Sam Brusco, Associate Editor05.19.17
It’s not often one hears, “Guess the 40th time’s the charm!”
For then-fledgling San Diego, Calif.-based Rocket Chemical Company, it took 40 attempts to get its water displacing formula to work. In 1953, the small lab and its three-person staff achieved the invention of a line of rust-prevention solvents and degreasers to be used in the aerospace industry. And although it had taken 39 failed attempts before getting it right, on the 40th, the company succeeded in a big way—WD-40 was born. (WD-40, by the way, stands for Water Displacement, 40th formula.)
Initially, aerospace contractor Convair used WD-40 to prevent the skin of the Atlas Missile from rusting and corroding. It was so successful in this endeavor that employees snuck cans of WD-40 home in their lunchboxes for household use. In fact, it was so useful around the home for tasks like extricating jammed screws and bolts and loosening stubborn zippers that Rocket Chemical Company made WD-40 available to San Diego consumers in 1958, and it was written up as a new consumer product in 1961. By 1965, airlines including Delta and United were using WD-40 on fixed and movable joints of their DC-8 and Boeing 720s for maintenance and overhaul.
WD-40’s claim to fame is a non-volatile, viscous oil that remains on its application surface for a long period of time, providing both lubrication and protection from moisture that is quite useful in both commercial and domestic settings. Since its creation, WD-40’s chemistry has been tuned and upgraded to support high temperature, pressure, and moisture environments as well as to last over 10 times longer than the original formula. According to the company, about four out of five households have a can of WD-40 in their home, and the official list enumerates more than 2,000 uses for the surface-treating oil.
Naturally, people began to believe that WD-40 could be used for creaky joints of a biological nature as well—a dire mistake. Using the lubricant as a remedy for arthritis or general joint soreness unfortunately became an unproven and potentially harmful practice. While it’s true that lubricating rusty metal joints is one of WD-40’s main uses, there are no scientific studies on safety or effectiveness on or in the body. WD-40 is also not intended to lubricate metal knee, hip, or shoulder replacements. (In fact, the bottle clearly states “could be harmful or fatal if swallowed.” That should be reason enough to avoid it.)
Surface modifications for musculoskeletal devices and implants must obviously address more than just their “squeakiness,” however. Antibacterial coatings should be applied to improve an instrument or implant’s safety, functionality, and longevity. A statistically significant number of patients are allergic to metal, so many implant manufacturers add hypoallergenic coatings to their devices. Perhaps most importantly, surface modifications are also made to enhance the structural and functional connection between living bone and the surface of a load-bearing implant.
“Some of the more common coatings we are seeing requested for orthopedic devices are geared toward instrument longevity, and enhancing wear resistance and allergy protection in implants,” noted Michael Venturini, president and CEO of Columbia City, Ind.-based DOT America Inc., a provider of specialty coatings and surface treatments for medical implants and instruments. “The most common of these coatings would be physical vapor deposition (PVD). Especially useful for patients with a high degree of sensitivity, PVD coatings such as TiNbN (titanium niobium nitride) and even TiN (titanium nitride) are extremely useful in protection from allergies as they show great results in friction erosion, eliminating the release of certain contact allergens such as nickel, cobalt, or chrome.”
“The biggest trend we see in the surface finishing of medical implants is reducing cost to surface finishing operations,” explained Brett Eldridge, sales manager, mass finishing equipment for Rösler Metal Finishing USA, a Battle Creek, Mich.-based provider of metal surface finishing and preparation. “Our mass finishing equipment, consumables, and processing expertise allow orthopedic manufactures to achieve repeatable surface finishes at lower costs. The cost savings come from eliminating various types of operations such as manual hand finishing, which can be inconsistent, and inconsistencies cause rework and scrap.”
Orthopedic devices sometimes present the challenge of a novel material composition. Polyetheretherketone (PEEK), though it’s been around for some time, is still considered “novel” as its usefulness in replacing titanium for certain musculoskeletal applications has only just begun to flourish. Some of PEEK’s material properties, however, make it less than ideal for long-term implantation, and applying a coating can help to make PEEK a better fit.
“For PEEK devices, the hydrophobic and bioinert properties of the substrate have warranted novel solutions to improve the clinical results,” illuminated Ulf Brogren, president of Austin, Texas-based Promimic U.S. Inc., a biomaterial company that develops and markets an implant surface that accelerates osseointegration. “As a result, many OEMs have started to coat PEEK with coatings such as plasma-sprayed hydroxyapatite (HA) or titanium. However, this has proven to be difficult since materials with different mechanical properties and hardness are combined. In the case of plasma sprayed HA onto PEEK, a stiff and brittle ceramic material [is] placed on a soft and flexible substrate. Naturally, these materials will respond completely different when they are subjected to stress, which can cause delamination and/or crack formation. One way around this problem is to use a coating so thin it does not introduce a load-bearing layer between the core implant and surrounding bone.”
“There are a few polymers required in orthopedic devices for their non-substitutable intrinsic value, like ultra-high-molecular-weight-polyethylene (UHMWPE), PEEK, etc.,” said Pierfrancesco Robotti, scientific marketing for Pergine Valsugana, Italy-based Eurocoating, a provider of thermal plasma spray (TPS) coatings for implantable orthopedic and dental applications. “These polymers are very elastic and quite sensitive to thermal stresses and, at the same time, are bioinert, not really osteointegrable directly. Limiting my answer to TPS technology, applying a coating to enhance affinity with bone at the surface means having all the potential consequences on the polymer itself in mind because of the coating process. Moreover, adhesion and cohesion of the coating applied on a polymer must reach certain defined minimum performances to guarantee the success of the device. On the other hand, extremely hard ceramic materials are not easy to coat with porous metal structures using TPS technology. Also, the most popular kinds of bioceramics for structural use (e.g., zirconia toughened alumina, or alumina toughened zirconia, etc.) experience a similar situation as the aforementioned polymers. Indeed they are basically bioinert and not easy to connect with porous metal structures. Using the TPS technology, a porous metal coating can be applied to change the picture at the interface with bone; again, wide considerations on consequences for the ceramic bulk must be made.”
Another common surface treatment for orthopedic devices is anodizing—an electrochemical process that converts a metal surface into a decorative, durable, corrosion-resistant finish. Typically, the anodizing process occurs on aluminum surfaces, but the process is also well-suited for titanium—a very common metal used in orthopedic instruments and implants.
“Titanium color anodizing is a multiple stage process,” explained Brian Sutton, technical director for Miraclean, an Ashville, N.Y.-based provider of precision ultrasonic cleaning equipment, nitric and citric passivations lines, automated FPI application lines, automated titanium anodize lines, and water-based chemistries. “When automated, fixtured parts automatically advance through clean, acid etch, and rinse steps that are time, temperature, and concentration controlled and monitored. The anodize tank is set up similarly to an electroplating tank with an anode, cathode, rectifier, and an electrolytic solution. The process does not color the titanium but rather creates a stable oxide layer on the surface of the titanium through the application of current. The ‘colors’ produced are actually interference perceived when wavelengths of light refract through the oxide layer and reflect off the substrate below it. Different voltages produce discretely different thicknesses of oxide layer which in turn result in different color effects. Time, temperature, chemistry concentration, and voltage all have a critical impact on the color effect that is produced.”
The process provides the advantage of hardening and coloring the surface without altering the titanium’s mechanical properties. However, though orthopedic instruments and implants are subjected to both hardening and coloring anodizing processes, they do not occur simultaneously.
“Titanium implants are commonly anodized Color or Type 2,” said Tim Zentz, sales manager for Danco Anodizing, a Warsaw, Ind.-based provider of surface preparation, anodizing, and identification techniques for metal parts. “There is no Type 3 titanium anodize, as there is no governing or regulatory body that has issued specifications for the process. Color titanium anodizing is primarily used for color identification, whereas Type 2 titanium anodizing is used for added lubricity, increased fatigue strength, or improved surface finish. Stainless steel instruments can also be passivated, electropolished, and/or low-friction chrome (LFC) coated. LFC increases the life of a stainless-steel instrument through steam autoclave up to 30-50 percent due to added corrosion resistance.”
Color anodizing orthopedic device components or instruments may seem like a purely aesthetic process, but this type of surface treatment offers benefits to the end-users—clinicians.
“Color anodizing of titanium is frequently implemented for identification purposes, for example to color-code sizes of bone screws and even the corresponding surgical instrumentation,” stated Sutton. “Bone plates and rods are also often color anodized.”
Better Bone Integration
The interface between an orthopedic implant and the surrounding tissue can have a significant effect on its clinical outcome. One of the main reasons for surface modifications is to facilitate osseointegration, the process of bone and tissue fusion to the implant to ensure implant fixation and long-term performance.
One method that has proven successful to better cozy orthopedic implants to bone and tissue is the abrasive blasting process. For example, abrasive air blasting propels a loose abrasive (such as aluminum oxide) with compressed air against the implant’s work surface to create a roughened pattern that enhances bone ingrowth. Similarly, wet blasting also uses an abrasive media and compressed air to achieve a desired finish, but adds the third element of liquid in order to broaden the levels of achievable surface finish—often resulting in a smoother and more consistent finish.
“Our mass finishing, air blasting, and wet blasting equipment have been used for surface modifications to enhance osseointegration,” explained Eldridge. “Air blasting and wet blasting are used to provide a roughened surface on the implant, which allows for bone growth to occur around the implant. Typically, the surface roughness (Ra) is greater than 200 microinches and this can be achieved with several different abrasive blast media, though this is nothing new.”
“What is new is that orthopedic customers want better repeatability on these surface finishes,” he continued. “We provide this repeatability through verified process controls in our automated air blast and wet blast machines. We can automate equipment to the extent of operators only needing to load and unload parts.”
Micro-abrasive blasting (also referred to as micro-blasting or precision blasting) uses a stream of abrasive, high-pressure, micron-sized particles propelled against a part. A small nozzle drives the stream, helping to focus and intensify it. Typically, this finishing process has been used to sanitize surgical instruments or deburr hypodermic needles, but finishing some orthopedic devices requires the equipment’s blast energy to be directed to a very small, precise area. Some are finding success in using this technique for smaller or more geometrically-complex orthopedic devices (such as those used in the extremities).
“Osseointegration is becoming a widely requested surface modification,” commented Venturini. “DOT has been successful with microblasting, using resorbable blasting medias as well as aluminum oxide to create a rough surface that promotes osseointegration while not showing any significant dimensional loss.”
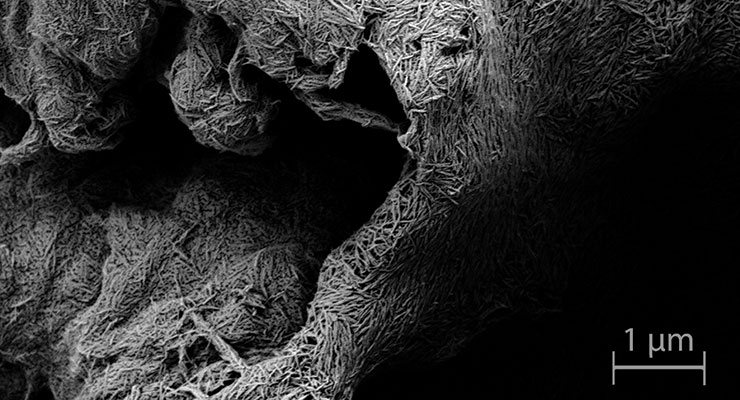
An orthopedic implant’s hydrophilic properties are important to consider for improved osseointegration as well. The first stage of implant integration involves adsorption of blood and extracellular fluids—which contain about 90 percent water—onto the implant’s surface. The proteins and ions present in these fluids that adsorb on the surface are both hydrophilic and serve as the platform for bone cell growth. As such, a hydrophilic surface serves as a better substrate for bone cell growth than a hydrophobic surface.
“An important aspect for osseointegration is the implant’s wetting behavior, i.e., if it is hydrophilic or not,” said Brogren. “A good wetting behavior of the implant will lead to a faster integration of the implant. This effect is dependent upon how the implant is handled, cleaned, and packaged.”
Coatings can also be applied to facilitate implant hydrophilicity as well as independently stimulate bone growth. One such coating material that has become quite common to facilitate osseointegration is the naturally occurring mineral form of calcium apatite called hydroxyapatite—or HA for short.
“Typically, porous coatings are requested, with absolute large porosity (>40 percent), large size pores (>200 um), with, at the same time, good enough mechanical performances to guarantee integrity under the physiological cycling loads. On top, a change of the chemical nature can be requested: for example, pass from a CoCr (cobalt chromium) bulk to a porous titanium surface, from titanium alloy to hydroxyapatite, etc.,” commented Robotti. “The most interesting novelty comes in the new couplings, such as titanium coating on PEEK, titanium coating on polyethylene, or titanium coating on ZTA (zirconia toughened alumina). However, these configurations need accurate TPS process control to guarantee solid results.”
HA coatings have become particularly sought-after for orthopedic applications largely because up to 50 percent of human bone volume is composed of HA. The material is already used as a filler to replace amputated bone and has found success in use as a coating to promote bone ingrowth in prosthetic devices. Titanium coatings have traditionally been used because they are capable of connecting structurally and functionally with bone, but HA is becoming increasingly popular due to its enhanced osseointegrative capability as a result of bone material mimicry.
HAnano Surface, one such HA-based coating, was introduced in the United States for the orthopedic market in 2016. Just 20 nanometers thin and applicable to a wide variety of implant materials, HAnano has demonstrated increased anchoring strength and integration with surrounding bone tissue.
“HAnano (which Danco-Warsaw offers through a license agreement with Promimic AB) is a 0.02-micron thin layer of hydroxyapatite that can be applied to nearly all implantable devices, including PEEK, titanium, stainless steel, 3D-printed/porous structures, cobalt chrome, etc.,” explained Zentz. “This technology not only has the added benefit of application to nearly all implantable surfaces, but also resolves regulatory concerns about the potential issue of traditional HA that can flake off.”
The coating, composed of bio-mimicking synthetic bone materials, facilitates the hydrophilic surface properties integral to an implant that will successfully integrate with bone.
“What we also know is that a coating such as HAnano Surface does not only improve the hydrophilicity initially, but that this effect is stable over time,” stated Brogren. “Thus, by choosing an appropriate coating, the wetting behavior will be enhanced also after storing of the product. Because of the increased focus on osseointegration in orthopedics, a hot topic for the coming years will be how to improve hydrophilicity of implants, and how to maintain this effect on the implant—from manufacturing until placed into the patient. Surface modifications and coatings can play an important role in this; we have shown that HAnano Surface maintains a hydrophilic implant surface over a long time.”
Covering All the Bases
Suppliers and OEMs alike have come to somewhat of a consensus that additive manufacturing (AM) is the way of the future for orthopedic devices. That technological approach also enables specific surface design—3D-printed titanium could have its porous structure, shape-morphology, porosity, or stiffness tailored to each specific use case.
“Orthopedic OEMs can take advantage of AM by building up components made of an implantable metal material with complex multilevel topography and, at the same time, complex shape,” commented Robotti. “It’s even better if these components are expected with multiple variations to accommodate the request of many sizes or the differences between left-right, or even gender differences. Moreover, AM technology shows high production flexibility to be competitively exploited both for production of custom-made parts and for massive manufacturing. With all of this, you might not need further additive treatments.”
However, some of 3D printing’s technological limitations are bringing about further applications for surface modification.
“On the other hand, most of the AM technologies allow to play with one single material per device,” Robotti continued. “In this case, surface treatment or coatings on AM parts can be attractive. However, in complex porous structures (such as those manufactured with AM), TPS can’t be the elective choice for surface modification due to its capability to coat only in the line of sight. Chemical and electrochemical treatments may better infiltrate the porous structure, bringing the surface modifications desired within the porosity. Examples are biomimetic coatings based on calcium phosphates, titanium oxide modifications, etc.”
Additive manufacturing of orthopedic implants also challenges the continued trend toward more efficient osseointegration, and provokes novel surface modification strategies.
“Additive manufacturing gives a nice macro and micro texturing, but on the nano-scale, these production methods smoothen out the surface and decrease the surface energy. Smooth surfaces on the micro and nano-scale do not improve osseointegration,” explained Brogren. “Therefore, surface modifications and coatings that introduce a roughness on the nano level and attain higher surface energy, improved wetting behavior, and higher protein adsorption will be the next generation of these devices. Obviously, to be able to modify the substrate deep into the porous structure with a coating or surface modification, it cannot be a line-of-sight technology. So, for these applications, wet chemistry solutions is the way of the future.”
In his prediction for the future of surface modification for musculoskeletal instruments and implants, Zentz anticipates that “continued focus on surface modifications that improve patient outcomes, including faster osseointegration, anti-microbial properties, and coatings that perform well in bad bone applications (osteoporatic, diabetic, smoking, etc…) will be important.”
Finally, as orthopedic device geometries become more and more complex (whether as a result of progress in additive manufacturing techniques or otherwise), those providing surface treatments have them covered.
“As device engineers bring new geometries to be color anodized or new effects they wish to create, system parameters and parts fixturing will be tweaked and refined to meet the new goals,” said Sutton. “Our motto is ‘if they dream it, we will come.’”
For then-fledgling San Diego, Calif.-based Rocket Chemical Company, it took 40 attempts to get its water displacing formula to work. In 1953, the small lab and its three-person staff achieved the invention of a line of rust-prevention solvents and degreasers to be used in the aerospace industry. And although it had taken 39 failed attempts before getting it right, on the 40th, the company succeeded in a big way—WD-40 was born. (WD-40, by the way, stands for Water Displacement, 40th formula.)
Initially, aerospace contractor Convair used WD-40 to prevent the skin of the Atlas Missile from rusting and corroding. It was so successful in this endeavor that employees snuck cans of WD-40 home in their lunchboxes for household use. In fact, it was so useful around the home for tasks like extricating jammed screws and bolts and loosening stubborn zippers that Rocket Chemical Company made WD-40 available to San Diego consumers in 1958, and it was written up as a new consumer product in 1961. By 1965, airlines including Delta and United were using WD-40 on fixed and movable joints of their DC-8 and Boeing 720s for maintenance and overhaul.
WD-40’s claim to fame is a non-volatile, viscous oil that remains on its application surface for a long period of time, providing both lubrication and protection from moisture that is quite useful in both commercial and domestic settings. Since its creation, WD-40’s chemistry has been tuned and upgraded to support high temperature, pressure, and moisture environments as well as to last over 10 times longer than the original formula. According to the company, about four out of five households have a can of WD-40 in their home, and the official list enumerates more than 2,000 uses for the surface-treating oil.
Naturally, people began to believe that WD-40 could be used for creaky joints of a biological nature as well—a dire mistake. Using the lubricant as a remedy for arthritis or general joint soreness unfortunately became an unproven and potentially harmful practice. While it’s true that lubricating rusty metal joints is one of WD-40’s main uses, there are no scientific studies on safety or effectiveness on or in the body. WD-40 is also not intended to lubricate metal knee, hip, or shoulder replacements. (In fact, the bottle clearly states “could be harmful or fatal if swallowed.” That should be reason enough to avoid it.)
Surface modifications for musculoskeletal devices and implants must obviously address more than just their “squeakiness,” however. Antibacterial coatings should be applied to improve an instrument or implant’s safety, functionality, and longevity. A statistically significant number of patients are allergic to metal, so many implant manufacturers add hypoallergenic coatings to their devices. Perhaps most importantly, surface modifications are also made to enhance the structural and functional connection between living bone and the surface of a load-bearing implant.
“Some of the more common coatings we are seeing requested for orthopedic devices are geared toward instrument longevity, and enhancing wear resistance and allergy protection in implants,” noted Michael Venturini, president and CEO of Columbia City, Ind.-based DOT America Inc., a provider of specialty coatings and surface treatments for medical implants and instruments. “The most common of these coatings would be physical vapor deposition (PVD). Especially useful for patients with a high degree of sensitivity, PVD coatings such as TiNbN (titanium niobium nitride) and even TiN (titanium nitride) are extremely useful in protection from allergies as they show great results in friction erosion, eliminating the release of certain contact allergens such as nickel, cobalt, or chrome.”
“The biggest trend we see in the surface finishing of medical implants is reducing cost to surface finishing operations,” explained Brett Eldridge, sales manager, mass finishing equipment for Rösler Metal Finishing USA, a Battle Creek, Mich.-based provider of metal surface finishing and preparation. “Our mass finishing equipment, consumables, and processing expertise allow orthopedic manufactures to achieve repeatable surface finishes at lower costs. The cost savings come from eliminating various types of operations such as manual hand finishing, which can be inconsistent, and inconsistencies cause rework and scrap.”
Orthopedic devices sometimes present the challenge of a novel material composition. Polyetheretherketone (PEEK), though it’s been around for some time, is still considered “novel” as its usefulness in replacing titanium for certain musculoskeletal applications has only just begun to flourish. Some of PEEK’s material properties, however, make it less than ideal for long-term implantation, and applying a coating can help to make PEEK a better fit.
“For PEEK devices, the hydrophobic and bioinert properties of the substrate have warranted novel solutions to improve the clinical results,” illuminated Ulf Brogren, president of Austin, Texas-based Promimic U.S. Inc., a biomaterial company that develops and markets an implant surface that accelerates osseointegration. “As a result, many OEMs have started to coat PEEK with coatings such as plasma-sprayed hydroxyapatite (HA) or titanium. However, this has proven to be difficult since materials with different mechanical properties and hardness are combined. In the case of plasma sprayed HA onto PEEK, a stiff and brittle ceramic material [is] placed on a soft and flexible substrate. Naturally, these materials will respond completely different when they are subjected to stress, which can cause delamination and/or crack formation. One way around this problem is to use a coating so thin it does not introduce a load-bearing layer between the core implant and surrounding bone.”
“There are a few polymers required in orthopedic devices for their non-substitutable intrinsic value, like ultra-high-molecular-weight-polyethylene (UHMWPE), PEEK, etc.,” said Pierfrancesco Robotti, scientific marketing for Pergine Valsugana, Italy-based Eurocoating, a provider of thermal plasma spray (TPS) coatings for implantable orthopedic and dental applications. “These polymers are very elastic and quite sensitive to thermal stresses and, at the same time, are bioinert, not really osteointegrable directly. Limiting my answer to TPS technology, applying a coating to enhance affinity with bone at the surface means having all the potential consequences on the polymer itself in mind because of the coating process. Moreover, adhesion and cohesion of the coating applied on a polymer must reach certain defined minimum performances to guarantee the success of the device. On the other hand, extremely hard ceramic materials are not easy to coat with porous metal structures using TPS technology. Also, the most popular kinds of bioceramics for structural use (e.g., zirconia toughened alumina, or alumina toughened zirconia, etc.) experience a similar situation as the aforementioned polymers. Indeed they are basically bioinert and not easy to connect with porous metal structures. Using the TPS technology, a porous metal coating can be applied to change the picture at the interface with bone; again, wide considerations on consequences for the ceramic bulk must be made.”
Another common surface treatment for orthopedic devices is anodizing—an electrochemical process that converts a metal surface into a decorative, durable, corrosion-resistant finish. Typically, the anodizing process occurs on aluminum surfaces, but the process is also well-suited for titanium—a very common metal used in orthopedic instruments and implants.
“Titanium color anodizing is a multiple stage process,” explained Brian Sutton, technical director for Miraclean, an Ashville, N.Y.-based provider of precision ultrasonic cleaning equipment, nitric and citric passivations lines, automated FPI application lines, automated titanium anodize lines, and water-based chemistries. “When automated, fixtured parts automatically advance through clean, acid etch, and rinse steps that are time, temperature, and concentration controlled and monitored. The anodize tank is set up similarly to an electroplating tank with an anode, cathode, rectifier, and an electrolytic solution. The process does not color the titanium but rather creates a stable oxide layer on the surface of the titanium through the application of current. The ‘colors’ produced are actually interference perceived when wavelengths of light refract through the oxide layer and reflect off the substrate below it. Different voltages produce discretely different thicknesses of oxide layer which in turn result in different color effects. Time, temperature, chemistry concentration, and voltage all have a critical impact on the color effect that is produced.”
The process provides the advantage of hardening and coloring the surface without altering the titanium’s mechanical properties. However, though orthopedic instruments and implants are subjected to both hardening and coloring anodizing processes, they do not occur simultaneously.
“Titanium implants are commonly anodized Color or Type 2,” said Tim Zentz, sales manager for Danco Anodizing, a Warsaw, Ind.-based provider of surface preparation, anodizing, and identification techniques for metal parts. “There is no Type 3 titanium anodize, as there is no governing or regulatory body that has issued specifications for the process. Color titanium anodizing is primarily used for color identification, whereas Type 2 titanium anodizing is used for added lubricity, increased fatigue strength, or improved surface finish. Stainless steel instruments can also be passivated, electropolished, and/or low-friction chrome (LFC) coated. LFC increases the life of a stainless-steel instrument through steam autoclave up to 30-50 percent due to added corrosion resistance.”
Color anodizing orthopedic device components or instruments may seem like a purely aesthetic process, but this type of surface treatment offers benefits to the end-users—clinicians.
“Color anodizing of titanium is frequently implemented for identification purposes, for example to color-code sizes of bone screws and even the corresponding surgical instrumentation,” stated Sutton. “Bone plates and rods are also often color anodized.”
Better Bone Integration
The interface between an orthopedic implant and the surrounding tissue can have a significant effect on its clinical outcome. One of the main reasons for surface modifications is to facilitate osseointegration, the process of bone and tissue fusion to the implant to ensure implant fixation and long-term performance.
One method that has proven successful to better cozy orthopedic implants to bone and tissue is the abrasive blasting process. For example, abrasive air blasting propels a loose abrasive (such as aluminum oxide) with compressed air against the implant’s work surface to create a roughened pattern that enhances bone ingrowth. Similarly, wet blasting also uses an abrasive media and compressed air to achieve a desired finish, but adds the third element of liquid in order to broaden the levels of achievable surface finish—often resulting in a smoother and more consistent finish.
“Our mass finishing, air blasting, and wet blasting equipment have been used for surface modifications to enhance osseointegration,” explained Eldridge. “Air blasting and wet blasting are used to provide a roughened surface on the implant, which allows for bone growth to occur around the implant. Typically, the surface roughness (Ra) is greater than 200 microinches and this can be achieved with several different abrasive blast media, though this is nothing new.”
“What is new is that orthopedic customers want better repeatability on these surface finishes,” he continued. “We provide this repeatability through verified process controls in our automated air blast and wet blast machines. We can automate equipment to the extent of operators only needing to load and unload parts.”
Micro-abrasive blasting (also referred to as micro-blasting or precision blasting) uses a stream of abrasive, high-pressure, micron-sized particles propelled against a part. A small nozzle drives the stream, helping to focus and intensify it. Typically, this finishing process has been used to sanitize surgical instruments or deburr hypodermic needles, but finishing some orthopedic devices requires the equipment’s blast energy to be directed to a very small, precise area. Some are finding success in using this technique for smaller or more geometrically-complex orthopedic devices (such as those used in the extremities).
“Osseointegration is becoming a widely requested surface modification,” commented Venturini. “DOT has been successful with microblasting, using resorbable blasting medias as well as aluminum oxide to create a rough surface that promotes osseointegration while not showing any significant dimensional loss.”
An orthopedic implant’s hydrophilic properties are important to consider for improved osseointegration as well. The first stage of implant integration involves adsorption of blood and extracellular fluids—which contain about 90 percent water—onto the implant’s surface. The proteins and ions present in these fluids that adsorb on the surface are both hydrophilic and serve as the platform for bone cell growth. As such, a hydrophilic surface serves as a better substrate for bone cell growth than a hydrophobic surface.
“An important aspect for osseointegration is the implant’s wetting behavior, i.e., if it is hydrophilic or not,” said Brogren. “A good wetting behavior of the implant will lead to a faster integration of the implant. This effect is dependent upon how the implant is handled, cleaned, and packaged.”
Coatings can also be applied to facilitate implant hydrophilicity as well as independently stimulate bone growth. One such coating material that has become quite common to facilitate osseointegration is the naturally occurring mineral form of calcium apatite called hydroxyapatite—or HA for short.
“Typically, porous coatings are requested, with absolute large porosity (>40 percent), large size pores (>200 um), with, at the same time, good enough mechanical performances to guarantee integrity under the physiological cycling loads. On top, a change of the chemical nature can be requested: for example, pass from a CoCr (cobalt chromium) bulk to a porous titanium surface, from titanium alloy to hydroxyapatite, etc.,” commented Robotti. “The most interesting novelty comes in the new couplings, such as titanium coating on PEEK, titanium coating on polyethylene, or titanium coating on ZTA (zirconia toughened alumina). However, these configurations need accurate TPS process control to guarantee solid results.”
HA coatings have become particularly sought-after for orthopedic applications largely because up to 50 percent of human bone volume is composed of HA. The material is already used as a filler to replace amputated bone and has found success in use as a coating to promote bone ingrowth in prosthetic devices. Titanium coatings have traditionally been used because they are capable of connecting structurally and functionally with bone, but HA is becoming increasingly popular due to its enhanced osseointegrative capability as a result of bone material mimicry.
HAnano Surface, one such HA-based coating, was introduced in the United States for the orthopedic market in 2016. Just 20 nanometers thin and applicable to a wide variety of implant materials, HAnano has demonstrated increased anchoring strength and integration with surrounding bone tissue.
“HAnano (which Danco-Warsaw offers through a license agreement with Promimic AB) is a 0.02-micron thin layer of hydroxyapatite that can be applied to nearly all implantable devices, including PEEK, titanium, stainless steel, 3D-printed/porous structures, cobalt chrome, etc.,” explained Zentz. “This technology not only has the added benefit of application to nearly all implantable surfaces, but also resolves regulatory concerns about the potential issue of traditional HA that can flake off.”
The coating, composed of bio-mimicking synthetic bone materials, facilitates the hydrophilic surface properties integral to an implant that will successfully integrate with bone.
“What we also know is that a coating such as HAnano Surface does not only improve the hydrophilicity initially, but that this effect is stable over time,” stated Brogren. “Thus, by choosing an appropriate coating, the wetting behavior will be enhanced also after storing of the product. Because of the increased focus on osseointegration in orthopedics, a hot topic for the coming years will be how to improve hydrophilicity of implants, and how to maintain this effect on the implant—from manufacturing until placed into the patient. Surface modifications and coatings can play an important role in this; we have shown that HAnano Surface maintains a hydrophilic implant surface over a long time.”
Covering All the Bases
Suppliers and OEMs alike have come to somewhat of a consensus that additive manufacturing (AM) is the way of the future for orthopedic devices. That technological approach also enables specific surface design—3D-printed titanium could have its porous structure, shape-morphology, porosity, or stiffness tailored to each specific use case.
“Orthopedic OEMs can take advantage of AM by building up components made of an implantable metal material with complex multilevel topography and, at the same time, complex shape,” commented Robotti. “It’s even better if these components are expected with multiple variations to accommodate the request of many sizes or the differences between left-right, or even gender differences. Moreover, AM technology shows high production flexibility to be competitively exploited both for production of custom-made parts and for massive manufacturing. With all of this, you might not need further additive treatments.”
However, some of 3D printing’s technological limitations are bringing about further applications for surface modification.
“On the other hand, most of the AM technologies allow to play with one single material per device,” Robotti continued. “In this case, surface treatment or coatings on AM parts can be attractive. However, in complex porous structures (such as those manufactured with AM), TPS can’t be the elective choice for surface modification due to its capability to coat only in the line of sight. Chemical and electrochemical treatments may better infiltrate the porous structure, bringing the surface modifications desired within the porosity. Examples are biomimetic coatings based on calcium phosphates, titanium oxide modifications, etc.”
Additive manufacturing of orthopedic implants also challenges the continued trend toward more efficient osseointegration, and provokes novel surface modification strategies.
“Additive manufacturing gives a nice macro and micro texturing, but on the nano-scale, these production methods smoothen out the surface and decrease the surface energy. Smooth surfaces on the micro and nano-scale do not improve osseointegration,” explained Brogren. “Therefore, surface modifications and coatings that introduce a roughness on the nano level and attain higher surface energy, improved wetting behavior, and higher protein adsorption will be the next generation of these devices. Obviously, to be able to modify the substrate deep into the porous structure with a coating or surface modification, it cannot be a line-of-sight technology. So, for these applications, wet chemistry solutions is the way of the future.”
In his prediction for the future of surface modification for musculoskeletal instruments and implants, Zentz anticipates that “continued focus on surface modifications that improve patient outcomes, including faster osseointegration, anti-microbial properties, and coatings that perform well in bad bone applications (osteoporatic, diabetic, smoking, etc…) will be important.”
Finally, as orthopedic device geometries become more and more complex (whether as a result of progress in additive manufacturing techniques or otherwise), those providing surface treatments have them covered.
“As device engineers bring new geometries to be color anodized or new effects they wish to create, system parameters and parts fixturing will be tweaked and refined to meet the new goals,” said Sutton. “Our motto is ‘if they dream it, we will come.’”