for both small and large joints. But customers need to be served very differently in each of these segments, and it’s important to recognize that the anatomy is very different, therefore the products must be different,” Abdelnaby told Orthopedic Design & Technology. “There is core trauma, which typically encompasses larger bone structures and then there are extremities. The bone structure in extremities is different than in a tibia or a hip.”
Markedly different, actually. The tibia and hip have relatively simple architecture—the tibia (shin bone) forms the knee joint with the femur and the ankle joint with the fibula and tarsus, while the hip joint connects the os coxal (hip) and femur, the latter being the longest, heaviest, and strongest bone in the body.
The hand, conversely, is comprised of 27 distinct bones, each of which is a fraction of the size of a major joint connector. The scaphoid, for instance—the large carpal bone buttressing the radius below the thumb—is roughly the size and shape of a medium cashew.
Such discrepancies in size and structure require creative yet comprehensive solutions that integrate fracture pattern, location, bone quality, and implant placement. One comparable elixir is offered by Addison, Texas-based OsteoMed, which manufactures a hand plating system that gives surgeons locking, non-locking, and cannulated repair options. The solution encompasses five screw types, three K-Wires, 53 plates, and angled locking technology up to 22 degrees; the low-profile titanium plates come in four sizes (1.2-2.4 mm), and are anatomically contoured with a polished surface for biocompatibility with bone and soft tissue. Moreover, the system’s headless screws feature narrow trailing diameters, making them ideal remedies for distal interphalangeal joint injuries and scaphoid fractures.
Structural diversity is also inherent in Acumed LLC’s Acu-Loc 2 Wrist Plating System, a solution designed to treat multiple fracture patterns of the distal radius and distal ulna. Offering surgeons 48 different wrist plating options, the Acu-Loc 2 features various anatomically pre-contoured plates that help restore original bone geometry and mend both intra-articular or extra-articular fractures. The system improves upon its predecessor (the original Acu-Loc) with a two-part cannulated compression screw that captures and stabilizes dorsal and volar fragments, and 2.3 mm locking variable angle screws designed to help capture specific bone fragments. Additionally, the compression screw—dubbed Frag-Loc—can be inserted through a small incision, making it a more attractive option for patients and surgeons riding the minimally invasive bandwagon.
“The natural evolution of the orthopedic market from largely open, less instrumented fusions to less invasive, highly reproducible, motion-preserving approaches has, for the most part, already occurred in other areas of orthopedics such as large joint and spine,” explained Ryan D. Schlotterback, chief commercial officer for Nextremity Solutions Inc., a privately held musculoskeletal product development company based in Warsaw, Ind. “Those of us in the extremity space are challenged to address these same market needs and bring solutions that help make our surgeons better while allowing our patients to get back to daily life sooner with less pain. We still have some work to do to bring our clinical outcomes up to par with some of the other areas of orthopedics.”
Nextremity is doing its part to raise the clinical bar with its Re+Line Bunion Correction System and Nextra Hammertoe Correction System.
The Re+Line device is a low-profile tension-band compression plate designed to help surgeons perform precise, repeatable bunion correction procedures. The technology, developed specifically for small and large correction Chevron/Austin osteotomies, allows for medial plate positioning, and resists deforming forces while providing compression across the osteotomy site. The Re+Line system has been associated with faster healing, earlier weight-bearing and less postoperative pain compared with other surgical techniques.
Some of those same clinical benefits are possible with the Nextra Hammertoe device, a system touted by the company as “the industry’s only adjustable two-piece hammertoe implant.” Completely reversible and fully adjustable in-situ, the product features a natural 10-degree angulation and a design that enhances precision, adjustability, bone-to- bone apposition, and stability.
Studies have shown the Hammertoe device to be clinically superior to traditional Kirschner Wires (K-Wires), achieving a bone fusion rate more than five times higher than its conventional counterpart (84 percent vs. 16 percent). Patient satisfaction levels are better too, as recipients have reported greater pain relief, higher activity levels, and fewer related disabilities with the Nextra implant.
“We believe we’ll continue to see less invasive, more robust and reproducible solutions come to the extremity space,” Schlotterback said. “Not many patients enjoy, nor can they afford, being off their feet for six to eight weeks. Our surgeons believe there are better ways to approach these pathologies. Biologics and some of the newer resorbable materials will continue to play a larger role.”
So will carbon fiber technology and nickel-titanium alloys.
Carbon fiber is a relative newcomer to the orthopedic arena, having been worshipped for years by the automotive and aerospace industries for its incredible strength-to-weight ratio. But it is quickly gaining favor with implant manufacturers for its high heat tolerance, elasticity, corrosion resistance, and radiolucency.
Helping spearhead the carbon fiber revolution is CarboFix Orthopedics Ltd. of Herzeliya, Israel. The company has developed continuous carbon fiber-reinforced PEEK (polyether ether ketone) implants that are stronger, more pliable, and more durable than traditional metal. In comparative fatigue experiments of Ø8.5 mm nails, CarboFix nails lasted more than nine times longer than stainless steel rods and 100 times longer than titanium rods, according to company data (4 million cycles vs. 440,000 and 40,000 cycles, respectively).
The material’s radiolucency renders CarboFix implants nearly invisible on imaging scans, thereby allowing physicians to better gauge the healing process. The products, in fact, have contrived such an effective vanishing act that surgeons have dubbed the company’s carbon fiber nails the “invisible nail.” To keep them from being completely hidden, CarboFix implants are marked with faint radio-opaque markers, and some of the products have predrilled holes so they can be attached with standard metal screws.
“There are all sorts of new technologies emerging in the orthopedic medical device world,” declared Dave Neal, president and founder of STAT Design LLC, a Morris Plains, N.J.-based outsourcing partner for the orthopedic industry. “Special CAD software, such as that by nTopology, adds porous structure to designs assisting with the bone in-growth feature in implants. Materials such as PEEK are being used more, along with the addition of titanium coatings applied on this material.”
Indeed, titanium has long been medicine’s metal of choice due to its corrosion resistance, flexibility, strength, and biocompatibility—attributes that are nearly identical to carbon fiber polymers. The most common types of medical-grade titanium are 6AL4V and 6AL4V ELI, alloys comprised of 6 percent aluminum and
4 percent vanadium.
When combined with nickel, however, titanium becomes a radically different alloy, transforming itself into a virtual self-forming elastic. The amalgamated material, known as nitinol, essentially can change and recover its shape even in the face of opposing forces. It can recover up to 10 times more strain and possesses more deformability compared with traditional metals, enabling it to respond better to loading changes.
A staple in the cardiovascular sector for at least two decades, nitinol is now finding more widespread use in orthopedics as a material for guidewires, staples, anchors, and internal fixation products. In bone fusion and fracture fixation devices, nitinol “provides both the capacity for sustained compression during postoperative healing and the opportunity to minimize device stiffness and mitigate stress shielding compared to rigid titanium implants,” according to an American Society for Metals paper.
One of the first nitinol implant manufacturers was BioMedical Enterprises Inc. (BME), a privately held firm purchased by DePuy Synthes last summer for an undisclosed sum. The San Antonio, Texas-based company developed musculoskeletal implants to treat bunions, hammertoes, and other foot and ankle deformities as well as hand and wrist deformities; its products were sold ready-to-use on a sterilized, pre-loaded, fully disposable insertion tool. The deal boosted DePuy’s portfolio of foot, ankle, hand, and wrist products.
“Small companies have the best chance of success by making their extremity devices so attractive that the larger companies are seeking them out for an acquisition,” Neal said. “In the past, the big orthopedic companies tended to ignore the [extremities] market, opting to concentrate on the larger hip and knee markets. Now they are adding this segment to their portfolio either through in-house product development or through an acquisition.”
Or, in the case of Zimmer Holdings Inc., both. The company significantly expanded its reach into the global extremity market with the $13.4 billion purchase of former rival Biomet Inc. in 2015. The combined firm’s sports medicine, extremities, and trauma (SET) division is its third-largest unit, responsible for 21 percent of total company revenue in 2016, up from 20.2 percent the previous year and 16.3 percent in 2012.
Zimmer Biomet also augmented its SET portfolio with the April 2016 purchase of Cayenne Medical Inc., a designer, developer, and seller of soft tissue repair and reconstruction products for knees, shoulders, and extremities; and its global distribution agreement with Nextremity Solutions. The October 2016 deal positions Zimmer Biomet to distribute Nextremity Solutions’ Nextra hammertoe correction system, MSP metatarsal shortening system, Re+Line bunion correction system and Arcus stable system.
“To stay innovative, Zimmer Biomet believes it is important to focus on unmet needs for the patient, for the surgeon, and for the hospital. We believe that it is imperative to consider each of these stakeholders when contemplating new solution strategies,” observed Steve Vankoski, Zimmer Biomet’s senior director of global marketing, Extremities. “Zimmer Biomet also contends that it must continue to focus on those underserved patients who may benefit from orthopedic innovation that is simply not available to them yet. And lastly, to not pigeonhole one’s thinking and look for assets and opportunities outside of the extremities market that may serve as solutions in the extremities space. Innovation can be achieved through organic growth or acquisitions.”
Organic growth at Zimmer Biomet stems partly from the company’s foray into patient-specific instrumentation and reverse shoulder arthroplasties. Debuting in the United States in 2003, reverse shoulder arthroplasties have become more prevalent in the last decade, thanks to a rise in sports-related and traumatic injuries (particularly rotator cuff tears), age-related conditions (arthritis), and better fracture management. The global market for this procedure is estimated to swell more than 12 percent over the next three years, with stemless designs fueling much of the growth.
In tapping this previously underserved sector, Zimmer Biomet developed the Trabecular Metal Reverse Shoulder System, a solution featuring Trabecular metal base plates and 60-degree humeral components designed to reduce the likelihood of scapular notching in patients with rotator cuff deficiencies. Two years ago, the company began offering the system with Vivacit-E (vitamin E) ultra-high molecular weight polyethylene liners, an option that gives surgeons a high-tech bearing surface for better oxidative stability and low wear. The device has been used in more than 32,000 procedures worldwide since 2006.
Zimmer Biomet’s most recent shoulder market entry is the Comprehensive Vault Reconstruction System (VRS), a patient-matched glenoid implant for victims of severely deficient rotator cuffs and/or extensive bone loss. The system uses computed tomography imaging and advanced three-dimensional reconstruction techniques to create an implant model that helps surgeons plan for the placement, size, and trajectory of the central and peripheral screws, ensuring the best possible fixation.
The model is used to create customized implants that fill bone voids and can accommodate a bone loss envelope of approximately 50 by 50 by 35 mm based on pre-surgical evaluation. When enough bone exists to enhance fixation in the deficient glenoid vault, surgeons may add a custom boss to the implant along with a matching custom guide. The implant model, bone model, and custom boss reaming guide are all made from a sterilizable material and are intended to be single-use disposable instruments as well as tools for the initial plan review.
“With the VRS, we can develop any shape glenoid as long as the implant falls within a certain volumetric space and utilizes a specific number of screws, ensuring the implant fits like a glove,” Vankoski explained. “Additionally, there is no need to remove any more bone than the patient has already lost. More than 60 VRS cases have been performed since September [2016]. While this volume is not tremendously high, the technology is valuable because it offers customization, helping patients who may not have any other solution.”
While such trump cards are becoming a fundamental part of most large-joint solutions, they have yet to really infiltrate the extremity sector. Zimmer Biomet’s PSI Shoulder and Signature Glenoid system are among the few offerings available for small bone specialists; other options are provided by Wright Medical Technologies Inc., which markets the PROPHECY patient-specific total ankle guides, and Additive Orthopaedic LLC’s customized 3D printed titanium digital fusion implant. The latter product received U.S. Food and Drug Administration (FDA) 510(k) clearance last summer.
3D printing is giving patients further recourse against hard-to-treat conditions as well. Last year, Zimmer Biomet introduced a 3D-printed technology for joint fusion therapy in the foot and ankle, and Chinese doctors used 3D-printed grafts in two ankle joint deformity surgeries. 4WEB Medical has also targeted the planet’s $3.82 billion foot and ankle device market with its line of 74 osteotomy implants.
A lower extremity focus is bound to serve these companies well, as the worldwide foot and ankle sector is forecast to grow 7.2 percent annually over the next three years to reach $5.41 billion by 2020. Research firm Markets and Markets attributes the surge in market value to an increase in sports-related injuries, diabetes, and arthritis cases; an influx of new products; and a greater number of injured baby boomers.
Smith & Nephew plc is taking aim at this lucrative market through the toe. The United Kingdom-based orthopedic behemoth developed a hammertoe solution comprised of three separate repair options. Its HAT-TRICK Lesser Toe Repair System features products for metatarsophalangeal ligament repair and reconstruction, a metatarsal osteotomy guide, and a revisable, all-PEEK implant for proximal Inter-phalanges fusion.
The company is also fighting for market share with the help of its BST-CarGel, a first-line cartilage repair product it acquired in early 2016 from Montreal-based Piramal Healthcare Limited. Approved for use in various countries, including Australia, Canada and most of Europe, BST-CarGel is a biopolymer-based solution that is mixed with a patient’s blood and implanted into the joint after a microfracture procedure. The solution can be delivered arthroscopically to treat damaged synovial joint cartilage in the knee, hip, ankle, and shoulder. Once implanted, it acts as a scaffold, adhering to the cartilage surface to stabilize the blood clot while new cartilage is regenerated.
“The extremity market is ready for continued innovation to improve mobility, reduce pain, and increase patient satisfaction,” reflected John Clausen, vice president of trauma and extremities marketing at Smith and Nephew. “This can be seen with such products as the Smith & Nephew HAT-TRICK and VLP Mini-Mod. We are just starting to see the large joint companies switch their engineering focus to extremity solutions. This competition will spur the smaller companies to increase their innovation strength as well. With this much focus on small joints, we are entering into a very exciting time to see some unique product innovations which will benefit patients.”
References
To read the participants' full responses, please view the following links:
More of a Niche Market - Dave Neal, president and founder of STAT Design LLC
Innovation With Speed - Ryan D. Schlotterback, chief commercial officer of Nextremity Solutions Inc.
Big Trends Towards Small Bone Plating - John Clausen, vice president of trauma and extremities marketing for Smith & Nephew plc
Extremities Market is Still Maturing - Steve Vankoski, senior director of global marketing for Zimmer Biomet
The Intricacy of Extremity Anatomy - Emad Abdelnaby, senior director, Trauma and CMF for DePuy Synthes
Dave Neal, president and founder of Morris Plains, N.J.-based STAT Design LLC - See more at: http://www.odtmag.com/contents/view_online-exclusives/2017-06-05/more-of-a-niche-market/#sthash.EpiMCFNG.dpuf