Seth Goldenberg , Vice President, Veeva MedTech11.15.22
The medtech industry is dealing with growing complexity in getting products to patients, including new regulatory requirements, intense competition, and the increasing cost of compliance. The European Medical Device Regulation (EU MDR), for example, emphasizes transparency and provides a regulatory framework to ensure device safety and performance. With EU MDR, experts predict that approximately 30% of small medtech manufacturers may go out of business because they don’t have the capabilities to fulfill the requirements.1 Companies will need to take action and address the shortcomings of existing operational systems to prevent this outcome.
Only 56% of senior regulatory leaders say their companies completed or began modernizing global regulatory operations, leaving nearly half of organizations inhibited by traditional, older ways of working.2 The reliance on manual and siloed processes increases the risk of falling behind on regulatory requirements, since companies can’t keep up with the regulatory rate of change.
Medtech companies can gain a competitive edge by streamlining regulatory affairs using technology. Transforming regulatory operations can drive more process harmonization and standardization across the product lifecycle. In this article, we’ll discuss the state of regulatory affairs and identify opportunities for greater efficiency.
Consider that:
The regulatory function is an integral part of an organization, from pre-market strategy to post-market surveillance efforts, and leaders recognize it is time for a new approach. Numerous organizations have initiatives to integrate regulatory affairs across traditionally segregated departments and regions. This can enhance content management processes for real-time access to documents and enable workflow automation for greater flexibility and speed.
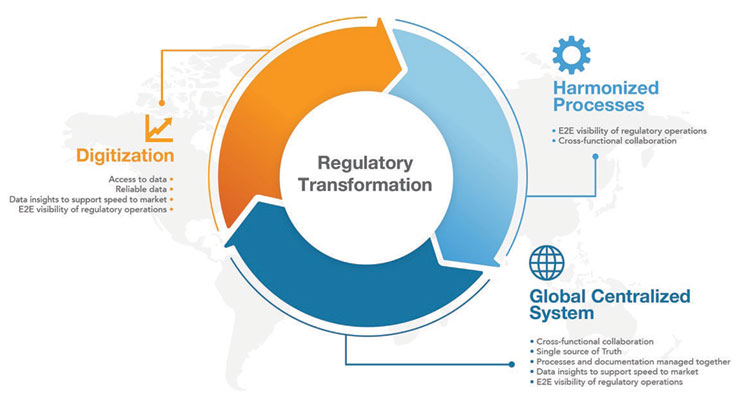
Implementing unified cloud-based regulatory applications that meet unique medtech requirements can help companies achieve this goal. Regulatory teams can access reliable data remotely and gain visibility across products or regions. Harmonized processes enable cross-functional collaboration for greater speed to insights and innovation. With a centralized global system, users gain a single source of truth, where processes and documentation can be managed together.
The modernization efforts lead to accurate and reliable data for more insights and better decision-making. Regulatory teams can then start collecting information sooner and with confidence to commercialize products in new countries. This is a significant improvement over manual processes and isolated activities that present problems when sharing and analyzing results.
With a foundation that enables end-to-end regulatory visibility, teams can respond swiftly to internal and external events affecting submissions and registrations. The improvements allow a shift in how medtech regulatory affairs are managed, moving away from reactive methods toward more proactive approaches. Still, only 7% of medtech companies report generating, saving, and managing product regulatory information online. With regulatory authorities scrutinizing consistent usage of content and information across documents, reports, and other assets, the time for change is now.
As the pace of innovation accelerates, regulatory strategy is becoming critical in medtech organizations. With visibility and clarity across products and regions, regulatory affairs can deliver an early understanding of the commercial strategy and product distribution and provide a strategic advantage in driving product success.
References
Seth J. Goldenberg, Ph.D., is responsible for Veeva MedTech’s global strategy, including customer engagement, market adoption, and product development. Goldenberg has nearly 20 years of experience supporting medtech companies as they navigate complex regulations and improve market access. Before joining Veeva, Goldenberg was director of product development strategy at North American Science Associates (NAMSA), where he supported medical device companies from inception through commercialization and post-market activities. Outside of Veeva, Goldenberg is an active member of the Regulatory Affairs Professionals Society (RAPS) and is the "entrepreneur in residence" at the Pennsylvania Pediatric Device Consortium. He holds a doctorate in pharmacology from the University of Washington and a master's degree from the school of biomedical engineering at Drexel University. He can be reached at seth.goldenberg@veeva.com.
Only 56% of senior regulatory leaders say their companies completed or began modernizing global regulatory operations, leaving nearly half of organizations inhibited by traditional, older ways of working.2 The reliance on manual and siloed processes increases the risk of falling behind on regulatory requirements, since companies can’t keep up with the regulatory rate of change.
Medtech companies can gain a competitive edge by streamlining regulatory affairs using technology. Transforming regulatory operations can drive more process harmonization and standardization across the product lifecycle. In this article, we’ll discuss the state of regulatory affairs and identify opportunities for greater efficiency.
Shortcomings in Submission Planning and Regulatory Interactions
The current condition of medtech regulatory operations, including worldwide compliance and visibility—speed to market, and post-market compliance—highlights that more work lies ahead.Consider that:
- 71% of companies develop submission content in isolated, regional document management systems
- 57% generate performance and KPI reports manually
- 57% report inconsistent or unstructured data gathering when organizing and tracking international registrations
- Over 66% of respondents still manage submission documents on local laptops, file shares, or regional document management systems2
The regulatory function is an integral part of an organization, from pre-market strategy to post-market surveillance efforts, and leaders recognize it is time for a new approach. Numerous organizations have initiatives to integrate regulatory affairs across traditionally segregated departments and regions. This can enhance content management processes for real-time access to documents and enable workflow automation for greater flexibility and speed.
Establishing a Foundation for Advanced Regulatory Affairs
To modernize regulatory strategy, companies need seamless collaboration between business and IT teams to evaluate and implement new systems and processes. Focusing on three areas (Figure 1), including digitization, harmonized processes, and a centralized global system, strongly supports end-to-end regulatory operations visibility.Implementing unified cloud-based regulatory applications that meet unique medtech requirements can help companies achieve this goal. Regulatory teams can access reliable data remotely and gain visibility across products or regions. Harmonized processes enable cross-functional collaboration for greater speed to insights and innovation. With a centralized global system, users gain a single source of truth, where processes and documentation can be managed together.
The modernization efforts lead to accurate and reliable data for more insights and better decision-making. Regulatory teams can then start collecting information sooner and with confidence to commercialize products in new countries. This is a significant improvement over manual processes and isolated activities that present problems when sharing and analyzing results.
With a foundation that enables end-to-end regulatory visibility, teams can respond swiftly to internal and external events affecting submissions and registrations. The improvements allow a shift in how medtech regulatory affairs are managed, moving away from reactive methods toward more proactive approaches. Still, only 7% of medtech companies report generating, saving, and managing product regulatory information online. With regulatory authorities scrutinizing consistent usage of content and information across documents, reports, and other assets, the time for change is now.
Moving From Vision to Reality
Two common goals propelling regulatory teams to transform operations are improving compliance and accelerating time-to-market. To keep track of approvals and submission revisions throughout the entire product development lifecycle, it is essential to have processes and documents that work together and support seamless submissions. As more medtech companies start to think about modernizing their regulatory operations, they must first lay the groundwork by bringing together key stakeholders, processes, and systems necessary for success. With clear organizational alignment and digital technology, medtech can harmonize and centralize regulatory operations.As the pace of innovation accelerates, regulatory strategy is becoming critical in medtech organizations. With visibility and clarity across products and regions, regulatory affairs can deliver an early understanding of the commercial strategy and product distribution and provide a strategic advantage in driving product success.
References
- CX International Symposium, Medical device industry leaders call for three big changes to “devastating” EU MDR legislation, 2019
- Veeva MedTech, MedTech 2021 Regulatory Pulse Benchmark Report, 2021
Seth J. Goldenberg, Ph.D., is responsible for Veeva MedTech’s global strategy, including customer engagement, market adoption, and product development. Goldenberg has nearly 20 years of experience supporting medtech companies as they navigate complex regulations and improve market access. Before joining Veeva, Goldenberg was director of product development strategy at North American Science Associates (NAMSA), where he supported medical device companies from inception through commercialization and post-market activities. Outside of Veeva, Goldenberg is an active member of the Regulatory Affairs Professionals Society (RAPS) and is the "entrepreneur in residence" at the Pennsylvania Pediatric Device Consortium. He holds a doctorate in pharmacology from the University of Washington and a master's degree from the school of biomedical engineering at Drexel University. He can be reached at seth.goldenberg@veeva.com.