Sam Brusco, Associate Editor11.13.23
The beginnings of intramedullary (IM) fixation go all the way back to the 16th century. The conquistadors in Mexico documented that Aztec physicians would use solid, wooden pegs in order to treat non-union fractures. In the procedure, the bone was scraped and a stick of resinous wood was put into the medullary canal to set the bone firmly.
In the late 19th century, ivory IM nails were introduced in Germany for femoral shaft fractures and became the material of choice for IM nails until 1910. This period also witnessed the development of interlocking nails and pins made from ivory to boost rotational stability. However, it later became apparent that ivory materials are resorbable—meaning ivory implants couldn’t offer sufficient stability to health fractures, particularly for cases involving delayed unions.
This led to experimentation with other metallic nails over the years: aluminum IM splints for femoral fractures, silver pins for upper limb fractures, and stainless steel or chromicized brass nails were used.
The modern use of stainless steel IM nails as the gold standard to treat femoral shaft fractures began with German surgeon Gerhard Küntscher during World War II. Then called the Küntscher nail, his first invention was a V-shaped, stainless steel IM nail. Küntscher published results of 39 cases in 1939 and, in 1947, another study with 107 war patients whose femoral and tibia shaft fractures were treated with IM nails. The nail’s use expanded in the United States after the first American soldiers arrived back from the war with German IM nails.
In the 1960s, titanium was introduced for IM nails after image intensifiers were invented. These nails boasted a Young Modulus closer to intact diaphyseal cortical bone, higher fatigue and yield tensile strength, and lower density compared to the stainless steel IM nails. Another turning point came about in 1986, when the non-slotted, femoral IM nail was made possible using a gun-drilling, computerized machining technique. This allowed construction of a nail with lower bending stiffness and better rotational stability. Non-slotted IM nails subsequently became the preferred technology up to the 21st century.
Three main novel IM nails have been identified in the last 20 years: expandable, coated, and carbon fiber nails.
Orthopedic device giant Stryker’s legacy T2 nailing platform is one of the mainstays of the company’s orthopedic trauma surgery technology portfolio. The T2 platform of IM nails are able to treat a wide range of fractures in the upper and lower extremities. The nails feature type II anodization for better fatigue strength, active internal compression, and a common instrumentation platform. They also tout radiolucent targeting arms with easy assembly and friction lock technology, Stryker describes.
The latest in this line, the T2 Alpha femur retrograde nailing system, hit the market in August 2021. According to Stryker, it features a redefined, data-driven nail design and dedicated proximal targeting system that could reduce the number of X-ray shots as compared to freehand locking.
“T2 Alpha Femur Retrograde is based on decades of experience with the Stryker T2 system,” Bernd Simon, senior principal engineer of R&D at Stryker, told ODT. “It is designed to provide an intraoperative adaptability with more fixation options in the nail, internal and external compression and the option to use Advanced Locking Screws for added axial and angular stability.1 The length-dependent curvature of the nail shows superior anatomic fit,2 and ease of use and usability of the system was rated as intuitive by surgeons in an internal survey.”3
In September 2022, Stryker rolled out the Gamma4 system, the latest generation of its 30-year legacy intramedullary (IM) nailing system for hip fractures. Gamma4 treats stable and unstable hip fractures as well as stabilizes bones and corrects bone deformities in the intracapsular, trochanteric, subtrochanteric, and shaft regions of the femur (including osteoporotic and osteopenic bone).
The system also features a Precision pin, which reduces potential for skiving. A redefined nail design features length-dependent RoC, shortened proximal body, and chamfered distal tip with pre-inserted set screw. The nail also has an integrated instrument platform.
“Gamma4 is designed to improve user experience and anatomical compliance for hip fracture treatment with a length dependent radius of curvature for long nails,4 additional implant sizes and nail features, such as the shortened proximal nail body and the beveled nail tip,” said Simon. “Gamma4 offers a more streamlined procedural workflow5 with a preloaded cannulated set screw, the precision pin for lag screw placement, and compatibility with the same core instrument tray as T2 Alpha.”
IM nails are among the many devices in the market to treat traumatic orthopedic injuries. In September this year, Stryker also received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Pangea line of variable-angle plating and instruments for the femur, fibula, tibia, humerus, and utility. The anatomically contoured implants were designed through the collaborative efforts of orthopedic surgeons and can address a variety of fracture patterns. The company also said the instrumentation and implant trays include 20 anatomic plates and 13 utility plates that are all accessible in one platform.
The Pangea systems were featured at this year’s Orthopaedic Trauma Association meeting.
“With these new systems, we are now offering surgeons a comprehensive portfolio that supports a wide range of their trauma needs,” Stryker Vice President and General Manager of Trauma Eric Tamweber told the press in a news release.
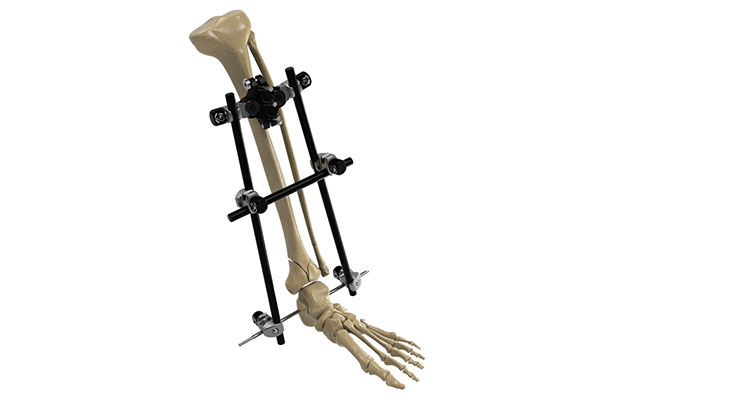
Other devices for surgeries to realign fractures and repair traumatic tears include cannulated, solid, and fenestrated screws, pins, rods, anchors, and external fixation devices. In external fixation, screws are placed into the bone above and below a bone fracture, and a device is attached to the screws from outside the skin to keep fractured bones stabilized and in alignment. It can be adjusted outside the body to ensure bones remain in an optimal position during the healing process. External fixators are commonly used in children and when the skin over the fracture has been damaged.
In late September, Orthofix commercially launched and completed the first U.S. cases with its newest stable external fixation system, Galaxy Fixation Gemini. The modular system treats fractures of both the upper and lower limbs and is provided in several sterile procedure kit configurations as a quick, off-the-shelf solution, according to the company.
“In trauma settings, it is critical that we respond quickly to manage the damage to the impacted limb,” Dr. Evengy Dyskin, clinical assistant professor of orthopedics at the University of Buffalo, said in a press release detailing the technology’s launch. “The all-inclusive nature of the sterile kits eliminates spatial constraints, enabling application in a trauma bay or intensive care unit setting, which would not be possible with a traditional pin-to-bar system.”
Orthofix touts the Galaxy Fixation Gemini ankle kit as the only pin-to-bar system with specific clamps available in a sterile kit configuration. The ankle kit includes a double multi-screw clamp the company claims facilitates quick tibial half-pin insertion. It’s also complemented by foot support and first metatarsal sterile kits, when a more robust construct is desired.
Smaller orthopedic companies are able to innovate within the trauma market as well.
In late September, Acuitive Technologies revealed it had won FDA clearance for its CITRELOCK DUO fixation device for bicep tears caused by injury or overuse. The tendon-friendly spiral thread is made from resorbable material, whose composition matches native bone’s inorganic content. The soft spiral thread was designed to prevent tendon laceration and its compressive strength mimics cortical bone, with a modulus the company said is similar to trabecular bone. The material’s elastomeric properties aim to improve pull-out strength as well.
CITRELOCK DUO’s controlled resorption rate seeks to mitigate post-operative chronic inflammation.
“Our customers are looking for a fixation device with unique mechanical properties that protects the tendon’s integrity during insertion while providing improved fixation within the bone tunnel. Additionally, the CITRELOCK DUO provides more predictable resorption while avoiding the chronic inflammation reported with other biodegradable polymers. With its innovative CITREGEN material technology and unique thread design, the CITRELOCK DUO fills the clinical need surgeons have been seeking,” Acuitive Vice President of Product Development Rui Ferreira told the press.
Manufacturing partners can also help orthopedic trauma surgery device makers navigate regulatory hurdles, ensure compliance with industry standards, and scale production up or down based on demand. In order to gather more information about the market forces impacting design and manufacturing of devices used in orthopedic trauma surgeries, ODT spoke to five experts in this field over the past few weeks.
Mike Couch: There is a trend toward using advanced materials, such as titanium alloys and biocompatible polymers, to create implants that are lightweight, durable, and biocompatible. Additive manufacturing techniques like 3D printing have gained popularity for creating customized implants and instruments as well. This allows for precise, patient-specific solutions.
Instruments and implants designed for minimally invasive procedures are also in high demand. These devices are typically smaller and require higher precision in manufacturing. Integration of digital technologies like augmented reality (AR) and virtual reality (VR) for surgical planning and training is becoming more common. This requires the development of specialized instruments and implants.
Manufacturers also need to stay updated with evolving regulatory requirements and ensure their products meet the necessary quality and safety standards.
Charles Hansford: Trauma, of all the orthopedic families, is the most limited regarding growth for additive manufacturing (AM) production. Plates and screws, although some are manufactured additively, are not cost effective for higher volume production. The greatest opportunities for growth that we have seen are for bone templates, some limited fixation screws (with porous lattice), patient specific implants, and instrument development. Craniomaxillofacial trauma/reconstruction and oncology patient-specific products are the largest growth areas in the trauma space for AM.
Brandon Miller: Trauma has a couple of trends we have noticed. One trend is the continued increase in the use of single-patient use disposable instruments. Many instrument kits are being made from new materials and are being placed in a convenient kit. Another trend is trauma companies are evaluating new implant manufacturing methods. Many companies are expressing interest in 3D printing. These porous structures assist with bone in-growth at the fracture site. Often, these printed devices are candidates for technologically advanced surfaces that make the implant more bioactive or durable.
Ben Thompson: We continue to see strong demand for our proprietary porous coating technology because of the clinical benefits of promoting bone ingrowth. The coating allows for an accurate, secure, and confident scratch-fit and a clinically proven ingrowth structure for long-term implant success.
Two trends in trauma include anatomically correct plating systems and 3D printing or additive manufacturing. 3D printing of patient-specific implants and implants with complex geometries are strengths for our additive manufacturing with over 10 years of experience at our Slice Manufacturing Studio site in Akron, Ohio, which is a pioneer in production metal 3D printing advanced processes with high standards. 3D printing enables complex device features that cannot be produced by traditional manufacturing methods.
Mike Treleaven: As an end-to-end contract manufacturer of medical devices, we help bring our customers’ products to market. We get involved early in the process with DFM (Design for Manufacturing), prototyping, and pilot production. We've had an increasing number of OEMs approach us needing our help to bring their robotic and computer-assisted devices to life for knee and hip replacement surgeries. The products we produce play a large part in helping surgeons perform knee and hip procedures more precisely and consistently than with manual techniques.
Brusco: How can a manufacturing partner benefit orthopedic trauma surgery device OEMs over those OEMs doing the work in-house?
Couch: Our engineers have specialized knowledge and experience in manufacturing orthopedic trauma devices. They can provide expertise in design, material selection, and production processes. Outsourcing to a CMO can also be cost-effective, as they often have established manufacturing processes, economies of scale, and access to a network of suppliers.
CMOs can expedite the production process, helping OEMs get their products to market more quickly. CMOs can also help OEMs navigate regulatory hurdles and ensure compliance with industry standards, reducing the risk of product recalls or regulatory issues.
CMOs can scale production up or down based on demand as well, providing OEMs with flexibility in managing their product portfolio.
Miller: We have in-house material science and product development teams. Often, OEM engineers have little to no experience designing 3D-printed implants. Our team, having designed many additive implants, knows the design changes necessary to maximize mechanical strength, which is needed for regulatory approval. Coupled with that, we have many surfaces that we develop and offer as a way for an OEM to offer new technologies to their customers, without the burdensome time and budgetary requirements to develop in-house. Simply put, working with a solution provider like us can shorten time to market while lowering R&D expenses and COGS.
Thompson: We have launched hundreds of new products, including reamers for femoral, tibial, humeral, ankle fusion, and clavicle nails as both an exclusive contract manufacturer and private label manufacturer of record. We have also built customer-centric processes and acquired new technologies to help our customers meet changing market needs. Our capabilities span concept to launch and include expertise in engineering, design and development, regulatory, quality, project management, instrument kitting, launch and inventory planning, integrated business planning, and supply chain management. For OEMs who are resource constrained, we have a highly skilled product development team to provide a rapid, turnkey solution to help OEMs bring new and differentiated products to market.
Brusco: Anything else to say regarding the orthopedic trauma surgery device market?
Treleaven: We’ve carved out unique capabilities for niche markets. For example, we’ve taken an innovative approach for manufacturing intramedullary nails. Believe it or not, intramedullary nails are complicated to manufacture and require multiple machining forming steps. It’s not a “one-size-fits-all” situation because when they are inserted into bones, they must have precise anatomical bends. Traditional methods of bending metal tend to damage or distort the holes and slots used for screws. We developed a proprietary method for creating a precise anatomical bend without damaging the nails’ special features.
References
In the late 19th century, ivory IM nails were introduced in Germany for femoral shaft fractures and became the material of choice for IM nails until 1910. This period also witnessed the development of interlocking nails and pins made from ivory to boost rotational stability. However, it later became apparent that ivory materials are resorbable—meaning ivory implants couldn’t offer sufficient stability to health fractures, particularly for cases involving delayed unions.
This led to experimentation with other metallic nails over the years: aluminum IM splints for femoral fractures, silver pins for upper limb fractures, and stainless steel or chromicized brass nails were used.
The modern use of stainless steel IM nails as the gold standard to treat femoral shaft fractures began with German surgeon Gerhard Küntscher during World War II. Then called the Küntscher nail, his first invention was a V-shaped, stainless steel IM nail. Küntscher published results of 39 cases in 1939 and, in 1947, another study with 107 war patients whose femoral and tibia shaft fractures were treated with IM nails. The nail’s use expanded in the United States after the first American soldiers arrived back from the war with German IM nails.
In the 1960s, titanium was introduced for IM nails after image intensifiers were invented. These nails boasted a Young Modulus closer to intact diaphyseal cortical bone, higher fatigue and yield tensile strength, and lower density compared to the stainless steel IM nails. Another turning point came about in 1986, when the non-slotted, femoral IM nail was made possible using a gun-drilling, computerized machining technique. This allowed construction of a nail with lower bending stiffness and better rotational stability. Non-slotted IM nails subsequently became the preferred technology up to the 21st century.
Three main novel IM nails have been identified in the last 20 years: expandable, coated, and carbon fiber nails.
Orthopedic device giant Stryker’s legacy T2 nailing platform is one of the mainstays of the company’s orthopedic trauma surgery technology portfolio. The T2 platform of IM nails are able to treat a wide range of fractures in the upper and lower extremities. The nails feature type II anodization for better fatigue strength, active internal compression, and a common instrumentation platform. They also tout radiolucent targeting arms with easy assembly and friction lock technology, Stryker describes.
The latest in this line, the T2 Alpha femur retrograde nailing system, hit the market in August 2021. According to Stryker, it features a redefined, data-driven nail design and dedicated proximal targeting system that could reduce the number of X-ray shots as compared to freehand locking.
“T2 Alpha Femur Retrograde is based on decades of experience with the Stryker T2 system,” Bernd Simon, senior principal engineer of R&D at Stryker, told ODT. “It is designed to provide an intraoperative adaptability with more fixation options in the nail, internal and external compression and the option to use Advanced Locking Screws for added axial and angular stability.1 The length-dependent curvature of the nail shows superior anatomic fit,2 and ease of use and usability of the system was rated as intuitive by surgeons in an internal survey.”3
In September 2022, Stryker rolled out the Gamma4 system, the latest generation of its 30-year legacy intramedullary (IM) nailing system for hip fractures. Gamma4 treats stable and unstable hip fractures as well as stabilizes bones and corrects bone deformities in the intracapsular, trochanteric, subtrochanteric, and shaft regions of the femur (including osteoporotic and osteopenic bone).
The system also features a Precision pin, which reduces potential for skiving. A redefined nail design features length-dependent RoC, shortened proximal body, and chamfered distal tip with pre-inserted set screw. The nail also has an integrated instrument platform.
“Gamma4 is designed to improve user experience and anatomical compliance for hip fracture treatment with a length dependent radius of curvature for long nails,4 additional implant sizes and nail features, such as the shortened proximal nail body and the beveled nail tip,” said Simon. “Gamma4 offers a more streamlined procedural workflow5 with a preloaded cannulated set screw, the precision pin for lag screw placement, and compatibility with the same core instrument tray as T2 Alpha.”
IM nails are among the many devices in the market to treat traumatic orthopedic injuries. In September this year, Stryker also received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Pangea line of variable-angle plating and instruments for the femur, fibula, tibia, humerus, and utility. The anatomically contoured implants were designed through the collaborative efforts of orthopedic surgeons and can address a variety of fracture patterns. The company also said the instrumentation and implant trays include 20 anatomic plates and 13 utility plates that are all accessible in one platform.
The Pangea systems were featured at this year’s Orthopaedic Trauma Association meeting.
“With these new systems, we are now offering surgeons a comprehensive portfolio that supports a wide range of their trauma needs,” Stryker Vice President and General Manager of Trauma Eric Tamweber told the press in a news release.
Other devices for surgeries to realign fractures and repair traumatic tears include cannulated, solid, and fenestrated screws, pins, rods, anchors, and external fixation devices. In external fixation, screws are placed into the bone above and below a bone fracture, and a device is attached to the screws from outside the skin to keep fractured bones stabilized and in alignment. It can be adjusted outside the body to ensure bones remain in an optimal position during the healing process. External fixators are commonly used in children and when the skin over the fracture has been damaged.
In late September, Orthofix commercially launched and completed the first U.S. cases with its newest stable external fixation system, Galaxy Fixation Gemini. The modular system treats fractures of both the upper and lower limbs and is provided in several sterile procedure kit configurations as a quick, off-the-shelf solution, according to the company.
“In trauma settings, it is critical that we respond quickly to manage the damage to the impacted limb,” Dr. Evengy Dyskin, clinical assistant professor of orthopedics at the University of Buffalo, said in a press release detailing the technology’s launch. “The all-inclusive nature of the sterile kits eliminates spatial constraints, enabling application in a trauma bay or intensive care unit setting, which would not be possible with a traditional pin-to-bar system.”
Orthofix touts the Galaxy Fixation Gemini ankle kit as the only pin-to-bar system with specific clamps available in a sterile kit configuration. The ankle kit includes a double multi-screw clamp the company claims facilitates quick tibial half-pin insertion. It’s also complemented by foot support and first metatarsal sterile kits, when a more robust construct is desired.
Smaller orthopedic companies are able to innovate within the trauma market as well.
In late September, Acuitive Technologies revealed it had won FDA clearance for its CITRELOCK DUO fixation device for bicep tears caused by injury or overuse. The tendon-friendly spiral thread is made from resorbable material, whose composition matches native bone’s inorganic content. The soft spiral thread was designed to prevent tendon laceration and its compressive strength mimics cortical bone, with a modulus the company said is similar to trabecular bone. The material’s elastomeric properties aim to improve pull-out strength as well.
CITRELOCK DUO’s controlled resorption rate seeks to mitigate post-operative chronic inflammation.
“Our customers are looking for a fixation device with unique mechanical properties that protects the tendon’s integrity during insertion while providing improved fixation within the bone tunnel. Additionally, the CITRELOCK DUO provides more predictable resorption while avoiding the chronic inflammation reported with other biodegradable polymers. With its innovative CITREGEN material technology and unique thread design, the CITRELOCK DUO fills the clinical need surgeons have been seeking,” Acuitive Vice President of Product Development Rui Ferreira told the press.
Injury Innovations
Due to the speed of orthopedic device innovation and need to get devices to market as speedily as possible, orthopedic trauma surgery device OEMs might enlist a partner with specialized knowledge of manufacturing those devices. They can offer needed expertise in design, material selection, and production processes. Outsourcing some work to a manufacturing partner can also be cost-effective because they already possess established manufacturing processes, economies of scale, and access to a network of suppliers.Manufacturing partners can also help orthopedic trauma surgery device makers navigate regulatory hurdles, ensure compliance with industry standards, and scale production up or down based on demand. In order to gather more information about the market forces impacting design and manufacturing of devices used in orthopedic trauma surgeries, ODT spoke to five experts in this field over the past few weeks.
- Mike Couch, business development manager at Precision Medical Technologies.
- Charles Hansford, director of advanced development at Tecomet.
- Brandon Miller, director of commercial product development at Lincotek Medical.
- Ben Thompson, chief commercial officer at Avalign Technologies.
- Mike Treleaven, senior vice president of engineering at Tegra Medical.
Mike Couch: There is a trend toward using advanced materials, such as titanium alloys and biocompatible polymers, to create implants that are lightweight, durable, and biocompatible. Additive manufacturing techniques like 3D printing have gained popularity for creating customized implants and instruments as well. This allows for precise, patient-specific solutions.
Instruments and implants designed for minimally invasive procedures are also in high demand. These devices are typically smaller and require higher precision in manufacturing. Integration of digital technologies like augmented reality (AR) and virtual reality (VR) for surgical planning and training is becoming more common. This requires the development of specialized instruments and implants.
Manufacturers also need to stay updated with evolving regulatory requirements and ensure their products meet the necessary quality and safety standards.
Charles Hansford: Trauma, of all the orthopedic families, is the most limited regarding growth for additive manufacturing (AM) production. Plates and screws, although some are manufactured additively, are not cost effective for higher volume production. The greatest opportunities for growth that we have seen are for bone templates, some limited fixation screws (with porous lattice), patient specific implants, and instrument development. Craniomaxillofacial trauma/reconstruction and oncology patient-specific products are the largest growth areas in the trauma space for AM.
Brandon Miller: Trauma has a couple of trends we have noticed. One trend is the continued increase in the use of single-patient use disposable instruments. Many instrument kits are being made from new materials and are being placed in a convenient kit. Another trend is trauma companies are evaluating new implant manufacturing methods. Many companies are expressing interest in 3D printing. These porous structures assist with bone in-growth at the fracture site. Often, these printed devices are candidates for technologically advanced surfaces that make the implant more bioactive or durable.
Ben Thompson: We continue to see strong demand for our proprietary porous coating technology because of the clinical benefits of promoting bone ingrowth. The coating allows for an accurate, secure, and confident scratch-fit and a clinically proven ingrowth structure for long-term implant success.
Two trends in trauma include anatomically correct plating systems and 3D printing or additive manufacturing. 3D printing of patient-specific implants and implants with complex geometries are strengths for our additive manufacturing with over 10 years of experience at our Slice Manufacturing Studio site in Akron, Ohio, which is a pioneer in production metal 3D printing advanced processes with high standards. 3D printing enables complex device features that cannot be produced by traditional manufacturing methods.
Mike Treleaven: As an end-to-end contract manufacturer of medical devices, we help bring our customers’ products to market. We get involved early in the process with DFM (Design for Manufacturing), prototyping, and pilot production. We've had an increasing number of OEMs approach us needing our help to bring their robotic and computer-assisted devices to life for knee and hip replacement surgeries. The products we produce play a large part in helping surgeons perform knee and hip procedures more precisely and consistently than with manual techniques.
Brusco: How can a manufacturing partner benefit orthopedic trauma surgery device OEMs over those OEMs doing the work in-house?
Couch: Our engineers have specialized knowledge and experience in manufacturing orthopedic trauma devices. They can provide expertise in design, material selection, and production processes. Outsourcing to a CMO can also be cost-effective, as they often have established manufacturing processes, economies of scale, and access to a network of suppliers.
CMOs can expedite the production process, helping OEMs get their products to market more quickly. CMOs can also help OEMs navigate regulatory hurdles and ensure compliance with industry standards, reducing the risk of product recalls or regulatory issues.
CMOs can scale production up or down based on demand as well, providing OEMs with flexibility in managing their product portfolio.
Miller: We have in-house material science and product development teams. Often, OEM engineers have little to no experience designing 3D-printed implants. Our team, having designed many additive implants, knows the design changes necessary to maximize mechanical strength, which is needed for regulatory approval. Coupled with that, we have many surfaces that we develop and offer as a way for an OEM to offer new technologies to their customers, without the burdensome time and budgetary requirements to develop in-house. Simply put, working with a solution provider like us can shorten time to market while lowering R&D expenses and COGS.
Thompson: We have launched hundreds of new products, including reamers for femoral, tibial, humeral, ankle fusion, and clavicle nails as both an exclusive contract manufacturer and private label manufacturer of record. We have also built customer-centric processes and acquired new technologies to help our customers meet changing market needs. Our capabilities span concept to launch and include expertise in engineering, design and development, regulatory, quality, project management, instrument kitting, launch and inventory planning, integrated business planning, and supply chain management. For OEMs who are resource constrained, we have a highly skilled product development team to provide a rapid, turnkey solution to help OEMs bring new and differentiated products to market.
Brusco: Anything else to say regarding the orthopedic trauma surgery device market?
Treleaven: We’ve carved out unique capabilities for niche markets. For example, we’ve taken an innovative approach for manufacturing intramedullary nails. Believe it or not, intramedullary nails are complicated to manufacture and require multiple machining forming steps. It’s not a “one-size-fits-all” situation because when they are inserted into bones, they must have precise anatomical bends. Traditional methods of bending metal tend to damage or distort the holes and slots used for screws. We developed a proprietary method for creating a precise anatomical bend without damaging the nails’ special features.
References
- White Paper, Fatigue Strength of Locking Screws, T2-WP-6, Rev 1
- Internal Report № A0046864. Rev 1, Schönkirchen, Germany
- Stryker White Paper, Survey data on first clinical experiences with the T2 Alpha Femur Retrograde Nailing System, T2-WP-17, 11-2021
- Internal Report № D0000089997, Rev AA, Schönkirchen, Germany
- White Paper, Survey data on first clinical experiences with the Gamma4 Hip Fracture Nailing System, G4-WP-1, 09-2022