Dr. Kevin Nelson, Founder and CSO, TissueGen Inc.05.19.17
Progress in implantable and topical fiber-based delivery of biologics and pharmaceuticals is paving the way for the next generation of orthopedic medical products and breakthroughs in tissue engineering and regenerative medicine. In the past, the types of biologics and pharmaceuticals that could be loaded and delivered from implantable fibers while remaining viable had been limited by the fiber manufacturing process itself. Now, a patented wet extrusion manufacturing method is enabling production of fibers loaded with a broad range of drugs to facilitate controlled, sustained delivery to targeted sites within the body.
Controlled, sustained biologics or pharmaceutical delivery from biodegradable fibers has the potential to revolutionize many orthopedic applications. These drug-loaded fibers can now serve as a critical component in medical textiles for a wide range of applications, including the repair of soft tissue. Since human tissue targeted in orthopedic procedures (e.g., the repair of tendons, ligaments, and muscle tissue) is inherently fibrous in nature, fiber-based medical devices are highly compatible with surrounding tissue structure and organization.
A New Approach to Drug Delivery
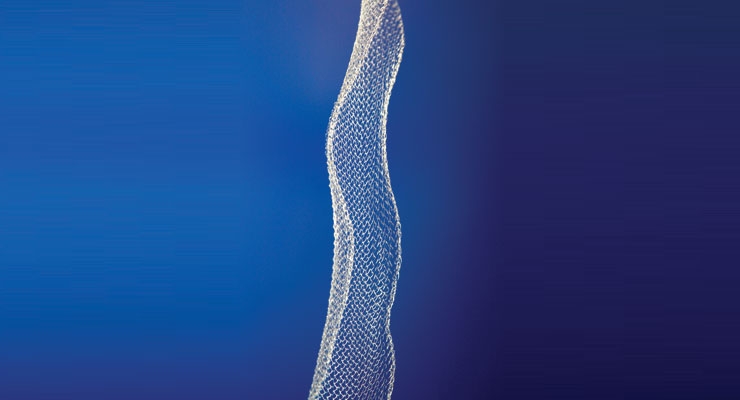
Medical fibers have typically been manufactured through melt extrusion at high temperatures and high shear stress—conditions that have severely limited the types of biologics and pharmaceuticals that could remain viable through the manufacturing process. To address these limitations, a patented wet-extrusion process has been developed for manufacturing biologics- and drug-loaded fiber (Figure 1), which occurs at or below body temperature and with relatively low shear stress. Coupled with proprietary drug protection technology, the viability of incorporated drugs is protected throughout the fiber manufacturing process so that a broad range of biologics and pharmaceuticals remain viable and may be delivered in controlled, sustained release profiles from fiber.
The Advantages of Drug-Loaded Fiber
Biologics- and pharmaceutical-loaded fiber can be offered in many physical formats—monofilament, hollow, bi-component (core-sheath), and even gel-filled center, resulting in round, flat, rectangular, or ribbon-shaped designs. The dimensions of the fiber may also be tailored for each particular application. Biologics- and pharmaceutical-loaded fiber is sufficiently strong to be woven, knitted, or braided, making it ideal for medical textile applications that may benefit from localized, non-systemic drug delivery.
Unlike traditional drug delivery formats such as microspheres and nanoparticles, biologics- and pharmaceutical-loaded fiber provides both mechanical and pharmacological support from the same device—a potentially significant advantage over other modes of drug delivery. Further, since these fibers can be extruded as a multi-layered, co-axial fiber with each layer containing a unique drug and polymer combination, the fiber may provide tailored release kinetics for multiple drugs from a single material.
Biologic and pharmaceutical-loaded fiber offers several other unique advantages over traditional drug delivery formats. Its long, cylindrical geometry provides a slower drug release rate than similar radius spherical geometries, resulting in inherently longer therapeutic windows for similar pharmaceutical concentrations. In addition, when it is manufactured in a highly controlled wet extrusion process, the resulting fibers are more uniform in size distribution than typically found with other formats. These fibers are also both readily implantable and maintain positional stability following implantation—a unique advantage when targeting specific tissue sites such as solid tumors. Finally, biologics- and pharmaceutical-loaded fiber may decrease toxicity risk to patients since it can be explanted in the case of an adverse reaction unlike microspheres and nanoparticles.
Modified Wet Extrusion for Drug-Loaded Fiber Manufacturing
Traditional melt extrusion of medical grade polymers typically exposes these materials to high shear stress and temperatures that often surpass 200 degrees Celsius—conditions that exceed retained viability limits for the vast majority of biologics and pharmaceuticals. As a result, the range of drugs that may be loaded and delivered with retained viability from melt-extrusion manufactured fibers is very limited.
Alternatively, wet extrusion has been proven to overcome the temperature and sheer stress limitations of melt extrusion. In the wet extrusion process, a polymer solution under pressure is injected through a spinneret into a coagulating bath. The coagulating bath is comprised of a solution that is highly miscible with the solvent used to dissolve the polymer and also a non-solvent for the polymer. As the polymer solution stream enters the coagulating bath, the solvent diffuses from the solution into the coagulating bath, thereby increasing the polymer concentration within the stream. Simultaneously, the polymer stream is exposed to the non-solvent of the coagulation bath and the combined effect causes the polymer molecules to precipitate out of solution to form a solid fiber. The polymer-based fiber is then pulled from the coagulation bath through a number of draw stations to stretch the fiber and align the polymer chains. While these draw stations typically incorporate ovens to heat the fiber during the drawing process, the temperature ranges are typically limited to those found in the human body. Through this drawing process, the residual solvents and non-solvents from the coagulating bath provide the molecular mobility required to align the polymer chains and create entanglement sites that impart high mechanical strength to the fiber.
While the solvents and non-solvents used in the wet extrusion process enable fiber formation at or below body temperature, most solvents and non-solvents required to produce fiber also destroy incorporated biologics and pharmaceuticals. To address this issue, proprietary methods have been developed to isolate the biologic or pharmaceutical from the harsh solvent system, thereby protecting the drug during extrusion. Prior to use in medical applications, however, the residual solvents must be removed from the fiber via a post-processing method to decrease residual solvent content to levels below the allowable limits set by the U.S. Food and Drug Administration. These post-processing methods must not denature the biologics or pharmaceuticals loaded to the biodegradable fiber so the drug retains viability post-manufacturing.
Enabling the Future of Medicine
Biologics- and pharmaceutical-loaded fiber is an ideal drug delivery platform for incorporation into a broad range of topical and implantable medical devices that may benefit from controlled release of biologics and pharmaceuticals with targeted, non-systemic delivery. Medical devices loaded with these fibers have the potential to revolutionize tissue engineering and regenerative medicine applications including spinal cord injury repair, nerve regeneration, orthopedic soft tissue repair, and many more.
Controlled, sustained drug delivery from biodegradable fiber may enable medical device developers to direct the body’s response and help orchestrate the healing process. With tailored release profiles of selected biologics or pharmaceuticals delivered from fiber, developers can mitigate unwanted reactions and promote desired tissue responses. Because incorporated drugs release at the surface of the fiber, therapeutic agents may be delivered to the local tissue rather than through systemic administration. This fiber-based drug delivery technology is a significant step forward in implantable drug delivery.
Biologics- and pharmaceutical-loaded fiber is well-suited for a wide range of current medical applications, including meshes (Figure 2), braids and weaves for medical textile applications, sutures, ligatures, and cardiovascular stents. Medical textiles constructed of these biodegradable fibers may serve as a tissue scaffold to deliver growth factors selectively and direct cell migration and tissue growth, thereby enabling tissue engineering and regenerative medicine applications.
Growth factors associated with tissue regeneration, acting as chemotactic agents, may be loaded in biodegradable fibers and can then be delivered to create concentration gradients to direct tissue growth. Even viral particles have been loaded into biodegradable fiber, subsequently implanted into immune-compromised animals, and shown to result in localized transfection of targeted cells.
Advantages for Tissue Engineering and Nerve Regeneration
When used as a three-dimensional scaffold, biodegradable fiber can create positionally stable chemical or biological concentration gradients along specific pathways. This is possible by either creating a concentration gradient down the length of a single fiber or by creating multiple fibers with uniform drug distribution and variable spacing within the scaffold. In fact, biodegradable fiber loaded with chemotactic factors for specific cell types can be used to create concentration gradients that encourage the target cells to migrate along pathways that are precisely determined by the fiber and effectively orchestrate parts of the wound healing process.
Beyond the many uses in advanced drug delivery, biologics- and pharmaceutical-loaded biodegradable fiber presents paradigm-shifting opportunities for nerve regeneration and tissue engineering applications. Both peripheral and central nervous system (CNS) repair—including spinal cord injury repair—may soon be possible.
Tissue Engineering
Medical textiles incorporating biologics- and pharmaceutical-loaded fiber hold tremendous promise for many tissue engineering applications. These fibers enable medical textiles to serve as active scaffolding for tissue engineering and regenerative medicine applications. These scaffolds may deliver growth factors to selectively direct cell migration and tissue growth. These resulting structures may enable improved site-specific drug release of more complex biologic combinations than ever before possible.
Biologics- and pharmaceutical-loaded fiber can now serve as a critical component in medical textiles, monofilament sutures, and ligatures such as those commonly found in orthopedic applications including ligament and tendon repair. Many components of the human body, particularly tissues involved in orthopedics such as tendons, ligaments, and muscle tissue, are inherently fibrous in nature. The fibrous biology of normal tissue suggests that a wound-healing device incorporating biodegradable fiber may be highly compatible with surrounding tissue structure and organization.
Nerve Regeneration
Spinal cord and CNS injuries are complex and the path to recovery has many challenges. The CNS is biologically different from the peripheral nervous system (PNS), especially in terms of wound healing. Research has shown that while axons do attempt to regenerate following injury, there are many roadblocks that impede functional recovery. For example, myelin (a substance that ensheathes many of the axons in our bodies to speed up the electrical signal conduction) contains proteins that inhibit the growth of the axons following injury. When the PNS is injured, the myelin is rapidly degraded by white blood cells and Schwann cells—the type of cells that form the myelin in the PNS. In CNS injury, however, clearance of myelin occurs much less effectively, creating a growth-prohibitive environment. In the PNS, Schwann cells produce nerve growth factor (NGF)—the beckoning call to regenerating axons to induce and guide their growth. The CNS has a diminished ability to provide this growth support following injury. Furthermore, the glial cells in the CNS form scar tissue very rapidly following injury, resulting in a glial scar consisting of growth inhibitory substances. This glial scar is highly effective at preventing regenerating axons from bridging even very small gaps. Biologics- and pharmaceutical-loaded fiber may address these challenges.
These fibers can enable the creation of three-dimensional concentration gradients of neurotrophic factors that are positionally stable over time. The resulting gradient scaffolds may be surgically implanted into an injured spinal cord in an orientation to create concentration gradients of the various neurotrophic factors that may selectively entice motor and sensory axons to cross the lesion in opposite directions in the injured spinal cord.
Medical devices are being developed with biodegradable fiber to advance the treatment and recovery of patients with spinal cord injuries. Currently in animal trials, TissueGen is applying biodegradable fiber drug-delivery technology to repair previously irreversible spinal cord injuries by delivering growth factors directly to the injury site. This approach may promote nerve regeneration without requiring tissue grafts or stem cell treatment. This approach could be indicated in patients with an acute spinal cord injury and may also apply to chronic injuries. As such, the number of potential recipients of this groundbreaking approach could total more than 250,000 in the United States alone.
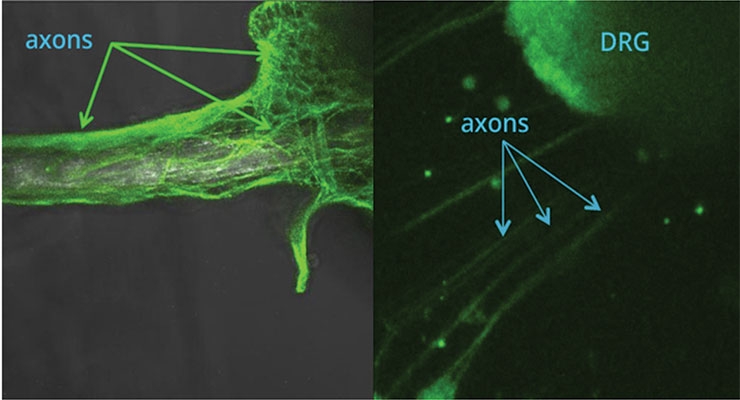
Biologics- and pharmaceutical-loaded fiber may also promote peripheral nerve regeneration. For example, parallel arrays of biodegradable fiber provide excellent scaffolding for guiding neurons. Fiber loaded with biologically active neurotrophic factors has been shown to attract neurons from isolated dorsal root ganglia (DRG) explants in cell culture experiments as shown in Figure 3. The image on the left depicts axons extending across and encapsulating a fiber that is loaded with NGF. These axons are derived from a DRG explant that is located off the photo to the right. The image on the right depicts a DRG explant extending axons directionally to the lower left side of the figure toward an NGF-loaded fiber not visible in the photograph.
Conclusion
Biologics- and pharmaceutical-loaded biodegradable fiber is advancing the state-of-the-art for controlled, sustained delivery from topical and implantable medical devices. Modified wet extrusion processes coupled with proprietary fiber manufacturing methods overcome the problems of heat and shear stress traditionally associated with melt extrusion and provide the potential benefit of stability and structural support not possible from other drug delivery formats such as microspheres and nanoparticles. Since patented manufacturing processes occur at or below body temperature, a broader range of sensitive biologics and pharmaceuticals than ever before possible may now be delivered from biodegradable implantable devices.
Since fiber can be both readily implantable and maintain positional stability, it offers a unique advantage when targeting specific sites within the body, enabling an entirely new approach to medical applications in advanced drug delivery, nerve regeneration, and tissue engineering.
Fiber’s advantages over traditional pharmaceutical delivery methods include:
Dr. Kevin Nelson is the founder and CSO of TissueGen Inc. He received his Ph.D. from The University of Texas Southwestern Medical Center at Dallas under the direction of Dr. Robert Eberhart. As a faculty member in Biomedical Engineering at the University of Texas at Arlington in 1996, Dr. Nelson joined a team working on an NIH grant to develop a fiber-based, biodegradable vascular stent with the goal of delivering gene therapy to the vessel wall. Simultaneously working with Dr. Nathan Schwade to develop drug-loaded microspheres for improved wound healing, he eventually combined the drug-loading techniques of microspheres with the fiber for the biodegradable stent, and fiber-based drug delivery was born. Eventually patented, this technology has been the focus of Dr. Nelson’s professional life and the driver behind TissueGen Inc.
Controlled, sustained biologics or pharmaceutical delivery from biodegradable fibers has the potential to revolutionize many orthopedic applications. These drug-loaded fibers can now serve as a critical component in medical textiles for a wide range of applications, including the repair of soft tissue. Since human tissue targeted in orthopedic procedures (e.g., the repair of tendons, ligaments, and muscle tissue) is inherently fibrous in nature, fiber-based medical devices are highly compatible with surrounding tissue structure and organization.
A New Approach to Drug Delivery
Medical fibers have typically been manufactured through melt extrusion at high temperatures and high shear stress—conditions that have severely limited the types of biologics and pharmaceuticals that could remain viable through the manufacturing process. To address these limitations, a patented wet-extrusion process has been developed for manufacturing biologics- and drug-loaded fiber (Figure 1), which occurs at or below body temperature and with relatively low shear stress. Coupled with proprietary drug protection technology, the viability of incorporated drugs is protected throughout the fiber manufacturing process so that a broad range of biologics and pharmaceuticals remain viable and may be delivered in controlled, sustained release profiles from fiber.
The Advantages of Drug-Loaded Fiber
Biologics- and pharmaceutical-loaded fiber can be offered in many physical formats—monofilament, hollow, bi-component (core-sheath), and even gel-filled center, resulting in round, flat, rectangular, or ribbon-shaped designs. The dimensions of the fiber may also be tailored for each particular application. Biologics- and pharmaceutical-loaded fiber is sufficiently strong to be woven, knitted, or braided, making it ideal for medical textile applications that may benefit from localized, non-systemic drug delivery.
Unlike traditional drug delivery formats such as microspheres and nanoparticles, biologics- and pharmaceutical-loaded fiber provides both mechanical and pharmacological support from the same device—a potentially significant advantage over other modes of drug delivery. Further, since these fibers can be extruded as a multi-layered, co-axial fiber with each layer containing a unique drug and polymer combination, the fiber may provide tailored release kinetics for multiple drugs from a single material.
Biologic and pharmaceutical-loaded fiber offers several other unique advantages over traditional drug delivery formats. Its long, cylindrical geometry provides a slower drug release rate than similar radius spherical geometries, resulting in inherently longer therapeutic windows for similar pharmaceutical concentrations. In addition, when it is manufactured in a highly controlled wet extrusion process, the resulting fibers are more uniform in size distribution than typically found with other formats. These fibers are also both readily implantable and maintain positional stability following implantation—a unique advantage when targeting specific tissue sites such as solid tumors. Finally, biologics- and pharmaceutical-loaded fiber may decrease toxicity risk to patients since it can be explanted in the case of an adverse reaction unlike microspheres and nanoparticles.
Modified Wet Extrusion for Drug-Loaded Fiber Manufacturing
Traditional melt extrusion of medical grade polymers typically exposes these materials to high shear stress and temperatures that often surpass 200 degrees Celsius—conditions that exceed retained viability limits for the vast majority of biologics and pharmaceuticals. As a result, the range of drugs that may be loaded and delivered with retained viability from melt-extrusion manufactured fibers is very limited.
Alternatively, wet extrusion has been proven to overcome the temperature and sheer stress limitations of melt extrusion. In the wet extrusion process, a polymer solution under pressure is injected through a spinneret into a coagulating bath. The coagulating bath is comprised of a solution that is highly miscible with the solvent used to dissolve the polymer and also a non-solvent for the polymer. As the polymer solution stream enters the coagulating bath, the solvent diffuses from the solution into the coagulating bath, thereby increasing the polymer concentration within the stream. Simultaneously, the polymer stream is exposed to the non-solvent of the coagulation bath and the combined effect causes the polymer molecules to precipitate out of solution to form a solid fiber. The polymer-based fiber is then pulled from the coagulation bath through a number of draw stations to stretch the fiber and align the polymer chains. While these draw stations typically incorporate ovens to heat the fiber during the drawing process, the temperature ranges are typically limited to those found in the human body. Through this drawing process, the residual solvents and non-solvents from the coagulating bath provide the molecular mobility required to align the polymer chains and create entanglement sites that impart high mechanical strength to the fiber.
While the solvents and non-solvents used in the wet extrusion process enable fiber formation at or below body temperature, most solvents and non-solvents required to produce fiber also destroy incorporated biologics and pharmaceuticals. To address this issue, proprietary methods have been developed to isolate the biologic or pharmaceutical from the harsh solvent system, thereby protecting the drug during extrusion. Prior to use in medical applications, however, the residual solvents must be removed from the fiber via a post-processing method to decrease residual solvent content to levels below the allowable limits set by the U.S. Food and Drug Administration. These post-processing methods must not denature the biologics or pharmaceuticals loaded to the biodegradable fiber so the drug retains viability post-manufacturing.
Enabling the Future of Medicine
Biologics- and pharmaceutical-loaded fiber is an ideal drug delivery platform for incorporation into a broad range of topical and implantable medical devices that may benefit from controlled release of biologics and pharmaceuticals with targeted, non-systemic delivery. Medical devices loaded with these fibers have the potential to revolutionize tissue engineering and regenerative medicine applications including spinal cord injury repair, nerve regeneration, orthopedic soft tissue repair, and many more.
Controlled, sustained drug delivery from biodegradable fiber may enable medical device developers to direct the body’s response and help orchestrate the healing process. With tailored release profiles of selected biologics or pharmaceuticals delivered from fiber, developers can mitigate unwanted reactions and promote desired tissue responses. Because incorporated drugs release at the surface of the fiber, therapeutic agents may be delivered to the local tissue rather than through systemic administration. This fiber-based drug delivery technology is a significant step forward in implantable drug delivery.
Biologics- and pharmaceutical-loaded fiber is well-suited for a wide range of current medical applications, including meshes (Figure 2), braids and weaves for medical textile applications, sutures, ligatures, and cardiovascular stents. Medical textiles constructed of these biodegradable fibers may serve as a tissue scaffold to deliver growth factors selectively and direct cell migration and tissue growth, thereby enabling tissue engineering and regenerative medicine applications.
Growth factors associated with tissue regeneration, acting as chemotactic agents, may be loaded in biodegradable fibers and can then be delivered to create concentration gradients to direct tissue growth. Even viral particles have been loaded into biodegradable fiber, subsequently implanted into immune-compromised animals, and shown to result in localized transfection of targeted cells.
Advantages for Tissue Engineering and Nerve Regeneration
When used as a three-dimensional scaffold, biodegradable fiber can create positionally stable chemical or biological concentration gradients along specific pathways. This is possible by either creating a concentration gradient down the length of a single fiber or by creating multiple fibers with uniform drug distribution and variable spacing within the scaffold. In fact, biodegradable fiber loaded with chemotactic factors for specific cell types can be used to create concentration gradients that encourage the target cells to migrate along pathways that are precisely determined by the fiber and effectively orchestrate parts of the wound healing process.
Beyond the many uses in advanced drug delivery, biologics- and pharmaceutical-loaded biodegradable fiber presents paradigm-shifting opportunities for nerve regeneration and tissue engineering applications. Both peripheral and central nervous system (CNS) repair—including spinal cord injury repair—may soon be possible.
Tissue Engineering
Medical textiles incorporating biologics- and pharmaceutical-loaded fiber hold tremendous promise for many tissue engineering applications. These fibers enable medical textiles to serve as active scaffolding for tissue engineering and regenerative medicine applications. These scaffolds may deliver growth factors to selectively direct cell migration and tissue growth. These resulting structures may enable improved site-specific drug release of more complex biologic combinations than ever before possible.
Biologics- and pharmaceutical-loaded fiber can now serve as a critical component in medical textiles, monofilament sutures, and ligatures such as those commonly found in orthopedic applications including ligament and tendon repair. Many components of the human body, particularly tissues involved in orthopedics such as tendons, ligaments, and muscle tissue, are inherently fibrous in nature. The fibrous biology of normal tissue suggests that a wound-healing device incorporating biodegradable fiber may be highly compatible with surrounding tissue structure and organization.
Nerve Regeneration
Spinal cord and CNS injuries are complex and the path to recovery has many challenges. The CNS is biologically different from the peripheral nervous system (PNS), especially in terms of wound healing. Research has shown that while axons do attempt to regenerate following injury, there are many roadblocks that impede functional recovery. For example, myelin (a substance that ensheathes many of the axons in our bodies to speed up the electrical signal conduction) contains proteins that inhibit the growth of the axons following injury. When the PNS is injured, the myelin is rapidly degraded by white blood cells and Schwann cells—the type of cells that form the myelin in the PNS. In CNS injury, however, clearance of myelin occurs much less effectively, creating a growth-prohibitive environment. In the PNS, Schwann cells produce nerve growth factor (NGF)—the beckoning call to regenerating axons to induce and guide their growth. The CNS has a diminished ability to provide this growth support following injury. Furthermore, the glial cells in the CNS form scar tissue very rapidly following injury, resulting in a glial scar consisting of growth inhibitory substances. This glial scar is highly effective at preventing regenerating axons from bridging even very small gaps. Biologics- and pharmaceutical-loaded fiber may address these challenges.
These fibers can enable the creation of three-dimensional concentration gradients of neurotrophic factors that are positionally stable over time. The resulting gradient scaffolds may be surgically implanted into an injured spinal cord in an orientation to create concentration gradients of the various neurotrophic factors that may selectively entice motor and sensory axons to cross the lesion in opposite directions in the injured spinal cord.
Medical devices are being developed with biodegradable fiber to advance the treatment and recovery of patients with spinal cord injuries. Currently in animal trials, TissueGen is applying biodegradable fiber drug-delivery technology to repair previously irreversible spinal cord injuries by delivering growth factors directly to the injury site. This approach may promote nerve regeneration without requiring tissue grafts or stem cell treatment. This approach could be indicated in patients with an acute spinal cord injury and may also apply to chronic injuries. As such, the number of potential recipients of this groundbreaking approach could total more than 250,000 in the United States alone.
Biologics- and pharmaceutical-loaded fiber may also promote peripheral nerve regeneration. For example, parallel arrays of biodegradable fiber provide excellent scaffolding for guiding neurons. Fiber loaded with biologically active neurotrophic factors has been shown to attract neurons from isolated dorsal root ganglia (DRG) explants in cell culture experiments as shown in Figure 3. The image on the left depicts axons extending across and encapsulating a fiber that is loaded with NGF. These axons are derived from a DRG explant that is located off the photo to the right. The image on the right depicts a DRG explant extending axons directionally to the lower left side of the figure toward an NGF-loaded fiber not visible in the photograph.
Conclusion
Biologics- and pharmaceutical-loaded biodegradable fiber is advancing the state-of-the-art for controlled, sustained delivery from topical and implantable medical devices. Modified wet extrusion processes coupled with proprietary fiber manufacturing methods overcome the problems of heat and shear stress traditionally associated with melt extrusion and provide the potential benefit of stability and structural support not possible from other drug delivery formats such as microspheres and nanoparticles. Since patented manufacturing processes occur at or below body temperature, a broader range of sensitive biologics and pharmaceuticals than ever before possible may now be delivered from biodegradable implantable devices.
Since fiber can be both readily implantable and maintain positional stability, it offers a unique advantage when targeting specific sites within the body, enabling an entirely new approach to medical applications in advanced drug delivery, nerve regeneration, and tissue engineering.
Fiber’s advantages over traditional pharmaceutical delivery methods include:
- Controlled, sustained delivery at therapeutic dosages
- Longer therapeutic windows for sensitive drugs
- Localized, non-systemic delivery of drugs at the target site
- Tailored release of multiple drugs from a single fiber
- Removable as needed in the event of a negative response to the device
Dr. Kevin Nelson is the founder and CSO of TissueGen Inc. He received his Ph.D. from The University of Texas Southwestern Medical Center at Dallas under the direction of Dr. Robert Eberhart. As a faculty member in Biomedical Engineering at the University of Texas at Arlington in 1996, Dr. Nelson joined a team working on an NIH grant to develop a fiber-based, biodegradable vascular stent with the goal of delivering gene therapy to the vessel wall. Simultaneously working with Dr. Nathan Schwade to develop drug-loaded microspheres for improved wound healing, he eventually combined the drug-loading techniques of microspheres with the fiber for the biodegradable stent, and fiber-based drug delivery was born. Eventually patented, this technology has been the focus of Dr. Nelson’s professional life and the driver behind TissueGen Inc.