Maria Shepherd, President and Founder, Medi-Vantage11.30.18
The news has been devastating; according to the National Institute on Drug Abuse (NIDA) website, “every day, more than 115 people in the United States die after overdosing on opioids…including prescription pain relievers, heroin, and synthetic opioids such as fentanyl…The Centers for Disease Control and Prevention estimates that the total ‘economic burden’ of prescription opioid misuse alone in the United States is $78.5 billion a year, including the costs of healthcare, lost productivity, addiction treatment, and criminal justice involvement.”1
While it’s been reported that orthopedic surgeons are prescribing fewer opioids than in prior years, an article on AthenaInsight stated, “the average day supply per prescription remained flat, at approximately 51-55 days.”2 This is significant since many patients report their first encounter with opioids is after surgery or a medical intervention.
Further, from the NIDA website, “In the late 1990s, pharmaceutical companies reassured the medical community that patients would not become addicted to prescription opioid pain relievers, and healthcare providers began to prescribe them at greater rates. This subsequently led to widespread diversion and misuse of these medications before it became clear that these medications could indeed be highly addictive. Opioid overdose rates began to increase. In 2015, more than 33,000 Americans died as a result of an opioid overdose…That same year, an estimated 2 million people in the United States suffered from substance use disorders related to prescription opioid pain relievers, and 591,000 suffered from a heroin use disorder (not mutually exclusive).”1
Why This Is Important
The NIDA estimates that 21 to 29 percent of patients who are prescribed opioids for chronic pain misuse them.1 Between 8 to 12 percent will develop an opioid-use habit.1 An estimated 4 to 6 percent of these former patients will transition from prescription opioids to heroin (Table 1).1 The result? The CDC reports that of the 47,055 drug overdose deaths in 2014, 61 percent involved an opioid.3 Further, “from 2014 to 2015, the death rate from synthetic opioids other than methadone, which includes fentanyl, increased by 72.2 percent, and heroin death rates increased by 20.6 percent.”3
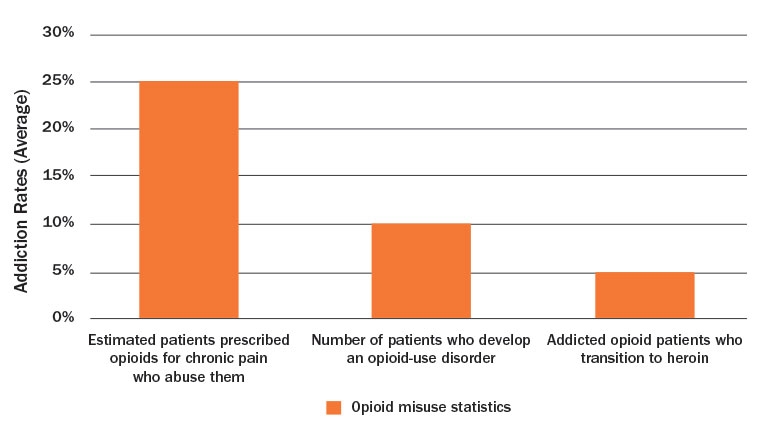
Table 1: NIDA Opioid Addiction Statistics1
Drug Misuse Trends Are Changing Healthcare
The CDC states that simply reducing medical use and the supply of addictive prescription medications will not tackle the overwhelming problem of untreated pain in the U.S. This means the solution to the opioid overdose crisis is deeply linked to pain management and the two problems must be addressed together. New medications tested in recent studies show promise for developing effective pain management without the risk of abuse or high numbers of fatal overdoses.4
These new classes of medications provide relief from pain without the rewarding effects and respiratory depression caused by current classes of approved pain meds. In addition, other non-opioid-based products like cannabinoids, sodium channel blockers, gene therapies, and medical devices that provide deep brain stimulation technologies also may lead to new therapies.
Neurostimulation—A Crowded Market
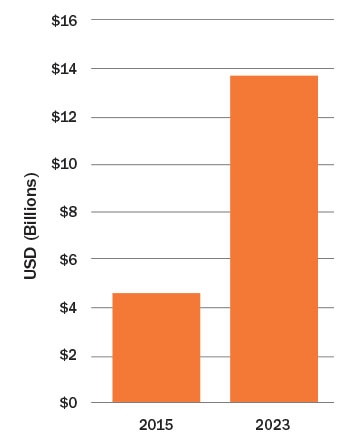
The top three device types in terms of market size and growth over the next five years are spinal cord, deep brain, and sacral nerve stimulation devices. Current indications for these devices include chronic pain, and many startup medtech companies are competing to gain share in these markets. It is not an easy market to enter because obstacles such as high device costs, low reimbursement, and poor awareness of referring physicians. The market for these three neurostim device types is projected to grow from $4.5 billion in 2015 to $13.7 billion in 2023 (Table 2).5
Innovation—Identifying New Spaces that Address Unmet Needs
While big and small players will compete aggressively for a foothold in the neurostim market, Medtronic has announced the launch of the Control Workflow system in the U.S. for use with its SynchroMed II intrathecal drug delivery system. The Medtronic pump and catheter are implanted under the skin to deliver medication into the intrathecal space of the spinal column. While many applications of neurostim are designed to treat the chronic pain patient after a problem with drug misuse has surfaced, Medtronic’s goal is to get to the potential opioid abuser before opioid misuse becomes an issue. Fortunately, early results look good. Combined with the use of the Medtronic pain pump, the Control Workflow system assists clinicians in weaning patients off oral opioids. Medtronic performed a retrospective analysis and learned that 51 percent of chronic non-malignant pain patients discontinued use of oral opioids after 12 months on the SynchroMed II pump.6
The team at Medtronic has put some thought into the design of the Control Workflow system. The device can be personalized for individual patients and provides guidance for oral opioid tapering. This assists physicians in creating treatment strategies for intrathecal medication with the lowest possible effective dose. The Control Workflow is especially helpful for patients because it places the medication at the source of the pain and may provide improved relief from pain with a lower number of drug complications while using a much smaller dose than oral medications.
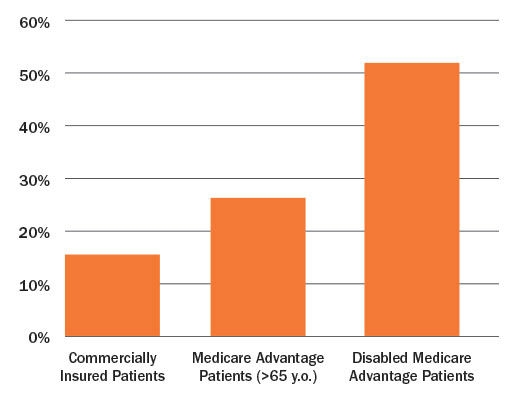
The Medtronic early intervention approach is needed. A recent report from researchers at the Mayo Clinic found that opioid prescription rates differ among insurers and those same rates, between 2007 and 2016, have not declined for commercially insured patients, despite efforts to reduce opioid prescribing.7 The Mayo Clinic study found that over 12 months, 14 percent of commercially insured patients, 26 percent of Medicare Advantage patients, and 52 percent of disabled Medicare Advantage patients received an opioid prescription (Table 3).7
The Medi-Vantage Perspective
The Medtronic solution gets ahead of the opioid misuse cycle before it starts, meeting the significant unmet need in chronic pain through a device when used for the appropriate patient segment. It has all the hallmarks we see in our product strategy research of an innovative device, by helping simplify drug delivery therapy, supporting oral opioid weaning, and providing patients with an efficacious pain relief alternative.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Inc., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.
While it’s been reported that orthopedic surgeons are prescribing fewer opioids than in prior years, an article on AthenaInsight stated, “the average day supply per prescription remained flat, at approximately 51-55 days.”2 This is significant since many patients report their first encounter with opioids is after surgery or a medical intervention.
Further, from the NIDA website, “In the late 1990s, pharmaceutical companies reassured the medical community that patients would not become addicted to prescription opioid pain relievers, and healthcare providers began to prescribe them at greater rates. This subsequently led to widespread diversion and misuse of these medications before it became clear that these medications could indeed be highly addictive. Opioid overdose rates began to increase. In 2015, more than 33,000 Americans died as a result of an opioid overdose…That same year, an estimated 2 million people in the United States suffered from substance use disorders related to prescription opioid pain relievers, and 591,000 suffered from a heroin use disorder (not mutually exclusive).”1
Why This Is Important
The NIDA estimates that 21 to 29 percent of patients who are prescribed opioids for chronic pain misuse them.1 Between 8 to 12 percent will develop an opioid-use habit.1 An estimated 4 to 6 percent of these former patients will transition from prescription opioids to heroin (Table 1).1 The result? The CDC reports that of the 47,055 drug overdose deaths in 2014, 61 percent involved an opioid.3 Further, “from 2014 to 2015, the death rate from synthetic opioids other than methadone, which includes fentanyl, increased by 72.2 percent, and heroin death rates increased by 20.6 percent.”3

Table 1: NIDA Opioid Addiction Statistics1
Drug Misuse Trends Are Changing Healthcare
The CDC states that simply reducing medical use and the supply of addictive prescription medications will not tackle the overwhelming problem of untreated pain in the U.S. This means the solution to the opioid overdose crisis is deeply linked to pain management and the two problems must be addressed together. New medications tested in recent studies show promise for developing effective pain management without the risk of abuse or high numbers of fatal overdoses.4

Table 2: Projected Global Market for Neurostimulation Implantables5
Neurostimulation—A Crowded Market
The top three device types in terms of market size and growth over the next five years are spinal cord, deep brain, and sacral nerve stimulation devices. Current indications for these devices include chronic pain, and many startup medtech companies are competing to gain share in these markets. It is not an easy market to enter because obstacles such as high device costs, low reimbursement, and poor awareness of referring physicians. The market for these three neurostim device types is projected to grow from $4.5 billion in 2015 to $13.7 billion in 2023 (Table 2).5
Innovation—Identifying New Spaces that Address Unmet Needs
While big and small players will compete aggressively for a foothold in the neurostim market, Medtronic has announced the launch of the Control Workflow system in the U.S. for use with its SynchroMed II intrathecal drug delivery system. The Medtronic pump and catheter are implanted under the skin to deliver medication into the intrathecal space of the spinal column. While many applications of neurostim are designed to treat the chronic pain patient after a problem with drug misuse has surfaced, Medtronic’s goal is to get to the potential opioid abuser before opioid misuse becomes an issue. Fortunately, early results look good. Combined with the use of the Medtronic pain pump, the Control Workflow system assists clinicians in weaning patients off oral opioids. Medtronic performed a retrospective analysis and learned that 51 percent of chronic non-malignant pain patients discontinued use of oral opioids after 12 months on the SynchroMed II pump.6
The team at Medtronic has put some thought into the design of the Control Workflow system. The device can be personalized for individual patients and provides guidance for oral opioid tapering. This assists physicians in creating treatment strategies for intrathecal medication with the lowest possible effective dose. The Control Workflow is especially helpful for patients because it places the medication at the source of the pain and may provide improved relief from pain with a lower number of drug complications while using a much smaller dose than oral medications.

Table 3: Opioid Prescription Rates Differ by Insurer7
The Medi-Vantage Perspective
The Medtronic solution gets ahead of the opioid misuse cycle before it starts, meeting the significant unmet need in chronic pain through a device when used for the appropriate patient segment. It has all the hallmarks we see in our product strategy research of an innovative device, by helping simplify drug delivery therapy, supporting oral opioid weaning, and providing patients with an efficacious pain relief alternative.
References
- http://bit.ly/odt181201
- http://bit.ly/odt181202
- http://bit.ly/odt181203
- http://bit.ly/odt181204
- http://bit.ly/odt181205
- http://bit.ly/odt181206
- http://bit.ly/odt181207
Maria Shepherd has more than 20 years of leadership experience in medical device/life science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Inc., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.