Muhammad Jamil, Ph.D., Computational Fluid Dynamics Engineer, StarFish Medical09.12.23
Developing a medical device is inherently associated with risk. Regulatory bodies such as Health Canada or the U.S. Food and Drug Administration (FDA) provide regulatory oversight for physical risk to the patient, because patient safety is critical. To minimize risk, companies invest heavily in research and development, multiple design iterations/explorations, prototyping, animal or bench experiments, or clinical trials.
However, there are significant costs related to all these processes and they prolong the time for a device to go from inception to market.
In silico medicine—where computer simulations based on the principles of engineering, mathematics, and computational sciences—is becoming an integral part of medical device development. It’s an attractive option because it can be a useful tool to minimize risk throughout a product’s life cycle through multiple design iterations at fraction of the cost. This provides access to a wealth of data not possible through animal/bench testing, carefully designing clinical trials to reduce costs, simulating situations which involve hazardous chemicals, or performing too risky or infeasible experiments.
The most important aspect of the risk is patient injury. Health Canada and FDA regulatory oversight tries to ensure physical risk is minimized. They categorize medical devices based on the risk(s) they pose to the patient, maintain active market surveillance, and publish relevant statistical data for the general public.
Health Canada MDI reports increased from 4,008 in 2008 to 11,307 in 2017. Moreover, the seriousness of the incidents also increased in the same period from 587 in 2008 to 1,721 in 2017. Incidents resulting in serious health risk or death must be reported within 10 days and are categorized as most serious.
There were 5,741 MDI reports submitted from December 2019 to February 2023. Although there is steady decline in the MDI reports, there’s still significant risk associated with the medical devices.
Medical devices are categorized by Health Canada between Class I-IV with increasing level of risk associated with them (Class IV carries the highest risk). Most risk is associated with insulin pumps, implantable cardioverter defibrillators (ICDs), tissue heart valves, etc. that resulted in patient deaths.¹
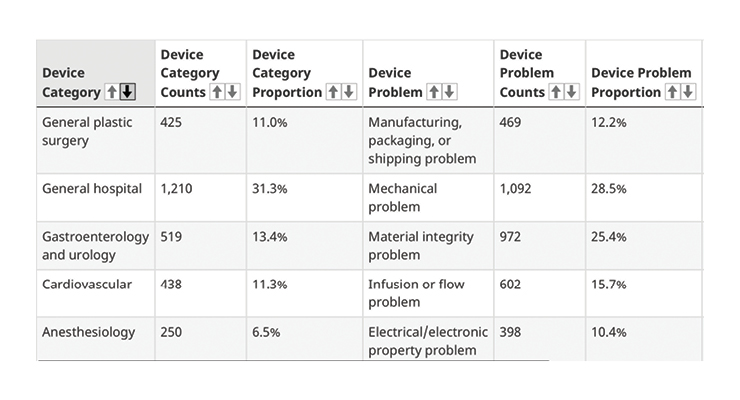
Based on reported data from December 2019 to February 2023 of MDI reports, more than 50% of the incidents were linked to mechanical, mechanical integrity, or flow/infusion problems.2
Based on 2009-2017 CMDSNet report data,3 more than 80% of the device problems were linked to defect, malfunction, failure, breakage, and leakage. A comparison of recent data to the past indicates the major issues remain the same.
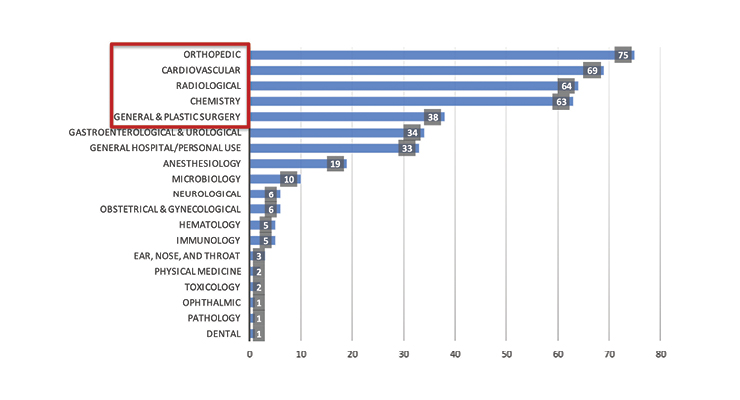
Based on FDA Class I recall data from January 2016 to December 2018, device design is the most common cause of recalls.4 Based on Class II recall data of devices that caused medium risk from October 2019 to September 2020, device design is the second major cause of recalls, after process control.
Most of the recalls were from devices used in orthopedic, cardiovascular, radiological, and chemical procedures—industries that require more invasive treatments are associated with higher risk. The data suggests that in silico medicine has huge potential to add value to medical device development through risk reduction.
The true value of in silico simulations compared to traditional methods is expressed in high-risk situations. Risk can be minimized by testing riskier medical device situations in silico. Invasive devices and implants can be tested in vivo without causing harm to patients. Utilizing virtual patient models reveals device behavior and predicts associated risks. Hazardous materials or environments can also be simulated in silico.
In summary, in silico simulations provide a more comprehensive design and development process to reduce risks associated with medical devices. In silico simulations can lower risk-associated device incidents, failures, and recalls, reducing the financial stakes.
References
Muhammad Jamil, Ph.D., is a computational fluid dynamics (CFD) engineer in the Design and Analysis group at StarFish Medical. As part of the design and development team, he specializes in using computational modeling and simulation in making design decisions and to accelerate the medical device development.
However, there are significant costs related to all these processes and they prolong the time for a device to go from inception to market.
In silico medicine—where computer simulations based on the principles of engineering, mathematics, and computational sciences—is becoming an integral part of medical device development. It’s an attractive option because it can be a useful tool to minimize risk throughout a product’s life cycle through multiple design iterations at fraction of the cost. This provides access to a wealth of data not possible through animal/bench testing, carefully designing clinical trials to reduce costs, simulating situations which involve hazardous chemicals, or performing too risky or infeasible experiments.
Is In Silico Medicine the Answer to Minimize Risk?
Before jumping to the question, it’s appropriate to define risk severity and delve into available statistical data published by regulatory bodies.The most important aspect of the risk is patient injury. Health Canada and FDA regulatory oversight tries to ensure physical risk is minimized. They categorize medical devices based on the risk(s) they pose to the patient, maintain active market surveillance, and publish relevant statistical data for the general public.
Medical Device Incidents (MDI) in Canada
Health Canada is responsible for maintaining effective regulatory oversight within Canada to minimize risk. Health Canada ensures post-market surveillance through MDI data reported via the Canada Vigilance-Medical Device System (CV-MDS) and Canadian Medical Devices Sentinel Network (CMDSNet) programs.Health Canada MDI reports increased from 4,008 in 2008 to 11,307 in 2017. Moreover, the seriousness of the incidents also increased in the same period from 587 in 2008 to 1,721 in 2017. Incidents resulting in serious health risk or death must be reported within 10 days and are categorized as most serious.
There were 5,741 MDI reports submitted from December 2019 to February 2023. Although there is steady decline in the MDI reports, there’s still significant risk associated with the medical devices.
Medical devices are categorized by Health Canada between Class I-IV with increasing level of risk associated with them (Class IV carries the highest risk). Most risk is associated with insulin pumps, implantable cardioverter defibrillators (ICDs), tissue heart valves, etc. that resulted in patient deaths.¹
Based on reported data from December 2019 to February 2023 of MDI reports, more than 50% of the incidents were linked to mechanical, mechanical integrity, or flow/infusion problems.2
Based on 2009-2017 CMDSNet report data,3 more than 80% of the device problems were linked to defect, malfunction, failure, breakage, and leakage. A comparison of recent data to the past indicates the major issues remain the same.
FDA Recall Data
FDA maintains the record of recall data in United States. FDA classifies recalls based on the probability of causing harm or adverse effects. Recalls with the highest probability of causing harm to patients are categorized as Class I; those posing the lowest probability of harm are classified as Class III.Based on FDA Class I recall data from January 2016 to December 2018, device design is the most common cause of recalls.4 Based on Class II recall data of devices that caused medium risk from October 2019 to September 2020, device design is the second major cause of recalls, after process control.
Most of the recalls were from devices used in orthopedic, cardiovascular, radiological, and chemical procedures—industries that require more invasive treatments are associated with higher risk. The data suggests that in silico medicine has huge potential to add value to medical device development through risk reduction.
In Silico Simulations Improve and Innovate Design Process
For the biggest risk factor—device design—in silico simulations provide the flexibility to test different designs and reduce risk by identifying failure modes, testing under extreme conditions, and identifying worst case scenarios.The true value of in silico simulations compared to traditional methods is expressed in high-risk situations. Risk can be minimized by testing riskier medical device situations in silico. Invasive devices and implants can be tested in vivo without causing harm to patients. Utilizing virtual patient models reveals device behavior and predicts associated risks. Hazardous materials or environments can also be simulated in silico.
In Silico Simulations Reduce Costs and Development Time
In silico simulations can form the basis of a robust development process, where different designs can be tested in short periods of time by predicting failure in advance, testing in extreme conditions, and designing bench experiments. Simulations can also be a tool to design and optimize clinical trials, greatly reducing time and costs.In summary, in silico simulations provide a more comprehensive design and development process to reduce risks associated with medical devices. In silico simulations can lower risk-associated device incidents, failures, and recalls, reducing the financial stakes.
References
Muhammad Jamil, Ph.D., is a computational fluid dynamics (CFD) engineer in the Design and Analysis group at StarFish Medical. As part of the design and development team, he specializes in using computational modeling and simulation in making design decisions and to accelerate the medical device development.