Mark Crawford, Contributing Writer03.24.22
As the orthopedic device market rebounds from COVID-19, product design and prototyping is on the rise. A growing need exists for prototype devices to prove new designs prior to full-scale launch, especially for more complex devices.
“Product development seems to be taking on more of the projects that have been on the back burner,” said David Neal, president of STAT DESIGN, a Lake Hopatcong, N.J.-based business that provides CAD design, development, and 3D printing services to the medical device industry. “With the daily fires to put out from COVID-19 slowing down a bit, there is more time to work on some of the other projects that may have gotten pushed down the priority list.”
Some of these projects involve a merging of technologies, including minimally invasive interventional approaches, advanced imaging and navigational systems, and greater adoption of robotics. “Product design has become more focused on procedural solutions rather than discrete devices,” said David Schechter, president of Meddux Development Corporation, a Boulder, Colo.-based medical device design, development, and manufacturing partner for complex single-use devices. “This requires a committed emphasis on human factors/usability engineering and incorporating a systems approach to product development.”
Quick turnaround is essential for prototyping these complex devices and a manufacturable design is critical for achieving that expectation. “We have experienced increased need for prototypes due to the expected upticks in surgeries and product development after the last two years of the pandemic,” said David Cabral, president and CEO of Five Star Companies, a New Bedford, Ma.-based contract manufacturer of precision instruments in the ortho, spine, and sports medicine markets. “Surgeries have opened up and product development is in high gear.”
There are several ways to outsource prototyping, including stand-alone prototype shops that specialize in quick turns of initial designs and 3D printing service bureaus that convert solid models into functional parts in metal or plastic in a matter of hours. A third option is “the contract manufacturer that can machine complex rapid prototypes and then smoothly transition into higher volume production,” said Philip Allen, vice president of sales and marketing for Lowell, a Minneapolis, Minn.-based contract manufacturer of complex implants and instruments for the orthopedic and cardiovascular markets. “Each of these three options fills an important role in the production of prototypes. As a contract manufacturer, our focus is on complex prototypes that lead to the production of complex implants and instruments.”
Latest Trends
Implant manufacturers are intent on finding new surface treatments to make their implants more effective and long-lasting. Porous surfaces (also known as porous coatings) have been used on orthopedic implants for more than 30 years to facilitate bony in-growth and long-term fixation. “With joint replacement now more common in younger and active patients, there is more demand for improved implant performance and safety, and porous coatings play an important role in successful implantation and longevity,” said Shilesh Jani, engineering manager for Orchid Orthopedic Solutions, a Mason, Mich.-based developer and contract manufacturer of medical devices and implants.
For implant surfaces, orthopedic OEMs are becoming more particular about certain factors, such as coating thickness, and want to increase pore size, porosity, and mechanical strength—all key factors that impact osseointegration and the safety of the implant. “OEMs also want coatings to be rougher, with a higher friction coefficient than for previous generations of coatings,” said Jani.
Allen sees a trend toward blending 3D-printed prototypes with machined complex features.
“There are tremendous advantages in using this approach,” said Allen. “Many 3D-printed prototypes have a tolerance range between ±0.003 inches to ±0.01 inches, depending on factors like part size, material selection, and technology being utilized. However, if features that are critical to the design must be held tighter, those features can be machined into the part. This approach combines the best of both worlds—the speed of additive manufacturing with the ability to add complex, close-tolerance features using a subtractive or machined approach.”
For example, a new cervical plate design requires a screw hole that must have a very specific feel for a surgeon when the screw is introduced to the plate during laboratory testing. If the prototype does not perform like the production part, the entire project can be at risk. A typical complex machined prototype of a cervical plate could take up to six weeks; plates made through a 3D-printed process take a week or so. A subtractive machining process can then be used to add the complex screw hole to the plate in another week—cutting the development time in half, yet retaining the features the surgeon had identified.
As devices become increasingly complex, with more parts and added functionality, there is greater need for design for manufacturing (DFM) to identify the best and most cost-effective manufacturing process. Using DFM during the prototyping stage will identify and work out any design flaws before the production process gets locked in. Finite element analysis (FEA) is a crucial DFM tool that predicts how a proposed medical device will respond to real-world forces such as vibration, load, heat, fluid flow, electrostatics, and other physical effects. FEA will reveal a product’s most probable points of failure, which can then be fixed through redesign if needed.
“We use FEA with a tighter range of material properties to ensure strength and durability without major functional or feature changes to the components,” said Dennis Sullivan, senior manager of advanced sourcing for Medtronic Surgical Robotics, a Minneapolis, Minn.-based developer and manufacturer of robotic surgical solutions. “Recently, we went from a machined aluminum housing to a casting. Using FEA, we identified a specific material from one of our suppliers that allowed us to maintain the original design. While we still need to validate the process, the design remains intact with minimal impact.”
DFI—design for inspection—is another process that evaluates how a component will be inspected and how the design can be modified to make the inspection process as clear as possible. Another helpful program is DFSS or design for Six Sigma, which improves the quality of the design/production process by incorporating lean processes and eliminating waste (fewer steps). All these methods, when undertaken during the design phase, optimize efficiency and quality and reduce the probability that redesign and reiteration will be required.
What OEMs Want
Speed is everything, especially during early concept development, when there are plenty of good ideas to test. It is essential for medical device manufacturers (MDMs) to have supply chain partners that can iterate and prototype quickly, so critical feedback from surgeons and clinicians can be included during the design cycle, saving time and money.
Accelerated design, production, and time to market are still the top requests of orthopedic companies.
“I want a supplier that will partner with me to do everything we can, up front, to ensure speed on the back end of a project,” said Sullivan. “This includes not only things like DFM and process development, but also putting fair contracts in place for mutual protection.”
First iterations typically require very fast lead times—in this early stage OEMs want to put a solid, 3D representation of the concept into the hands of their decision-makers, rather than simply show them an image on the screen. As the team gets closer to locking in a design, “it becomes more important to have a part that is as close to representing the final product as possible, to enable an easy transition to production,” said Allen. “In this stage, the ability to match the print and function for final testing becomes critical.”
Faster speed also has a role in the other top items on the OEM wish list. This growing variety of tasks reflects how OEMs increasingly expect their contract manufacturers (CMs) to take on more product development responsibilities in-house—even regulatory compliance expertise. Capabilities on the list include modifications to existing parts, compliance certifications, and cost controls (for example, competitive and/or effortless procurement of raw materials) that do not compromise device function.
“Quick turnaround speed is always essential,” said Robert Suric, global sales manager for gSource, an Emerson, N.J.-based provider of handheld, non-powered surgical instruments. “Faster product development can also be accomplished through a shorter supply chain, vertical integration, streamlined logistics, and supply chain management solutions that result in getting raw materials on time, or even having them in stock.”
With the COVID-19 pandemic still present, a vertically integrated supplier is both safer and faster (shorter supply chain).
“Large or small company, it doesn’t matter,” added Sullivan. “A supplier that can perform the component fabrication, including finishing and assembly, is in high demand. With the lack of resources these days, this is a more effective approach than trying to schedule multiple subcontractors, which inevitably creates delays and adversely affects project timelines.”
Although they are still highly cost-conscious, OEMs are interested in more multi-functional and complex designs, products, and assemblies and expect their CMs to have the answers. “A good example is an instrument that can insert, expand, rotate, and remove without the need to use multiple devices,” said Cabral. “This saves surgeons time, reduces fatigue, and typically results in better patient outcomes.”
Another cost-effective solution is reducing the weight of surgical instruments—although sales and marketing teams tout the benefit of combining instruments into a single kit, engineers struggle to keep the weight under 25 pounds (as per recommendations from the Association of periOperative Registered Nurses and the Association for the Advancement of Medical Instrumentation). Using modular handles and hollowing out instruments are two ways to keep the weight down. “As additive manufacturing continues to advance, it may not be long before instruments are routinely made with a shelled inner core to reduce weight,” said Neal. “Not only does this lighten the weight, these instruments cool down more quickly after autoclaving, which means they can be used sooner than solid-mass instruments, which take a longer time to cool.”
It is also critical to surgeons that their instruments have the right balance and “feel”—which is why manufacturers of these products expect their contract design and manufacturing partners to utilize human factors and usability engineering in the early design process. “We need to think procedurally to understand how our device solution is going to fit within the proposed clinical workflow,” said Schechter. “This typically involves detailed task mapping and storyboarding to understand user interactions with the device, systems we need to be compatible with, and clinical variability patient to patient that is likely to be encountered.”
Additive Manufacturing
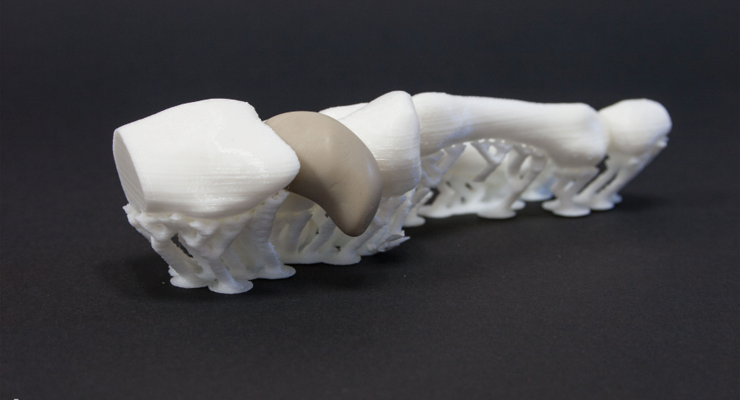
Additive manufacturing/3D printing (AM/3DP) is fast-becoming an essential tool for prototyping and design. Easy accessibility to these processes gives device manufacturers the ability to design and build prototypes and devices that cannot not be machined or built otherwise in a cost-effective manner—a capability that greatly expands engineering and design options. AM is used to print custom-made orthopedic implants using patient-specific measurements. Recent advancements in AM methods and materials have improved the regularity of internal pore structure patterns, creating complex, repeatable geometries that improve osseointegration.
“Rapid advancement in 3D printing technology and materials are giving us the ability to truly create ‘looks like, works like’ prototypes,” said Schechter. “We can even bring some of these materials through ethylene oxide sterilization, or even autoclave sterilization.”
Although lower-cost AM equipment is available, these machines tend to have limited capability (however, they do help small and mid-size CMs be more competitive). Higher-end machines are capable of producing medical-grade devices. For example, Apium Additive Technologies GmbH recently released its M220 3D printer, which is designed specifically to manufacture implants and other medical devices out of polyether ether ketone (PEEK). The machine features a sterile printing environment and new extrusion technology, which has a much higher feed force that enables more precise material flow. All components in contact with the material being printed are made of medically compatible materials—PEEK, titanium, 316L steel, and polytetrafluoroethylene (PTFE). Developed in cooperation with Evonik, the machine can make up to Class III deposits from PEEK.
Skåne University Hospital in Sweden recently became the first hospital in the world to 3D print its own cranioplasty implant using this technology. “With this method, we will have the entire chain, from designing and printing implants to fitting and surgery, in hospital,” said Einar Heiberg Brandt, medical engineer in Clinical Physiology and Nuclear Medicine at Skåne University Hospital and Wallenberg Centres for Molecular Medicine. “This will lead to faster surgeries and fewer complications. This is just the beginning—there are no barriers to writing other types of bone implants.”1
Although AM technology continues to develop at a rapid pace—as Apium shows—”the ability to use AM for high-volume runs is still not feasible yet,” said Brandon Mitchell, sales engineer with Lowell. “Also, surface finishes can still be problematic with AM. However, AM can be an excellent choice for parts that have complicated internal features, such as lattice structures, that cannot be manufactured with conventional machining. In general, however, it is still more cost-effective to run a low-volume part with CNC machining methods versus running it with AM. In general, if a part can be traditionally machined, then it probably should be.”
Innovation Overcomes Challenges
New technologies are difficult to get through the regulatory process, typically requiring large capital investments and years of clinical data gathering. However, with the right product and/or the right material, the payoff can be huge. With the pandemic easing, OEMs are cautiously investing more dollars in R&D, especially in product groups they already know.
For example, porous coatings are relatively stable, mature technologies. Newer surface modifications, such biomimetic coatings, may improve the already-high survivorship of primary implants with porous coatings. One area of current research is the mixing of therapeutic agents with the coatings that will elute via time-release mechanisms. “The challenge is to demonstrate the value of these methods through clinical trials, which are very expensive to conduct,” said Jani. “OEMs are reluctant to follow this path if the value is only a marginally improved outcome, even when the improvement is dramatic on a patient-by-patient level. For example, implant infection is generally thought to affect approximately 1 to 2 percent of primary implants. That may be a low incidence, but for the patient who does develop an infection, it is 100 percent undesirable. How do we cut the infection rate down to less than 0.1 percent? And can it be done in a manner that is cost-effective?”
In the constant race to develop new products and get them to market quickly, MDMs can sometimes lose sight of the importance of manufacturability of their design, which can be a costly mistake. They must be careful to understand the limitations of rapid prototyping manufacturing processes during the design cycle—especially considering the constant pressure to increase development speed and shorten time to market. Although rapid prototyping processes can quickly get parts into the hands of the engineering team to assess the feasibility of the design, the team must stay aware of how that product will be produced. “If they are not careful, too often developers will be led down a design path that ultimately is not manufacturable at scale and they will not be able to achieve a target cost point during commercial production,” advised Schechter.
Surgery robots have advanced greatly in terms of accuracy and patient outcomes. One of the most challenging parts of implant surgery is the accurate placement of the individual components of the implant to ensure stability and smooth operation. Advances in robotic arm technology provide enhanced visual, tactile, and auditory feedback, making it easier for the surgical team to achieve ideal placement. Surgical robots are also fast-becoming an integral part of a wide range of minimally invasive procedures, which is a key reason for the rapid expansion of these procedures and the quick recovery times for patients.
In contrast, smart devices for surgical interventions that utilize advanced sensors and artificial intelligence for active feedback are gaining attention. Handheld, stand-alone smart instruments with disposables are taking on the big robotic equipment. With the recent advancements in gaming and proximity sensors, spine screw, hip acetabulum, and shoulder alignment guides are being developed to locate implant positioning relative to the anatomy at a fraction of the cost of robotic systems. The disposables also may very well lend themselves to being built with the AM process, noted Neal. “3D printing is especially useful in the prototype stage,” he said. “It allows engineers to quickly iterate designs without the initial costs and lead times associated with one-off machining or investing in a mold.”
Partnering Up
The current supply chain crisis, especially in electronics, is not only creating scheduling delays, but entire projects may be put at risk if a component that was qualified and available during prototyping suddenly goes into shortage. “Our team of sourcing and supply chain professionals are constantly communicating with our suppliers and monitoring the industry to identify risks,” said Sullivan. “Decisions can then be made quickly to steer not only new product development, but also production to a healthy supply base, knowing that new components and materials must have a robust product verification and validation, which takes significant time in order to ensure patient safety.”
For best results, especially in the age of COVID-19, MDMs should work collaboratively with trusted suppliers and rely on their expertise to build a better product and navigate the supply chain (sometimes to find alternative materials and components that will keep a project on track when shortages arise). “We have many years of experience with products that are similar to what customers bring in,” said Cabral. “The manufacturing knowledge we have gained from making these products, as well as product development in other related fields, combined with our willingness to work diligently with the customer’s team, enables us to provide a device that totally meets the design requirements and exceeds a surgeon’s expectations.”
Reference
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.
“Product development seems to be taking on more of the projects that have been on the back burner,” said David Neal, president of STAT DESIGN, a Lake Hopatcong, N.J.-based business that provides CAD design, development, and 3D printing services to the medical device industry. “With the daily fires to put out from COVID-19 slowing down a bit, there is more time to work on some of the other projects that may have gotten pushed down the priority list.”
Some of these projects involve a merging of technologies, including minimally invasive interventional approaches, advanced imaging and navigational systems, and greater adoption of robotics. “Product design has become more focused on procedural solutions rather than discrete devices,” said David Schechter, president of Meddux Development Corporation, a Boulder, Colo.-based medical device design, development, and manufacturing partner for complex single-use devices. “This requires a committed emphasis on human factors/usability engineering and incorporating a systems approach to product development.”
Quick turnaround is essential for prototyping these complex devices and a manufacturable design is critical for achieving that expectation. “We have experienced increased need for prototypes due to the expected upticks in surgeries and product development after the last two years of the pandemic,” said David Cabral, president and CEO of Five Star Companies, a New Bedford, Ma.-based contract manufacturer of precision instruments in the ortho, spine, and sports medicine markets. “Surgeries have opened up and product development is in high gear.”
There are several ways to outsource prototyping, including stand-alone prototype shops that specialize in quick turns of initial designs and 3D printing service bureaus that convert solid models into functional parts in metal or plastic in a matter of hours. A third option is “the contract manufacturer that can machine complex rapid prototypes and then smoothly transition into higher volume production,” said Philip Allen, vice president of sales and marketing for Lowell, a Minneapolis, Minn.-based contract manufacturer of complex implants and instruments for the orthopedic and cardiovascular markets. “Each of these three options fills an important role in the production of prototypes. As a contract manufacturer, our focus is on complex prototypes that lead to the production of complex implants and instruments.”
Latest Trends
Implant manufacturers are intent on finding new surface treatments to make their implants more effective and long-lasting. Porous surfaces (also known as porous coatings) have been used on orthopedic implants for more than 30 years to facilitate bony in-growth and long-term fixation. “With joint replacement now more common in younger and active patients, there is more demand for improved implant performance and safety, and porous coatings play an important role in successful implantation and longevity,” said Shilesh Jani, engineering manager for Orchid Orthopedic Solutions, a Mason, Mich.-based developer and contract manufacturer of medical devices and implants.
For implant surfaces, orthopedic OEMs are becoming more particular about certain factors, such as coating thickness, and want to increase pore size, porosity, and mechanical strength—all key factors that impact osseointegration and the safety of the implant. “OEMs also want coatings to be rougher, with a higher friction coefficient than for previous generations of coatings,” said Jani.
Allen sees a trend toward blending 3D-printed prototypes with machined complex features.
“There are tremendous advantages in using this approach,” said Allen. “Many 3D-printed prototypes have a tolerance range between ±0.003 inches to ±0.01 inches, depending on factors like part size, material selection, and technology being utilized. However, if features that are critical to the design must be held tighter, those features can be machined into the part. This approach combines the best of both worlds—the speed of additive manufacturing with the ability to add complex, close-tolerance features using a subtractive or machined approach.”
For example, a new cervical plate design requires a screw hole that must have a very specific feel for a surgeon when the screw is introduced to the plate during laboratory testing. If the prototype does not perform like the production part, the entire project can be at risk. A typical complex machined prototype of a cervical plate could take up to six weeks; plates made through a 3D-printed process take a week or so. A subtractive machining process can then be used to add the complex screw hole to the plate in another week—cutting the development time in half, yet retaining the features the surgeon had identified.
As devices become increasingly complex, with more parts and added functionality, there is greater need for design for manufacturing (DFM) to identify the best and most cost-effective manufacturing process. Using DFM during the prototyping stage will identify and work out any design flaws before the production process gets locked in. Finite element analysis (FEA) is a crucial DFM tool that predicts how a proposed medical device will respond to real-world forces such as vibration, load, heat, fluid flow, electrostatics, and other physical effects. FEA will reveal a product’s most probable points of failure, which can then be fixed through redesign if needed.
“We use FEA with a tighter range of material properties to ensure strength and durability without major functional or feature changes to the components,” said Dennis Sullivan, senior manager of advanced sourcing for Medtronic Surgical Robotics, a Minneapolis, Minn.-based developer and manufacturer of robotic surgical solutions. “Recently, we went from a machined aluminum housing to a casting. Using FEA, we identified a specific material from one of our suppliers that allowed us to maintain the original design. While we still need to validate the process, the design remains intact with minimal impact.”
DFI—design for inspection—is another process that evaluates how a component will be inspected and how the design can be modified to make the inspection process as clear as possible. Another helpful program is DFSS or design for Six Sigma, which improves the quality of the design/production process by incorporating lean processes and eliminating waste (fewer steps). All these methods, when undertaken during the design phase, optimize efficiency and quality and reduce the probability that redesign and reiteration will be required.
What OEMs Want
Speed is everything, especially during early concept development, when there are plenty of good ideas to test. It is essential for medical device manufacturers (MDMs) to have supply chain partners that can iterate and prototype quickly, so critical feedback from surgeons and clinicians can be included during the design cycle, saving time and money.
Accelerated design, production, and time to market are still the top requests of orthopedic companies.
“I want a supplier that will partner with me to do everything we can, up front, to ensure speed on the back end of a project,” said Sullivan. “This includes not only things like DFM and process development, but also putting fair contracts in place for mutual protection.”
First iterations typically require very fast lead times—in this early stage OEMs want to put a solid, 3D representation of the concept into the hands of their decision-makers, rather than simply show them an image on the screen. As the team gets closer to locking in a design, “it becomes more important to have a part that is as close to representing the final product as possible, to enable an easy transition to production,” said Allen. “In this stage, the ability to match the print and function for final testing becomes critical.”
Faster speed also has a role in the other top items on the OEM wish list. This growing variety of tasks reflects how OEMs increasingly expect their contract manufacturers (CMs) to take on more product development responsibilities in-house—even regulatory compliance expertise. Capabilities on the list include modifications to existing parts, compliance certifications, and cost controls (for example, competitive and/or effortless procurement of raw materials) that do not compromise device function.
“Quick turnaround speed is always essential,” said Robert Suric, global sales manager for gSource, an Emerson, N.J.-based provider of handheld, non-powered surgical instruments. “Faster product development can also be accomplished through a shorter supply chain, vertical integration, streamlined logistics, and supply chain management solutions that result in getting raw materials on time, or even having them in stock.”
With the COVID-19 pandemic still present, a vertically integrated supplier is both safer and faster (shorter supply chain).
“Large or small company, it doesn’t matter,” added Sullivan. “A supplier that can perform the component fabrication, including finishing and assembly, is in high demand. With the lack of resources these days, this is a more effective approach than trying to schedule multiple subcontractors, which inevitably creates delays and adversely affects project timelines.”
Although they are still highly cost-conscious, OEMs are interested in more multi-functional and complex designs, products, and assemblies and expect their CMs to have the answers. “A good example is an instrument that can insert, expand, rotate, and remove without the need to use multiple devices,” said Cabral. “This saves surgeons time, reduces fatigue, and typically results in better patient outcomes.”
Another cost-effective solution is reducing the weight of surgical instruments—although sales and marketing teams tout the benefit of combining instruments into a single kit, engineers struggle to keep the weight under 25 pounds (as per recommendations from the Association of periOperative Registered Nurses and the Association for the Advancement of Medical Instrumentation). Using modular handles and hollowing out instruments are two ways to keep the weight down. “As additive manufacturing continues to advance, it may not be long before instruments are routinely made with a shelled inner core to reduce weight,” said Neal. “Not only does this lighten the weight, these instruments cool down more quickly after autoclaving, which means they can be used sooner than solid-mass instruments, which take a longer time to cool.”
It is also critical to surgeons that their instruments have the right balance and “feel”—which is why manufacturers of these products expect their contract design and manufacturing partners to utilize human factors and usability engineering in the early design process. “We need to think procedurally to understand how our device solution is going to fit within the proposed clinical workflow,” said Schechter. “This typically involves detailed task mapping and storyboarding to understand user interactions with the device, systems we need to be compatible with, and clinical variability patient to patient that is likely to be encountered.”
Additive Manufacturing
Additive manufacturing/3D printing (AM/3DP) is fast-becoming an essential tool for prototyping and design. Easy accessibility to these processes gives device manufacturers the ability to design and build prototypes and devices that cannot not be machined or built otherwise in a cost-effective manner—a capability that greatly expands engineering and design options. AM is used to print custom-made orthopedic implants using patient-specific measurements. Recent advancements in AM methods and materials have improved the regularity of internal pore structure patterns, creating complex, repeatable geometries that improve osseointegration.
“Rapid advancement in 3D printing technology and materials are giving us the ability to truly create ‘looks like, works like’ prototypes,” said Schechter. “We can even bring some of these materials through ethylene oxide sterilization, or even autoclave sterilization.”
Although lower-cost AM equipment is available, these machines tend to have limited capability (however, they do help small and mid-size CMs be more competitive). Higher-end machines are capable of producing medical-grade devices. For example, Apium Additive Technologies GmbH recently released its M220 3D printer, which is designed specifically to manufacture implants and other medical devices out of polyether ether ketone (PEEK). The machine features a sterile printing environment and new extrusion technology, which has a much higher feed force that enables more precise material flow. All components in contact with the material being printed are made of medically compatible materials—PEEK, titanium, 316L steel, and polytetrafluoroethylene (PTFE). Developed in cooperation with Evonik, the machine can make up to Class III deposits from PEEK.
Skåne University Hospital in Sweden recently became the first hospital in the world to 3D print its own cranioplasty implant using this technology. “With this method, we will have the entire chain, from designing and printing implants to fitting and surgery, in hospital,” said Einar Heiberg Brandt, medical engineer in Clinical Physiology and Nuclear Medicine at Skåne University Hospital and Wallenberg Centres for Molecular Medicine. “This will lead to faster surgeries and fewer complications. This is just the beginning—there are no barriers to writing other types of bone implants.”1
Although AM technology continues to develop at a rapid pace—as Apium shows—”the ability to use AM for high-volume runs is still not feasible yet,” said Brandon Mitchell, sales engineer with Lowell. “Also, surface finishes can still be problematic with AM. However, AM can be an excellent choice for parts that have complicated internal features, such as lattice structures, that cannot be manufactured with conventional machining. In general, however, it is still more cost-effective to run a low-volume part with CNC machining methods versus running it with AM. In general, if a part can be traditionally machined, then it probably should be.”
Innovation Overcomes Challenges
New technologies are difficult to get through the regulatory process, typically requiring large capital investments and years of clinical data gathering. However, with the right product and/or the right material, the payoff can be huge. With the pandemic easing, OEMs are cautiously investing more dollars in R&D, especially in product groups they already know.
For example, porous coatings are relatively stable, mature technologies. Newer surface modifications, such biomimetic coatings, may improve the already-high survivorship of primary implants with porous coatings. One area of current research is the mixing of therapeutic agents with the coatings that will elute via time-release mechanisms. “The challenge is to demonstrate the value of these methods through clinical trials, which are very expensive to conduct,” said Jani. “OEMs are reluctant to follow this path if the value is only a marginally improved outcome, even when the improvement is dramatic on a patient-by-patient level. For example, implant infection is generally thought to affect approximately 1 to 2 percent of primary implants. That may be a low incidence, but for the patient who does develop an infection, it is 100 percent undesirable. How do we cut the infection rate down to less than 0.1 percent? And can it be done in a manner that is cost-effective?”
In the constant race to develop new products and get them to market quickly, MDMs can sometimes lose sight of the importance of manufacturability of their design, which can be a costly mistake. They must be careful to understand the limitations of rapid prototyping manufacturing processes during the design cycle—especially considering the constant pressure to increase development speed and shorten time to market. Although rapid prototyping processes can quickly get parts into the hands of the engineering team to assess the feasibility of the design, the team must stay aware of how that product will be produced. “If they are not careful, too often developers will be led down a design path that ultimately is not manufacturable at scale and they will not be able to achieve a target cost point during commercial production,” advised Schechter.
Surgery robots have advanced greatly in terms of accuracy and patient outcomes. One of the most challenging parts of implant surgery is the accurate placement of the individual components of the implant to ensure stability and smooth operation. Advances in robotic arm technology provide enhanced visual, tactile, and auditory feedback, making it easier for the surgical team to achieve ideal placement. Surgical robots are also fast-becoming an integral part of a wide range of minimally invasive procedures, which is a key reason for the rapid expansion of these procedures and the quick recovery times for patients.
In contrast, smart devices for surgical interventions that utilize advanced sensors and artificial intelligence for active feedback are gaining attention. Handheld, stand-alone smart instruments with disposables are taking on the big robotic equipment. With the recent advancements in gaming and proximity sensors, spine screw, hip acetabulum, and shoulder alignment guides are being developed to locate implant positioning relative to the anatomy at a fraction of the cost of robotic systems. The disposables also may very well lend themselves to being built with the AM process, noted Neal. “3D printing is especially useful in the prototype stage,” he said. “It allows engineers to quickly iterate designs without the initial costs and lead times associated with one-off machining or investing in a mold.”
Partnering Up
The current supply chain crisis, especially in electronics, is not only creating scheduling delays, but entire projects may be put at risk if a component that was qualified and available during prototyping suddenly goes into shortage. “Our team of sourcing and supply chain professionals are constantly communicating with our suppliers and monitoring the industry to identify risks,” said Sullivan. “Decisions can then be made quickly to steer not only new product development, but also production to a healthy supply base, knowing that new components and materials must have a robust product verification and validation, which takes significant time in order to ensure patient safety.”
For best results, especially in the age of COVID-19, MDMs should work collaboratively with trusted suppliers and rely on their expertise to build a better product and navigate the supply chain (sometimes to find alternative materials and components that will keep a project on track when shortages arise). “We have many years of experience with products that are similar to what customers bring in,” said Cabral. “The manufacturing knowledge we have gained from making these products, as well as product development in other related fields, combined with our willingness to work diligently with the customer’s team, enables us to provide a device that totally meets the design requirements and exceeds a surgeon’s expectations.”
Reference
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.