ODT Staff08.15.18
ODT’s annual top companies list reflects the highest sales-generating firms developing and commercializing orthopedic technologies. Some of the most interesting innovations, however, originate with their smaller peers. Addressing an array of challenges for various clinical indications, these companies have generated attention due to their unique innovations. They may not ever make the Top 10 list, but could very well improve the care patients receive. Following are six companies that have captured the attention of the ODT editors.
Bioness & Mainstay Medical
Sam Brusco • Associate Editor
According to Centers for Disease Control and Prevention (CDC) studies, as many as one in four patients undergoing long-term therapy in primary care environments grapple with opioid-use disorder. Further, the number of U.S. opioid prescriptions and sales has increased fourfold since 1999, and the number of deaths from prescription opioid-related overdoses topped 165,000.
The “one size fits all” mentality of using opioids to manage chronic pain is chiefly to blame for these worrisome statistics. Patients—and even their clinicians—may not be aware of alternate routes to take to reduce pain to a manageable level. Anesthetic injections and/or nerve ablation treatments can temporarily quell the agony, but pain relief from ablation usually lasts a maximum of two years. For especially enduring chronic or orthopedic pain, wearable or implantable devices that deliver therapeutic neurostimulation to treat the affected nerve may offer welcome relief.
Revamping Rehab
In 2004, a group of visionaries united to establish Bioness Inc. to help those with neurological impairments regain their independence. Shortly thereafter, Alfred E. Mann—the entrepreneur and philanthropist behind Pacesetter Systems (now owned by Abbott Labs) and MiniMed (now owned by Medtronic)—gave his support to the fledgling company. As a result, Bioness was formed to develop and distribute neuromodulation devices into the field of rehabilitation. Bioness then partnered with NESS Ltd. to release two devices in the U.S.—the H200 Hand Rehabilitation System for patients with upper limb impairments, and the L300 Foot Drop System to assist those with foot drop (a dropping of the forefoot due to weakness, irritation, or damage to the common fibular nerve) in regaining a normal gait.
The greater majority of Bioness’ products—including L300 and H200—employ functional electrical stimulation (FES) to offer functional and therapeutic benefits for patients with central nervous disorders and orthopedic injuries. L300 consists of three main components: a functional stimulation cuff, control unit, and gait sensor that all communicate wirelessly. While walking, the gait sensor detects events and transmits signals to synchronize the electrical pulses, stimulating the foot muscles to lift off the ground at the appropriate time. H200 operates on the similar FES principle, but focuses on reaching, grasping, opening, and closing the hand. It can be used as part of a rehab regimen for stroke, brain injury, spinal cord injury, or any other disorders of the central nervous system resulting in upper limb functional deficits and impairments. Bioness also offers the Vector Gait Safety System, a rehabilitation module designed for physical and occupational therapy that provides patients recovering from stroke, amputations, and orthopedic, brain, and spinal cord injuries a safe and real-world environment to undergo physical therapy.
Bioness made its foray into implantable technologies with 2015’s FDA nod for StimRouter, a neuromodulation device to treat chronic peripheral pain. Rather than a large implanted component, StimRouter employs a 15 cm passive lead inserted percutaneously under local anesthesia. The tip of the lead is placed near the target peripheral nerve causing the chronic pain. The receiver is on the skin’s surface so the patient can program their level of neurostimulation.
Coming Back to Life
About two thirds of people suffer from low back pain at some point in their life. In most, the pain resolves after a few months, but for some, it becomes a chronic condition. It can begin as a simple strain or sprain of one of the joints of the spine. The brain responds to joint pain by limiting painful movements, one of which is suppressing the activation of the muscles stabilizing the lower back. As this happens, the spine ends up moving more easily into painful positions, resulting in an ongoing cycle of chronic low back pain (CLBP) and muscle weakening.
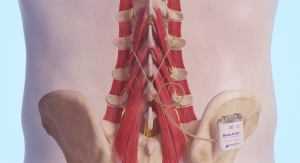
Many studies point to muscle control reactivation to break this vicious cycle. Physiotherapists can offer specific exercise programs to regain control of the spine stabilizing muscles, but sometimes conventional treatments aren’t enough to restore it. Aware of this, Dublin, Ireland-based medical device company Mainstay Medical developed the ReActiv8 implantable restorative neurostimulation system for people with disabling CLBP.
ReActiv8 is a small implanted device that stimulates the nerves responsible for activating the key muscles stabilizing the lower back. Once implanted, patients can deliver half-hour sessions twice daily via a handheld, wireless remote control. The hope is that repeated stimulation will eventually “teach” the brain and body to control the spine stabilizing muscles in between sessions, ultimately allowing the body to recover from CLBP.
The ReActiv8 implant is currently CE marked. As of July 9, all implantations for the U.S. IDE (Investigational Device Exemption) study, ReActiv8-B, have been completed. A total of 204 patients were implanted in the study, and Mainstay Medical expects to announce a full data readout from the trial toward the end of this year. The study intends to gather data to support a pre-market approval (PMA) application to the FDA in preparation for U.S. commercialization.
According to Mainstay’s annual report, the company generated $300,000 of revenue last year, entirely based on European sales. Initial activities were focused in Germany, with five centers in Germany and Ireland containing implanted patients as of fiscal 2017’s end. 2017 was also the first year Mainstay Medical posted any revenue at all. The company also has subsidiaries operating in the United States, Australia, Germany, and the Netherlands.
WishBone Medical Inc. & Pega Medical
Michael Barbella • Managing Editor
There are times Sarah Sibbel, M.D., feels more like a carpenter than a pediatric hand surgeon.
She has plates, screws, scissors, saws, hammers, and various power tools at her disposal—most of which are somewhat problematic for her patients. A (rare) specialist in brachial plexus birth palsy and congenital hand differences, Sibbel works exclusively with children, an easily overlooked and historically neglected component of the global healthcare spectrum.
“I treat kids’ hands...that’s their ultimate organ of exploration when they’re young,” Sibbel says in an online video for Children’s Hospital Colorado, where she is surgical director of the Brachial Plexus Clinic and director of the Pediatric Hand and Upper Extremity Program. “The saying is kids are not just little adults, and they’re not. They’re growing, they have growth plates, and they heal differently, oftentimes faster than adults do. I think it’s important to respect that aspect of taking care of children.”
Respect is hard to come by in pediatric orthopedics, though. Too small and generally too healthy to attract investor interest, children traditionally have been treated with jury-rigged adult devices (many of which are used off-label). Kid-sized implants are so rare, in fact, that surgeons have resorted to sawing grownup devices in half or mix-and-matching products with body parts (i.e., using spinal rods for femoral repair) in order to provide treatment.
Such makeshift solutions are becoming a bit less common nowadays as the medical device industry and the U.S. Food and Drug Administration address the longstanding shortfall in pediatric device development with grant programs and competitions. Those efforts could eventually help make the sector more appealing to financiers and boost the number of companies focused on juvenile orthopedics.
A Nod to Aunt Osti
Pediatric orthopedics apparently is part of the Deeter family heritage.
More than a century ago, Nick Deeter’s great great Aunt Osti (formal name: Ostia Lucille Smith) was one of the first female sales representatives in orthopedics for Revra DePuy, inventor of the fiber splint and founder of DePuy Manufacturing (now DePuy Synthes, part of Johnson & Johnson). Smith’s first sale was a juvenile ankle bone splint to a Florida doctor.
More than a century later, Deeter is keeping Smith’s legacy alive through his efforts to provide children with proper medical devices. He founded juvenile orthopedic implant/instrument manufacturer OrthoPediatrics in 2006, and WishBone Medical Inc. last year, becoming its CEO and board chairman.
“Ninety-five percent of the orthopedic implants going into children today are not specifically designed for them,” Deeter says on WishBone Medical’s website. “Ten years ago when I started OrthoPediatrics, I felt it was unacceptable for a child to get an adult device that was bent, cut, and altered just to try and make it fit while ignoring the growth plates and possibly causing more harm. There is still a lot of work to be done on behalf of our kids.”
Indeed there is, and WishBone Medical has wasted little time immersing itself in its mission. The Warsaw, Ind.-based firm sets itself apart from OrthoPediatrics by billing itself as a provider of “anatomically appropriate” implants and instruments in sterile packed, single-use disposable kits. The company has had a fairly active year thus far, releasing its first products in January and forging a partnership with the Hospital for Special Surgery (HSS).
WishBone Medical’s Broken Screw Removal System, available in three sizes (2.7mm, 3.5mm, 4.5mm), is designed to remove broken, stripped, and cold-welded screws. Its K-Wire Systems come in five different configurations—Pediatric Elbow Combo Kit, Pediatric Long Bone Combo Kit, and three Pediatric Extremity Combo Kits; each system includes two different sizes of wires, protective pin covers, and instruments.
The company also hammered out a U.S. distribution agreement with Implanet in March, and is collaborating with HSS to develop a pediatric hip implant. The product will specifically be designed to address the treatment of juvenile idiopathic arthritis and/or congenital dysplastic hip disease.
“We look forward to working with HSS on a Pediatric Total Hip system designed just for kids,” Deeter said in announcing the partnership.
A True VOICE for Children
Well before Deeter set out to make his Aunt Osti proud, Canadian firm Pega Medical was serving unmet needs in the pediatric orthopedic space. Founded in 1997, the Laval, Quebec-based company has developed seven different product sets for children, including pins, plates, screws, intramedullary nails, a self-extending rod, and a modular blade-plate system for femoral osteotomies and fractures.
With its products reaching more than 50 countries worldwide, Pega Medical considers itself a small international company with both a “brain” and “heart.” The latter is clearly evident in the firm’s devotion to underprivileged children worldwide suffering from bone diseases.
Through its CSR initiative, Pega Medical encourages surgeons to donate implants and instruments to their peers involved in medical missions to underdeveloped countries. Every surgeon who registers in the CSR program is given a VOICE (Virtual Orthopedic Implant Coin Exchange); surgeons who use Pega Medical devices obtain one VOICE for every implant purchased by their hospital. The VOICEs can be accumulated or donated to any cause in which a colleague is involved. The company donates one implant for every 50 mission VOICES accrued, and one instrument set for every 150 mission VOICES collected.
“We want to become a company that makes a real and durable difference for underprivileged children suffering from bone diseases around the world,” the company states on its website. “Pega Medical is recognized not only as an innovator with distinct devices reaching children with orthopedic conditions in over 50 countries around the world, but also as a company who puts the care of patients and the assistance to the surgeons as its first priority wherever they live (a small international company with a heart).”
OrthoSensor & Gramercy Extremity Orthopedics
Sean Fenske • Editor-in-Chief
The medtech industry continues to get “smarter”; that is, the devices themselves are being developed with artificial intelligence, sensors, communication-enabling technologies, and more to enhance the quality of care provided. Orthopedics, however, is one sector lagging behind in this trend. The implants going into patients today are very similar in terms of functionality to their counterparts of recent years. Indeed, fabrication techniques improve, materials may vary, and surfaces are modified with different treatments, but overall, the implants are no “smarter” than they were in the past.
In recent years, several companies have emerged seeking to change the “intelligence” behind orthopedic devices. The two companies featured in the following section have put products onto the market that offer smarter solutions to more traditional options. OrthoSensor has developed Verasense, which helps surgeons ensure the correct balancing of an implant during total knee arthroplasty. Gramercy Extremity Orthopedics provides a cart system to make extremities surgical procedures, as well as component ordering, much more efficient.
A Balancing Act
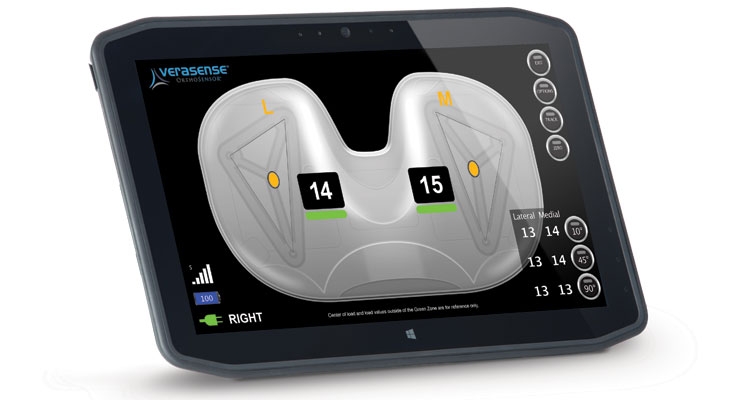
Founded in 2006, OrthoSensor’s vision is to utilize data to improve the decision-making process in orthopedic procedures. Toward this end, the company has developed the Verasense device. This technology is used to balance the knee implant during a primary or revision total knee replacement procedure. An implantable, disposable component couples wirelessly with an intra-operative monitor that offers surgeons feedback on the implant positioning in real time. It eliminates the “feel” aspect many surgeons rely on during a knee implant procedure. Upon use, the device is discarded and replaced with a permanent total knee replacement system component.
The results speak for themselves. According to the company, in a two-year multicenter study involving 135 patients, data reflected a 97 percent satisfaction rating from patients (i.e., an indication of “satisfied” or “very satisfied” at two-years post-op was provided). The study also revealed that knees balanced with Verasense achieved a KSS score of 178.6, which was 17.6 points higher than the average score of unbalanced patients. The average KSS pain score for those patients where the Verasense technology was used was 96.2 (8.1 points higher than unbalanced) and the average KSS function score was 82.4 (9.5 points higher than unbalanced). These results were announced at the 2015 American Academy of Orthopaedic Surgeons Annual Meeting.
In addition, OrthoSensor has established strategic partnerships with several leading industry providers of knee implant systems. Smith & Nephew co-markets Verasense along with its Journey II and Legion Complete Knee Systems. Stryker positions the technology as able to be used with its Triathlon Knee System. Leveraging its co-marketing agreement with OrthoSensor, Zimmer Biomet offers the solution for its Persona, NexGen, and Vanguard Knee Systems.
“Our Journey II Total Knee System with Verilast Technology is already delivering improved knee motion, patient function, and implant wear reduction,” said Gaurav Agarwal, then president of orthopaedic reconstruction for Smith & Nephew (at the time of the co-marketing announcement). “By combining our innovations with Verasense’s exacting component placement and soft-tissue balancing, our surgeons can deliver even more benefits to patients.”
One-Stop Shopping Cart
On the surface, the GEO Cart may look like an ordinary “cabinet” in which to store orthopedic extremities products. As the cliché goes, however, looks can be deceiving. Through the use of RFID technology, the GEO Cart delivery system, and sterilized single-use implants and instruments, Gramercy Extremity Orthopedics has developed a smart solution that makes product tracking and inventory replenishment much more efficient.
The GEO Cart is mobile and can be positioned either in the OR itself or just outside in a central location so as to service multiple rooms. It holds approximately 1,800 sterile packages containing over 2,000 parts that offer full traceability. Prior to a procedure or during the surgery itself, a nurse or the doctor can go to the cart and remove the required components. The Tyvek label on the package is removed and placed atop the cart. At the end of the day or at the completion of a case, a button is pressed and the cart is scanned to determine inventory utilized. Based on the scan findings, an order form is automatically opened and sent to GEO for fulfillment. In addition, a copy of the order form is also provided to the hospital or surgical center’s materials manager or financial department for processing.
In May 2017, the company announced the first surgery had been performed with the use of the GEO Bone Screw and GEO Cart systems, performed by Dr. Peter A. Blume, D.P.M., F.A.C.F.A.S. at Shoreline Surgery Center LLC in Guilford, Conn. “I was extremely impressed and pleased with the outcome,” said Dr. Blume. “The ease in which the system operated was noted by the entire Surgical Center staff and felt that the systems created many efficiencies that are lacking in today’s operating environment. The surgical back table was free of clutter and the implant and instruments were truly best of class. The GEO Cart finally addresses all the pitfalls when operating with orthopedic hardware, whether it be at the Surgery Center or in the hospital setting.
Bioness & Mainstay Medical
Sam Brusco • Associate Editor
According to Centers for Disease Control and Prevention (CDC) studies, as many as one in four patients undergoing long-term therapy in primary care environments grapple with opioid-use disorder. Further, the number of U.S. opioid prescriptions and sales has increased fourfold since 1999, and the number of deaths from prescription opioid-related overdoses topped 165,000.
The “one size fits all” mentality of using opioids to manage chronic pain is chiefly to blame for these worrisome statistics. Patients—and even their clinicians—may not be aware of alternate routes to take to reduce pain to a manageable level. Anesthetic injections and/or nerve ablation treatments can temporarily quell the agony, but pain relief from ablation usually lasts a maximum of two years. For especially enduring chronic or orthopedic pain, wearable or implantable devices that deliver therapeutic neurostimulation to treat the affected nerve may offer welcome relief.
Revamping Rehab
In 2004, a group of visionaries united to establish Bioness Inc. to help those with neurological impairments regain their independence. Shortly thereafter, Alfred E. Mann—the entrepreneur and philanthropist behind Pacesetter Systems (now owned by Abbott Labs) and MiniMed (now owned by Medtronic)—gave his support to the fledgling company. As a result, Bioness was formed to develop and distribute neuromodulation devices into the field of rehabilitation. Bioness then partnered with NESS Ltd. to release two devices in the U.S.—the H200 Hand Rehabilitation System for patients with upper limb impairments, and the L300 Foot Drop System to assist those with foot drop (a dropping of the forefoot due to weakness, irritation, or damage to the common fibular nerve) in regaining a normal gait.
The greater majority of Bioness’ products—including L300 and H200—employ functional electrical stimulation (FES) to offer functional and therapeutic benefits for patients with central nervous disorders and orthopedic injuries. L300 consists of three main components: a functional stimulation cuff, control unit, and gait sensor that all communicate wirelessly. While walking, the gait sensor detects events and transmits signals to synchronize the electrical pulses, stimulating the foot muscles to lift off the ground at the appropriate time. H200 operates on the similar FES principle, but focuses on reaching, grasping, opening, and closing the hand. It can be used as part of a rehab regimen for stroke, brain injury, spinal cord injury, or any other disorders of the central nervous system resulting in upper limb functional deficits and impairments. Bioness also offers the Vector Gait Safety System, a rehabilitation module designed for physical and occupational therapy that provides patients recovering from stroke, amputations, and orthopedic, brain, and spinal cord injuries a safe and real-world environment to undergo physical therapy.
Bioness made its foray into implantable technologies with 2015’s FDA nod for StimRouter, a neuromodulation device to treat chronic peripheral pain. Rather than a large implanted component, StimRouter employs a 15 cm passive lead inserted percutaneously under local anesthesia. The tip of the lead is placed near the target peripheral nerve causing the chronic pain. The receiver is on the skin’s surface so the patient can program their level of neurostimulation.
Coming Back to Life
About two thirds of people suffer from low back pain at some point in their life. In most, the pain resolves after a few months, but for some, it becomes a chronic condition. It can begin as a simple strain or sprain of one of the joints of the spine. The brain responds to joint pain by limiting painful movements, one of which is suppressing the activation of the muscles stabilizing the lower back. As this happens, the spine ends up moving more easily into painful positions, resulting in an ongoing cycle of chronic low back pain (CLBP) and muscle weakening.
Many studies point to muscle control reactivation to break this vicious cycle. Physiotherapists can offer specific exercise programs to regain control of the spine stabilizing muscles, but sometimes conventional treatments aren’t enough to restore it. Aware of this, Dublin, Ireland-based medical device company Mainstay Medical developed the ReActiv8 implantable restorative neurostimulation system for people with disabling CLBP.
ReActiv8 is a small implanted device that stimulates the nerves responsible for activating the key muscles stabilizing the lower back. Once implanted, patients can deliver half-hour sessions twice daily via a handheld, wireless remote control. The hope is that repeated stimulation will eventually “teach” the brain and body to control the spine stabilizing muscles in between sessions, ultimately allowing the body to recover from CLBP.
The ReActiv8 implant is currently CE marked. As of July 9, all implantations for the U.S. IDE (Investigational Device Exemption) study, ReActiv8-B, have been completed. A total of 204 patients were implanted in the study, and Mainstay Medical expects to announce a full data readout from the trial toward the end of this year. The study intends to gather data to support a pre-market approval (PMA) application to the FDA in preparation for U.S. commercialization.
According to Mainstay’s annual report, the company generated $300,000 of revenue last year, entirely based on European sales. Initial activities were focused in Germany, with five centers in Germany and Ireland containing implanted patients as of fiscal 2017’s end. 2017 was also the first year Mainstay Medical posted any revenue at all. The company also has subsidiaries operating in the United States, Australia, Germany, and the Netherlands.
WishBone Medical Inc. & Pega Medical
Michael Barbella • Managing Editor
There are times Sarah Sibbel, M.D., feels more like a carpenter than a pediatric hand surgeon.
She has plates, screws, scissors, saws, hammers, and various power tools at her disposal—most of which are somewhat problematic for her patients. A (rare) specialist in brachial plexus birth palsy and congenital hand differences, Sibbel works exclusively with children, an easily overlooked and historically neglected component of the global healthcare spectrum.
“I treat kids’ hands...that’s their ultimate organ of exploration when they’re young,” Sibbel says in an online video for Children’s Hospital Colorado, where she is surgical director of the Brachial Plexus Clinic and director of the Pediatric Hand and Upper Extremity Program. “The saying is kids are not just little adults, and they’re not. They’re growing, they have growth plates, and they heal differently, oftentimes faster than adults do. I think it’s important to respect that aspect of taking care of children.”
Respect is hard to come by in pediatric orthopedics, though. Too small and generally too healthy to attract investor interest, children traditionally have been treated with jury-rigged adult devices (many of which are used off-label). Kid-sized implants are so rare, in fact, that surgeons have resorted to sawing grownup devices in half or mix-and-matching products with body parts (i.e., using spinal rods for femoral repair) in order to provide treatment.
Such makeshift solutions are becoming a bit less common nowadays as the medical device industry and the U.S. Food and Drug Administration address the longstanding shortfall in pediatric device development with grant programs and competitions. Those efforts could eventually help make the sector more appealing to financiers and boost the number of companies focused on juvenile orthopedics.
A Nod to Aunt Osti
Pediatric orthopedics apparently is part of the Deeter family heritage.
More than a century ago, Nick Deeter’s great great Aunt Osti (formal name: Ostia Lucille Smith) was one of the first female sales representatives in orthopedics for Revra DePuy, inventor of the fiber splint and founder of DePuy Manufacturing (now DePuy Synthes, part of Johnson & Johnson). Smith’s first sale was a juvenile ankle bone splint to a Florida doctor.
More than a century later, Deeter is keeping Smith’s legacy alive through his efforts to provide children with proper medical devices. He founded juvenile orthopedic implant/instrument manufacturer OrthoPediatrics in 2006, and WishBone Medical Inc. last year, becoming its CEO and board chairman.
“Ninety-five percent of the orthopedic implants going into children today are not specifically designed for them,” Deeter says on WishBone Medical’s website. “Ten years ago when I started OrthoPediatrics, I felt it was unacceptable for a child to get an adult device that was bent, cut, and altered just to try and make it fit while ignoring the growth plates and possibly causing more harm. There is still a lot of work to be done on behalf of our kids.”
Indeed there is, and WishBone Medical has wasted little time immersing itself in its mission. The Warsaw, Ind.-based firm sets itself apart from OrthoPediatrics by billing itself as a provider of “anatomically appropriate” implants and instruments in sterile packed, single-use disposable kits. The company has had a fairly active year thus far, releasing its first products in January and forging a partnership with the Hospital for Special Surgery (HSS).
WishBone Medical’s Broken Screw Removal System, available in three sizes (2.7mm, 3.5mm, 4.5mm), is designed to remove broken, stripped, and cold-welded screws. Its K-Wire Systems come in five different configurations—Pediatric Elbow Combo Kit, Pediatric Long Bone Combo Kit, and three Pediatric Extremity Combo Kits; each system includes two different sizes of wires, protective pin covers, and instruments.
The company also hammered out a U.S. distribution agreement with Implanet in March, and is collaborating with HSS to develop a pediatric hip implant. The product will specifically be designed to address the treatment of juvenile idiopathic arthritis and/or congenital dysplastic hip disease.
“We look forward to working with HSS on a Pediatric Total Hip system designed just for kids,” Deeter said in announcing the partnership.
A True VOICE for Children
Well before Deeter set out to make his Aunt Osti proud, Canadian firm Pega Medical was serving unmet needs in the pediatric orthopedic space. Founded in 1997, the Laval, Quebec-based company has developed seven different product sets for children, including pins, plates, screws, intramedullary nails, a self-extending rod, and a modular blade-plate system for femoral osteotomies and fractures.
With its products reaching more than 50 countries worldwide, Pega Medical considers itself a small international company with both a “brain” and “heart.” The latter is clearly evident in the firm’s devotion to underprivileged children worldwide suffering from bone diseases.
Through its CSR initiative, Pega Medical encourages surgeons to donate implants and instruments to their peers involved in medical missions to underdeveloped countries. Every surgeon who registers in the CSR program is given a VOICE (Virtual Orthopedic Implant Coin Exchange); surgeons who use Pega Medical devices obtain one VOICE for every implant purchased by their hospital. The VOICEs can be accumulated or donated to any cause in which a colleague is involved. The company donates one implant for every 50 mission VOICES accrued, and one instrument set for every 150 mission VOICES collected.
“We want to become a company that makes a real and durable difference for underprivileged children suffering from bone diseases around the world,” the company states on its website. “Pega Medical is recognized not only as an innovator with distinct devices reaching children with orthopedic conditions in over 50 countries around the world, but also as a company who puts the care of patients and the assistance to the surgeons as its first priority wherever they live (a small international company with a heart).”
OrthoSensor & Gramercy Extremity Orthopedics
Sean Fenske • Editor-in-Chief
The medtech industry continues to get “smarter”; that is, the devices themselves are being developed with artificial intelligence, sensors, communication-enabling technologies, and more to enhance the quality of care provided. Orthopedics, however, is one sector lagging behind in this trend. The implants going into patients today are very similar in terms of functionality to their counterparts of recent years. Indeed, fabrication techniques improve, materials may vary, and surfaces are modified with different treatments, but overall, the implants are no “smarter” than they were in the past.
In recent years, several companies have emerged seeking to change the “intelligence” behind orthopedic devices. The two companies featured in the following section have put products onto the market that offer smarter solutions to more traditional options. OrthoSensor has developed Verasense, which helps surgeons ensure the correct balancing of an implant during total knee arthroplasty. Gramercy Extremity Orthopedics provides a cart system to make extremities surgical procedures, as well as component ordering, much more efficient.
A Balancing Act
Founded in 2006, OrthoSensor’s vision is to utilize data to improve the decision-making process in orthopedic procedures. Toward this end, the company has developed the Verasense device. This technology is used to balance the knee implant during a primary or revision total knee replacement procedure. An implantable, disposable component couples wirelessly with an intra-operative monitor that offers surgeons feedback on the implant positioning in real time. It eliminates the “feel” aspect many surgeons rely on during a knee implant procedure. Upon use, the device is discarded and replaced with a permanent total knee replacement system component.
The results speak for themselves. According to the company, in a two-year multicenter study involving 135 patients, data reflected a 97 percent satisfaction rating from patients (i.e., an indication of “satisfied” or “very satisfied” at two-years post-op was provided). The study also revealed that knees balanced with Verasense achieved a KSS score of 178.6, which was 17.6 points higher than the average score of unbalanced patients. The average KSS pain score for those patients where the Verasense technology was used was 96.2 (8.1 points higher than unbalanced) and the average KSS function score was 82.4 (9.5 points higher than unbalanced). These results were announced at the 2015 American Academy of Orthopaedic Surgeons Annual Meeting.
In addition, OrthoSensor has established strategic partnerships with several leading industry providers of knee implant systems. Smith & Nephew co-markets Verasense along with its Journey II and Legion Complete Knee Systems. Stryker positions the technology as able to be used with its Triathlon Knee System. Leveraging its co-marketing agreement with OrthoSensor, Zimmer Biomet offers the solution for its Persona, NexGen, and Vanguard Knee Systems.
“Our Journey II Total Knee System with Verilast Technology is already delivering improved knee motion, patient function, and implant wear reduction,” said Gaurav Agarwal, then president of orthopaedic reconstruction for Smith & Nephew (at the time of the co-marketing announcement). “By combining our innovations with Verasense’s exacting component placement and soft-tissue balancing, our surgeons can deliver even more benefits to patients.”
One-Stop Shopping Cart
On the surface, the GEO Cart may look like an ordinary “cabinet” in which to store orthopedic extremities products. As the cliché goes, however, looks can be deceiving. Through the use of RFID technology, the GEO Cart delivery system, and sterilized single-use implants and instruments, Gramercy Extremity Orthopedics has developed a smart solution that makes product tracking and inventory replenishment much more efficient.
The GEO Cart is mobile and can be positioned either in the OR itself or just outside in a central location so as to service multiple rooms. It holds approximately 1,800 sterile packages containing over 2,000 parts that offer full traceability. Prior to a procedure or during the surgery itself, a nurse or the doctor can go to the cart and remove the required components. The Tyvek label on the package is removed and placed atop the cart. At the end of the day or at the completion of a case, a button is pressed and the cart is scanned to determine inventory utilized. Based on the scan findings, an order form is automatically opened and sent to GEO for fulfillment. In addition, a copy of the order form is also provided to the hospital or surgical center’s materials manager or financial department for processing.
In May 2017, the company announced the first surgery had been performed with the use of the GEO Bone Screw and GEO Cart systems, performed by Dr. Peter A. Blume, D.P.M., F.A.C.F.A.S. at Shoreline Surgery Center LLC in Guilford, Conn. “I was extremely impressed and pleased with the outcome,” said Dr. Blume. “The ease in which the system operated was noted by the entire Surgical Center staff and felt that the systems created many efficiencies that are lacking in today’s operating environment. The surgical back table was free of clutter and the implant and instruments were truly best of class. The GEO Cart finally addresses all the pitfalls when operating with orthopedic hardware, whether it be at the Surgery Center or in the hospital setting.