Maria Shepherd, President and Founder, Medi-Vantage10.02.17
Orthopedic device product innovation and pricing strategy must be closely linked. In orthopedic product development, the most important three questions are:
Defining the unmet need is only the first step. The second step is ensuring the product development process meets the unmet need of the orthopedic patient, surgeon, and payer; fits within the clinical workflow; and overcomes every predictable obstacle to use. Sustainability is assessed through device market research that identifies the market positions of direct and indirect competitors (there are so many competitors in orthopedics). Most critical (and most difficult) is naming the payer for the product, how much it is worth to them, and why?
Why This Is Important
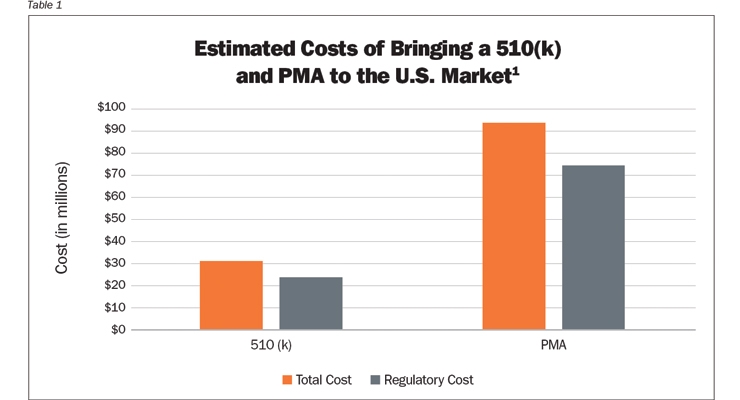
The trend in orthopedics is toward cellular-based repair, and bringing most of these products to market requires a PMA. It is expensive to bring a medical device to market. A Stanford study1 identified the average cost to bring a 510(k) product from concept to market at $31 million. Greater than 77 percent of the cost—approximately $24 million—was consumed on regulatory and U.S. Food and Drug Administration (FDA)-related activities. The cost of a PMA averaged $94 million, with $75 million spent on FDA-linked stages—nearly 80 percent of the total amount of bringing medtech devices to market launch (Table 1).
Orthopedic Device Pricing Strategy and Conjoint Analysis
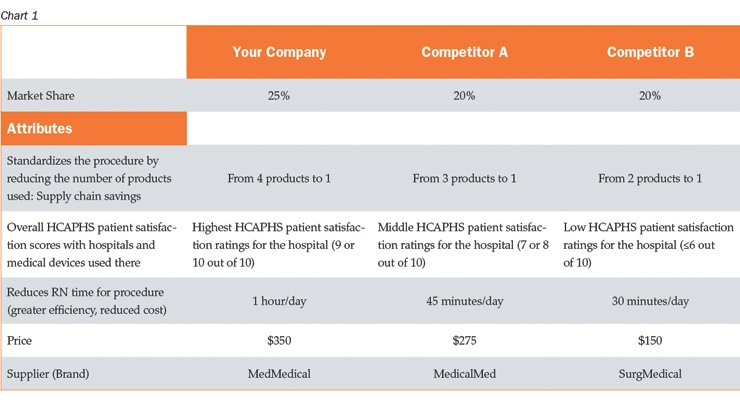
Want to know what your orthopedic surgeons are thinking when it comes to making product or pricing decisions? Conjoint analysis (also called Discrete Choice Analysis or sometimes MaxDiff) is an advanced market research strategy used in medtech to understand how surgeons (and more often now, payers and patients) make decisions about what they really value in medical device products and services. In conjoint analysis, we present surgeons, hospital administrators, insurers, and patients with choices to identify the drivers for the selections they make. The best part? You and your team end up with a market simulator (Chart 1), giving you the answers to questions like:
What Will a Market Simulator Do for Your Orthopedic Product Development Team?
The market simulator is an important tool that is the output of medtech conjoint analysis. The simulator is used to convert statistically significant conjoint data into a tool medtech executive management wants and needs—the ability to simulate market choices. A market simulator lets the product development team conduct “what-if” scenarios to investigate new product designs, product positioning, and price strategies. It helps the team identify market segments (i.e., groups of clinicians, hospital administrators, or patients with unique and targetable preferences).
HCAHPS Survey Experience Scores
If you haven’t heard about the HCAHPS survey, you will soon. HCAHPS is the first national, standardized, publicly reported survey of patients’ perspectives of hospital care. Hospitals are publicly reported on the Medicare website at www.medicare.gov/hospitalcompare. In 2012, the first HCAPHS patient surveys were performed (n=2.9 million) from 3,892 U.S. hospitals.2 The most common complaint? Forty percent of patients said hospitals are too noisy, and specifically cited hospital overhead broadcast systems. In response, U.S. hospitals have been installing smartphones and Bluetooth to encourage texting vs. placing calls or paging. In hospitals where caregivers were provided smartphones, a 78 percent decrease in overhead paging was observed.
Conclusion and Recommendations
A conjoint survey can be expensive, but with the cost of bringing a medical device to market assessed at $30 million to $94 million, the cost of a conjoint analysis and the confidence in decision-making it offers is short money. With any new orthopedic product, explore product design configurations and pricing strategies to reduce risk involved with bringing that device to market.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. Her career included a role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic. She was also director of marketing for Philips Medical and held senior management roles at Boston Scientific Corp. In 2007, she founded Medi-Vantage. Medi-Vantage provides marketing, business, and product development strategy research for the medical device, diagnostics, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught medtech marketing and product development courses and is a member of the Aligo Medtech Investment Committee. She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.
- Is there an unmet need that can be answered with an orthopedic device and what is it?
- Is the unmet need sustainable and for how long?
- Who (e.g., payer, hospital administrator, surgeon owner of an ASC) is going to pay for this product, how much, and why?
Defining the unmet need is only the first step. The second step is ensuring the product development process meets the unmet need of the orthopedic patient, surgeon, and payer; fits within the clinical workflow; and overcomes every predictable obstacle to use. Sustainability is assessed through device market research that identifies the market positions of direct and indirect competitors (there are so many competitors in orthopedics). Most critical (and most difficult) is naming the payer for the product, how much it is worth to them, and why?
Why This Is Important
The trend in orthopedics is toward cellular-based repair, and bringing most of these products to market requires a PMA. It is expensive to bring a medical device to market. A Stanford study1 identified the average cost to bring a 510(k) product from concept to market at $31 million. Greater than 77 percent of the cost—approximately $24 million—was consumed on regulatory and U.S. Food and Drug Administration (FDA)-related activities. The cost of a PMA averaged $94 million, with $75 million spent on FDA-linked stages—nearly 80 percent of the total amount of bringing medtech devices to market launch (Table 1).

Orthopedic Device Pricing Strategy and Conjoint Analysis
Want to know what your orthopedic surgeons are thinking when it comes to making product or pricing decisions? Conjoint analysis (also called Discrete Choice Analysis or sometimes MaxDiff) is an advanced market research strategy used in medtech to understand how surgeons (and more often now, payers and patients) make decisions about what they really value in medical device products and services. In conjoint analysis, we present surgeons, hospital administrators, insurers, and patients with choices to identify the drivers for the selections they make. The best part? You and your team end up with a market simulator (Chart 1), giving you the answers to questions like:
- Should we build in more features, or reduce the price?
- Which of these changes will take the most market share from our competitors?
- If we claim improved patient outcomes, what price premium will they pay?

What Will a Market Simulator Do for Your Orthopedic Product Development Team?
The market simulator is an important tool that is the output of medtech conjoint analysis. The simulator is used to convert statistically significant conjoint data into a tool medtech executive management wants and needs—the ability to simulate market choices. A market simulator lets the product development team conduct “what-if” scenarios to investigate new product designs, product positioning, and price strategies. It helps the team identify market segments (i.e., groups of clinicians, hospital administrators, or patients with unique and targetable preferences).
HCAHPS Survey Experience Scores
If you haven’t heard about the HCAHPS survey, you will soon. HCAHPS is the first national, standardized, publicly reported survey of patients’ perspectives of hospital care. Hospitals are publicly reported on the Medicare website at www.medicare.gov/hospitalcompare. In 2012, the first HCAPHS patient surveys were performed (n=2.9 million) from 3,892 U.S. hospitals.2 The most common complaint? Forty percent of patients said hospitals are too noisy, and specifically cited hospital overhead broadcast systems. In response, U.S. hospitals have been installing smartphones and Bluetooth to encourage texting vs. placing calls or paging. In hospitals where caregivers were provided smartphones, a 78 percent decrease in overhead paging was observed.
Conclusion and Recommendations
A conjoint survey can be expensive, but with the cost of bringing a medical device to market assessed at $30 million to $94 million, the cost of a conjoint analysis and the confidence in decision-making it offers is short money. With any new orthopedic product, explore product design configurations and pricing strategies to reduce risk involved with bringing that device to market.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. Her career included a role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic. She was also director of marketing for Philips Medical and held senior management roles at Boston Scientific Corp. In 2007, she founded Medi-Vantage. Medi-Vantage provides marketing, business, and product development strategy research for the medical device, diagnostics, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught medtech marketing and product development courses and is a member of the Aligo Medtech Investment Committee. She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.