Mark Crawford, Contributing Writer02.13.19
Surgical instrumentation is increasingly driven by more complexity, multifunctionality, ease of use, and human factors engineering. There is a growing market for new and innovative instruments to improve the end-user experience—for both surgeons and patients. OEMs work closely with surgeons on instrument designs to improve ease of use and ergonomics. This all adds to total cost, which makes cost efficiencies of paramount importance to OEMs. These can include simpler designs, reduced components, different materials, and easier assembly. Even simple adjustments on the shop floor that make production more efficient can make a difference in cost control.
Complex geometry, tight tolerances, reduction in size, and surface treatments and coatings often push advanced manufacturing technologies and additive manufacturing processes, including rapid prototyping, to their technological limits. Medical device manufactures (MDMs) want rapid design iterations and overall development timeline compression. Equally important is ensuring that a robust design for manufacturability (DFM) process is in place to mitigate any potential design or production issues before products hit the manufacturing floor.
With these ever-increasing challenges, OEMs are turning to contract manufacturers (CMs) to help them realize timely results, cost efficiencies, and other benefits that complement their own internal capabilities. For example, single-use (disposable) devices are increasingly viewed by OEMs as a way to deliver high performance at a lower cost, while also reducing the risk of healthcare-associated infections (HAIs), which can be associated with reusable instrumentation.
“As procedures increasingly move toward ambulatory surgical centers, companies are becoming more focused on ways to service the needs of those platforms,” said Chad Ryshkus, director of marketing and product development for MedTorque, a Kenosha, Wis.-based manufacturer of orthopedic instruments and implants, including single-procedure torque-limiting drivers. “Single-procedure instrument kits are now a cost-effective and logistically efficient solution for providing instruments, especially in those settings.”
Another trend in surgical device design and manufacturing is adding more functionality to instruments. With a wide spectrum of patient types and surgical procedures to consider, surgeons want a greater variety of tools, with advanced features to improve their efficiency in the operating room and speed the recovery of their patients. Some procedures, for example, may require an instrument that has more bend, flex, and reach, acting as an extension of a surgeon’s hands and creating more freedom and range of motion for minimally invasive procedures.
“Adding this kind of functionality to instruments requires great attention to detail in the design phase and manufacturing process to help avoid design challenges and pitfalls that can add costs to development,” said Steve Santoro, executive vice president for MICRO, a Somerset, N.J.-based contract manufacturing organization.
Latest Trends
The big trend in orthopedic manufacturing is designing more functionality into smaller products, which often requires advanced features, more complex components, and tighter tolerances—all designed to add value to the end-user experience. For example, the demand for “weight reduction, radiolucency, and minimal radio frequency interference is growing with the advent of advanced surgical procedures used to improve patient outcomes, as well as reduce operating room time [cost],” said John MacDonald, president of AIP Precision Machining, a Daytona Beach, Fla.-based contract manufacturer specializing in machining high-tolerance components for the orthopedic industry. “The trend toward advanced polymer materials, coupled with the functional demands, result in extremely difficult tolerances to maintain in any material, let alone polymers.”
Because many orthopedic and spine instruments are reusable, few design changes have been made in recent years to improve fit and performance, even with product line extensions or case and tray redesigns. A large percentage of instruments are made of stainless steel with silicon or similar overmolds for grip and designed for several years’ surgical use. “Lifecycle support for these instruments has become increasingly expensive with spares, repair, logistics, reprocessing, and general handling costs taking a big bite out of the profit-and-loss statement,” said James B. Schultz, vice president of customer solutions for ECA Medical, a Thousand Oaks, Calif.-based manufacturer of single-use torque limiting and surgery-ready procedure kits. “Therefore, robust and economically viable single-use instrumentation, such as sterile pack surgery ready kits, is making headway in a wide range of implant surgeries and outpatient procedures.”
More healthcare providers are turning toward single-use disposable surgical instruments versus reusable devices for patient safety reasons—an effort to prevent cross-contamination and infection and reduce the rate of occurrence of HAIs. Some reusable devices have complex designs and intricate parts, which can be challenging to clean thoroughly of blood, tissue, or other biological debris. Also, with the rise in super bugs that are resistant to antimicrobial therapies, reusable devices can be potentially fatal to patients when they are not fully sanitized between uses.
“Disposable surgical instruments are gaining favor since they are sterile off the shelf and individually packaged, which inherently reduces risk of infection,” said Santoro. “This also helps hospitals and other healthcare facilities reduce their sterilization and disinfection costs.”
Multifunctionality is also a strong theme in instrument manufacturing. Surgeons (and therefore OEMs) want instruments that are designed to let them do more with a single instrument, with less positional stress and muscle fatigue.
“With the requirement that instruments have the flexibility to ‘multi-task’ for the surgeon, we are building more complex assemblies that can provide three or more actions during a procedure,” said David Cabral, president of Five Star Companies, a full-service contract manufacturer of instruments for the orthopedic, spine, and sports medicine markets.
For example, the typical single-action grasper, inserter, and driver are now incorporated into a complex, multi-component device that will execute these activities nearly simultaneously. The challenges with making these types of instruments come into play at the assembly stage of the process. “With the engineered tolerances and variations, fitment of parts, and adherence to functionality, the skills required across the manufacturing spectrum is critical,” Cabral added. “This is where having a strong DFM team is imperative.”
What OEMs Want
As much as they want faster speed to market and lower costs, OEMs also seek deep subject knowledge and specific manufacturing proficiencies from their CMs—to complement or even substitute for their own internal core capabilities. OEMs expect their CMs to be knowledgeable enough to solve their pressing design and manufacturing problems. They also want more efficient turnaround due to streamlined project and supply chain management that their CMs can deliver, which are often smaller and nimbler than the OEMs themselves. CMs must also be skilled with DFM to help guide their customers from initial concept to final result, with minimal cost and greater time efficiencies.
“Being on top of new advancements and next-generation technologies is a value that contract manufacturers bring to their OEM relationships,” said Santoro. “Value is not always about lowest price, but about making the most cost-effective and efficient device that will be profitable and long lasting.”
That said, OEMs still push hard for reduced costs and faster speed to market. Aggressive pricing and reasonable lead times of 10 weeks or less are the standard requests and expectation.
“Our team adds Lean manufacturing strategies to our own unique brand of design for manufacturability to provide the added value our customers are looking for,” said John Phillips, president of operations for the implant and instrumentation division of Phillips Precision Medicraft, an Elmwood Park, N.J.-based manufacturer of advanced orthopedic implants, instrumentation, and sterilization delivery systems.
OEMs are always on the hunt for turnkey design and manufacturing solutions that provide measurable value. For example, single-use, sterile pack, and surgery-ready procedural kits that are optimized to produce the best outcomes at best value are in high demand. This is especially true for outpatient settings where operating room turnover is critical and inventory management and cost controls are paramount.
Another trend is toward single-use torque limiters for securing orthopedic and spine implants. “Reusable torque limiters are expensive to purchase and maintain and can easily fall out of calibration and become challenges to support over the lifecycle,” said Schultz. “Single-use factory set torque limiters are always in spec and provide perfect fixation throughout the procedure, reducing revision and infection risks and cutting overall costs.”
Innovative Thinking Required
The OEM call for speed-to-market and reduced pricing is the standard state in the industry, even as design and manufacturing become more complex, with smaller and more complex products made from advanced materials. Upgrades to technology can only do so much—boosting operational efficiencies are increasingly important to shave precious time and cost from manufacturing and delivery to market.
These pressing needs challenged Phillips Precision Medicraft to “create highly customized manufacturing solutions for our customers, as their requirements change,” said Phillips. “Since instrument designs continue to become more complex, it’s even more important to approach manufacturing solutions with innovative thinking and technology.”
Handheld surgical instruments have small components and require great attention to detail regarding function and design, including the geometry of the part and the material used. Getting these factors right often depends on translating input from surgeons and other key end users.
“As with most hand-held instruments, surgeons expect a certain ‘feel,’ whether it is a smoothness of operation, an assuring click that ensures engagement, or a visual indicator that the expected procedural outcome has been achieved,” said Cabral.
“Some minimally invasive tools for laparoscopic, endoscopic, and arthroscopic procedures require more flexibility and reach,” added Santoro. “Therefore, precision in design and manufacturing is essential for addressing the angles, degrees, and rotations required for specific instruments. Articulating instruments may also require more design planning because of the precision required during manufacturing.”
An increased number of components within a device tends to increase the level of manufacturing complexity. Inherent in these complex devices is a component tolerance stack that must be minimized at assembly—”this is where close customer interaction and DFM skills play a significant role in understanding the device’s use and the customer’s expectation and intent for performance and functionality,” said Cabral.
In addition to having strong assembly experience and command of controlling tolerances at the component level, CMs must also intimately understand how the instrument will be used in surgery and how all the components fit together to ensure the instrument functions as intended. It must also be designed robust enough to sustain repeated use without safety or efficacy challenges, while not being overly expensive to manufacture.
“Designing and manufacturing instrumentation requires good knowledge of the procedure and implant requirements so you can achieve needed results with optimized instrument sets and reduce overall upfront and lifecycle costs,” said Schultz. “Understanding loads, boundary conditions, stresses, and other factors are critical, as well as operational stack up and ergonomics.”
OEM designers (sometimes prodded by their CM partners) must keep DFM in mind; if ignored, the end result might be product that is over- or under-designed, leading to higher costs and time inefficiencies. A product that is over-designed may be functional, but too complicated to manufacture in high volumes. A product that is under-designed could fail to achieve its performance specifications. Therefore, it is advantageous for OEMs to bring in a qualified CM early in the process to solve potential problems that can be identified and addressed during the design phase—saving time, money, and other resources in the long run.
“Unfortunately,” noted Santoro, “product design engineers often have cost, time, or other pressures and dismiss manufacturing considerations, which can prove detrimental to ultimately producing the parts successfully.”
Technology Improvements
Precise electrochemical machining (PECM) is a technique that offers several advantages over other technologies in terms of material selection, surface finish, material removal rate (MRR), tolerance capabilities, grain structure, and other benefits.
“With PECM, greater precision and accuracy can be achieved resulting in highly complex geometrical shapes with high tolerance capabilities,” said Santoro.
PECM is a non-contact metal-shaping process that pairs a PECM machine with an electrolyte processing unit, power generator, and operator control station. Using electric current and an oscillating tool bathed in a conductive electrolyte (salt water), PECM dissolves metal by liquefication. A positively charged workpiece (the anode) takes the form of the negatively charged tool (the cathode) as the metal is dissolved. As a result, high-quality, burr-free parts can be produced without cracks or material deformities.
“MRR is not affected by the hardness of the metal and highly complex, contoured, and intricate shapes can be produced,” added Santoro. “Since the electrode or tool never touches the anode or workpiece, there is no wear on the final part and roughing, finishing, and polishing can all be accomplished in one operation.”
To improve operational efficiencies and speed, forward-thinking companies are taking on a culture of continuous improvement. Efficiencies that are gained—even small ones—can make an OEM or CM stand out from the crowd in terms of turnaround or cost. And although the Internet of Things will eventually lead the way when it comes to efficient manufacturing, other tools such as Lean are still highly effective in eliminating waste in the manufacturing process. For example, Phillips Precision Medicraft has created dedicated Lean manufacturing cells for select products, increased capacity by implementing new technologies, and managed manufacturing process controls effectively. “With a strong 5S Lean culture, we’ve also been able to increase overall speed and manufacturing output,” said Phillips. “We have also invested in new palletized machining centers to further reduce set-ups, run components on demand, and in some cases, run lights out.”
Looking to the Future
Design for manufacturability will continue to grow as a critical part of the design phase as OEMs make more complex products—both to maximize performance/safety and control lifecycle costs. Developing an efficient design with DFM is absolutely essential for keeping costs manageable and the return on investment high.
Additive manufacturing (AM) will also have a growing role in instrument manufacturing. The varied capabilities of AM technologies are changing the way OEMs look at designing instruments and systems. AM gives engineers an expanded design window to create life-saving products that cannot be manufactured in any other way. Although traditional methods are still effective, they are increasingly being integrated with AM systems to speed up the overall product development process—for example, making fixtures or prototypes.
The creation of 3D-printed surgical instruments might be closer than we imagine. The National Institutes of Health recently undertook a study that investigated the feasibility, design, and fabrication processes for making usable 3D-printed surgical instruments. Using a computer-aided design (CAD) package, the research team designed a surgical set consisting of hemostats, needle driver, scalpel handle, retractors, and forceps. These designs were then printed out using selective laser sintering (SLS). The products were evaluated by practicing general surgeons for ergonomic functionality and performance. Improvements were identified and adjustments made. Iterative cycles including design, production, and testing took an average of three days. After a series of five iterative cycles, the instruments were deemed suitable for performing surgical procedures.
“Functional 3D-printed surgical instruments are feasible,” wrote researchers in the World Journal of Surgery.1 “Advantages compared to traditional manufacturing methods include no increase in cost for increased complexity, accelerated design to production times, and surgeon-specific modifications.”
Widespread adoption of robotics will be in the future as well. Many large OEMs have invested heavily in robotic surgical platforms, which have produced a need for highly precise instruments with extremely tight tolerances, compared to traditional instrumentation.
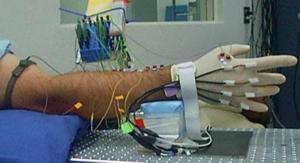
Advances in robotic technologies and software continue. For example, in 2017, researchers developed a new type of robotic arm that helps surgeons perform minimally invasive procedures. According to a press release, the researchers “looked at how different components in a mechanical system move in relation to one another, and how multiple chains of motion influence the performance of a system overall.
They then created an abstract representation, known as a constraint map, of a person’s arm movement—from the shoulder to the elbow to the forearm to the wrist to the fingers—with each joint represented.”
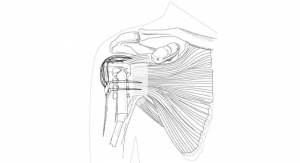
After mapping the motions of each joint and extending them beyond the hand to the motions of corresponding joints in the instrument, they eventually developed a streamlined mechanical instrument with precision pinchers. Commercialized by FlexDex Surgical, this robotic device precisely translates the surgeon’s hand, wrist, and arm movements from outside the patient into corresponding movements of an end-effector inside the patient’s body.
According to its inventors, this product provides the same sorts of benefits as robot-assisted surgery, such as greater precision and functionality, but at a lower cost compared to existing robotic surgical systems. The lower cost makes it possible for rural hospitals and other medical centers that can’t afford more expensive robot-assisted systems to perform these procedures.
“It will be interesting to see the impact that robotics will have on the development of instrumentation in the coming years,” said Phillips. “Improvements to and increased use of robotics in the operating room today will likely reduce the need for instrumentation in the years to come. This is the precise reason we are investing so heavily in the type of manufacturing technology that supports high-volume implant manufacturing.”
References
1 http://bit.ly/odt190160
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.
Complex geometry, tight tolerances, reduction in size, and surface treatments and coatings often push advanced manufacturing technologies and additive manufacturing processes, including rapid prototyping, to their technological limits. Medical device manufactures (MDMs) want rapid design iterations and overall development timeline compression. Equally important is ensuring that a robust design for manufacturability (DFM) process is in place to mitigate any potential design or production issues before products hit the manufacturing floor.
With these ever-increasing challenges, OEMs are turning to contract manufacturers (CMs) to help them realize timely results, cost efficiencies, and other benefits that complement their own internal capabilities. For example, single-use (disposable) devices are increasingly viewed by OEMs as a way to deliver high performance at a lower cost, while also reducing the risk of healthcare-associated infections (HAIs), which can be associated with reusable instrumentation.
“As procedures increasingly move toward ambulatory surgical centers, companies are becoming more focused on ways to service the needs of those platforms,” said Chad Ryshkus, director of marketing and product development for MedTorque, a Kenosha, Wis.-based manufacturer of orthopedic instruments and implants, including single-procedure torque-limiting drivers. “Single-procedure instrument kits are now a cost-effective and logistically efficient solution for providing instruments, especially in those settings.”
Another trend in surgical device design and manufacturing is adding more functionality to instruments. With a wide spectrum of patient types and surgical procedures to consider, surgeons want a greater variety of tools, with advanced features to improve their efficiency in the operating room and speed the recovery of their patients. Some procedures, for example, may require an instrument that has more bend, flex, and reach, acting as an extension of a surgeon’s hands and creating more freedom and range of motion for minimally invasive procedures.
“Adding this kind of functionality to instruments requires great attention to detail in the design phase and manufacturing process to help avoid design challenges and pitfalls that can add costs to development,” said Steve Santoro, executive vice president for MICRO, a Somerset, N.J.-based contract manufacturing organization.
Latest Trends
The big trend in orthopedic manufacturing is designing more functionality into smaller products, which often requires advanced features, more complex components, and tighter tolerances—all designed to add value to the end-user experience. For example, the demand for “weight reduction, radiolucency, and minimal radio frequency interference is growing with the advent of advanced surgical procedures used to improve patient outcomes, as well as reduce operating room time [cost],” said John MacDonald, president of AIP Precision Machining, a Daytona Beach, Fla.-based contract manufacturer specializing in machining high-tolerance components for the orthopedic industry. “The trend toward advanced polymer materials, coupled with the functional demands, result in extremely difficult tolerances to maintain in any material, let alone polymers.”
Because many orthopedic and spine instruments are reusable, few design changes have been made in recent years to improve fit and performance, even with product line extensions or case and tray redesigns. A large percentage of instruments are made of stainless steel with silicon or similar overmolds for grip and designed for several years’ surgical use. “Lifecycle support for these instruments has become increasingly expensive with spares, repair, logistics, reprocessing, and general handling costs taking a big bite out of the profit-and-loss statement,” said James B. Schultz, vice president of customer solutions for ECA Medical, a Thousand Oaks, Calif.-based manufacturer of single-use torque limiting and surgery-ready procedure kits. “Therefore, robust and economically viable single-use instrumentation, such as sterile pack surgery ready kits, is making headway in a wide range of implant surgeries and outpatient procedures.”
More healthcare providers are turning toward single-use disposable surgical instruments versus reusable devices for patient safety reasons—an effort to prevent cross-contamination and infection and reduce the rate of occurrence of HAIs. Some reusable devices have complex designs and intricate parts, which can be challenging to clean thoroughly of blood, tissue, or other biological debris. Also, with the rise in super bugs that are resistant to antimicrobial therapies, reusable devices can be potentially fatal to patients when they are not fully sanitized between uses.
“Disposable surgical instruments are gaining favor since they are sterile off the shelf and individually packaged, which inherently reduces risk of infection,” said Santoro. “This also helps hospitals and other healthcare facilities reduce their sterilization and disinfection costs.”
Multifunctionality is also a strong theme in instrument manufacturing. Surgeons (and therefore OEMs) want instruments that are designed to let them do more with a single instrument, with less positional stress and muscle fatigue.
“With the requirement that instruments have the flexibility to ‘multi-task’ for the surgeon, we are building more complex assemblies that can provide three or more actions during a procedure,” said David Cabral, president of Five Star Companies, a full-service contract manufacturer of instruments for the orthopedic, spine, and sports medicine markets.
For example, the typical single-action grasper, inserter, and driver are now incorporated into a complex, multi-component device that will execute these activities nearly simultaneously. The challenges with making these types of instruments come into play at the assembly stage of the process. “With the engineered tolerances and variations, fitment of parts, and adherence to functionality, the skills required across the manufacturing spectrum is critical,” Cabral added. “This is where having a strong DFM team is imperative.”
What OEMs Want
As much as they want faster speed to market and lower costs, OEMs also seek deep subject knowledge and specific manufacturing proficiencies from their CMs—to complement or even substitute for their own internal core capabilities. OEMs expect their CMs to be knowledgeable enough to solve their pressing design and manufacturing problems. They also want more efficient turnaround due to streamlined project and supply chain management that their CMs can deliver, which are often smaller and nimbler than the OEMs themselves. CMs must also be skilled with DFM to help guide their customers from initial concept to final result, with minimal cost and greater time efficiencies.
“Being on top of new advancements and next-generation technologies is a value that contract manufacturers bring to their OEM relationships,” said Santoro. “Value is not always about lowest price, but about making the most cost-effective and efficient device that will be profitable and long lasting.”
That said, OEMs still push hard for reduced costs and faster speed to market. Aggressive pricing and reasonable lead times of 10 weeks or less are the standard requests and expectation.
“Our team adds Lean manufacturing strategies to our own unique brand of design for manufacturability to provide the added value our customers are looking for,” said John Phillips, president of operations for the implant and instrumentation division of Phillips Precision Medicraft, an Elmwood Park, N.J.-based manufacturer of advanced orthopedic implants, instrumentation, and sterilization delivery systems.
OEMs are always on the hunt for turnkey design and manufacturing solutions that provide measurable value. For example, single-use, sterile pack, and surgery-ready procedural kits that are optimized to produce the best outcomes at best value are in high demand. This is especially true for outpatient settings where operating room turnover is critical and inventory management and cost controls are paramount.
Another trend is toward single-use torque limiters for securing orthopedic and spine implants. “Reusable torque limiters are expensive to purchase and maintain and can easily fall out of calibration and become challenges to support over the lifecycle,” said Schultz. “Single-use factory set torque limiters are always in spec and provide perfect fixation throughout the procedure, reducing revision and infection risks and cutting overall costs.”
Innovative Thinking Required
The OEM call for speed-to-market and reduced pricing is the standard state in the industry, even as design and manufacturing become more complex, with smaller and more complex products made from advanced materials. Upgrades to technology can only do so much—boosting operational efficiencies are increasingly important to shave precious time and cost from manufacturing and delivery to market.
These pressing needs challenged Phillips Precision Medicraft to “create highly customized manufacturing solutions for our customers, as their requirements change,” said Phillips. “Since instrument designs continue to become more complex, it’s even more important to approach manufacturing solutions with innovative thinking and technology.”
Handheld surgical instruments have small components and require great attention to detail regarding function and design, including the geometry of the part and the material used. Getting these factors right often depends on translating input from surgeons and other key end users.
“As with most hand-held instruments, surgeons expect a certain ‘feel,’ whether it is a smoothness of operation, an assuring click that ensures engagement, or a visual indicator that the expected procedural outcome has been achieved,” said Cabral.
“Some minimally invasive tools for laparoscopic, endoscopic, and arthroscopic procedures require more flexibility and reach,” added Santoro. “Therefore, precision in design and manufacturing is essential for addressing the angles, degrees, and rotations required for specific instruments. Articulating instruments may also require more design planning because of the precision required during manufacturing.”
An increased number of components within a device tends to increase the level of manufacturing complexity. Inherent in these complex devices is a component tolerance stack that must be minimized at assembly—”this is where close customer interaction and DFM skills play a significant role in understanding the device’s use and the customer’s expectation and intent for performance and functionality,” said Cabral.
In addition to having strong assembly experience and command of controlling tolerances at the component level, CMs must also intimately understand how the instrument will be used in surgery and how all the components fit together to ensure the instrument functions as intended. It must also be designed robust enough to sustain repeated use without safety or efficacy challenges, while not being overly expensive to manufacture.
“Designing and manufacturing instrumentation requires good knowledge of the procedure and implant requirements so you can achieve needed results with optimized instrument sets and reduce overall upfront and lifecycle costs,” said Schultz. “Understanding loads, boundary conditions, stresses, and other factors are critical, as well as operational stack up and ergonomics.”
OEM designers (sometimes prodded by their CM partners) must keep DFM in mind; if ignored, the end result might be product that is over- or under-designed, leading to higher costs and time inefficiencies. A product that is over-designed may be functional, but too complicated to manufacture in high volumes. A product that is under-designed could fail to achieve its performance specifications. Therefore, it is advantageous for OEMs to bring in a qualified CM early in the process to solve potential problems that can be identified and addressed during the design phase—saving time, money, and other resources in the long run.
“Unfortunately,” noted Santoro, “product design engineers often have cost, time, or other pressures and dismiss manufacturing considerations, which can prove detrimental to ultimately producing the parts successfully.”
Technology Improvements
Precise electrochemical machining (PECM) is a technique that offers several advantages over other technologies in terms of material selection, surface finish, material removal rate (MRR), tolerance capabilities, grain structure, and other benefits.
“With PECM, greater precision and accuracy can be achieved resulting in highly complex geometrical shapes with high tolerance capabilities,” said Santoro.
PECM is a non-contact metal-shaping process that pairs a PECM machine with an electrolyte processing unit, power generator, and operator control station. Using electric current and an oscillating tool bathed in a conductive electrolyte (salt water), PECM dissolves metal by liquefication. A positively charged workpiece (the anode) takes the form of the negatively charged tool (the cathode) as the metal is dissolved. As a result, high-quality, burr-free parts can be produced without cracks or material deformities.
“MRR is not affected by the hardness of the metal and highly complex, contoured, and intricate shapes can be produced,” added Santoro. “Since the electrode or tool never touches the anode or workpiece, there is no wear on the final part and roughing, finishing, and polishing can all be accomplished in one operation.”
To improve operational efficiencies and speed, forward-thinking companies are taking on a culture of continuous improvement. Efficiencies that are gained—even small ones—can make an OEM or CM stand out from the crowd in terms of turnaround or cost. And although the Internet of Things will eventually lead the way when it comes to efficient manufacturing, other tools such as Lean are still highly effective in eliminating waste in the manufacturing process. For example, Phillips Precision Medicraft has created dedicated Lean manufacturing cells for select products, increased capacity by implementing new technologies, and managed manufacturing process controls effectively. “With a strong 5S Lean culture, we’ve also been able to increase overall speed and manufacturing output,” said Phillips. “We have also invested in new palletized machining centers to further reduce set-ups, run components on demand, and in some cases, run lights out.”
Looking to the Future
Design for manufacturability will continue to grow as a critical part of the design phase as OEMs make more complex products—both to maximize performance/safety and control lifecycle costs. Developing an efficient design with DFM is absolutely essential for keeping costs manageable and the return on investment high.
Additive manufacturing (AM) will also have a growing role in instrument manufacturing. The varied capabilities of AM technologies are changing the way OEMs look at designing instruments and systems. AM gives engineers an expanded design window to create life-saving products that cannot be manufactured in any other way. Although traditional methods are still effective, they are increasingly being integrated with AM systems to speed up the overall product development process—for example, making fixtures or prototypes.
The creation of 3D-printed surgical instruments might be closer than we imagine. The National Institutes of Health recently undertook a study that investigated the feasibility, design, and fabrication processes for making usable 3D-printed surgical instruments. Using a computer-aided design (CAD) package, the research team designed a surgical set consisting of hemostats, needle driver, scalpel handle, retractors, and forceps. These designs were then printed out using selective laser sintering (SLS). The products were evaluated by practicing general surgeons for ergonomic functionality and performance. Improvements were identified and adjustments made. Iterative cycles including design, production, and testing took an average of three days. After a series of five iterative cycles, the instruments were deemed suitable for performing surgical procedures.
“Functional 3D-printed surgical instruments are feasible,” wrote researchers in the World Journal of Surgery.1 “Advantages compared to traditional manufacturing methods include no increase in cost for increased complexity, accelerated design to production times, and surgeon-specific modifications.”
Widespread adoption of robotics will be in the future as well. Many large OEMs have invested heavily in robotic surgical platforms, which have produced a need for highly precise instruments with extremely tight tolerances, compared to traditional instrumentation.
Advances in robotic technologies and software continue. For example, in 2017, researchers developed a new type of robotic arm that helps surgeons perform minimally invasive procedures. According to a press release, the researchers “looked at how different components in a mechanical system move in relation to one another, and how multiple chains of motion influence the performance of a system overall.
They then created an abstract representation, known as a constraint map, of a person’s arm movement—from the shoulder to the elbow to the forearm to the wrist to the fingers—with each joint represented.”
After mapping the motions of each joint and extending them beyond the hand to the motions of corresponding joints in the instrument, they eventually developed a streamlined mechanical instrument with precision pinchers. Commercialized by FlexDex Surgical, this robotic device precisely translates the surgeon’s hand, wrist, and arm movements from outside the patient into corresponding movements of an end-effector inside the patient’s body.
According to its inventors, this product provides the same sorts of benefits as robot-assisted surgery, such as greater precision and functionality, but at a lower cost compared to existing robotic surgical systems. The lower cost makes it possible for rural hospitals and other medical centers that can’t afford more expensive robot-assisted systems to perform these procedures.
“It will be interesting to see the impact that robotics will have on the development of instrumentation in the coming years,” said Phillips. “Improvements to and increased use of robotics in the operating room today will likely reduce the need for instrumentation in the years to come. This is the precise reason we are investing so heavily in the type of manufacturing technology that supports high-volume implant manufacturing.”
References
1 http://bit.ly/odt190160
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.