08.12.22
Breaking from tradition can be a challenging road to travel. Within healthcare, the risk can be even greater as a novel product may face an uphill climb in its journey towards successful market adoption. As such, the ODT Editors have spotlighted six companies outside the Top 10 they feel are making an impact on the device sector with more unique and newsworthy technologies.
Michael Barbella • Managing Editor
Jeff Karp owes his research career to Spider-Man.
Yes, that Spider-Man—the web-slinging, comic book superhero who assumes inhuman, arachnid-like abilities after being bitten by a radioactive spider.
Stan Lee would be proud.
Spidey caught Karp’s eye from the pages of a medical journal, where he accompanied an article describing a waterproof dry adhesive inspired by the Tokay gecko’s sticky feet (and Spider-Man’s own sticky limbs). Reading the article inspired Karp to develop an adhesive surgical patch to replace the sutures and staples used in wound closure. He pitched the idea to the National Science Foundation, and along with a global research team, created a waterproof adhesive bandage made of “biorubber.”
For the bandage’s second-generation version, Karp and his team turned to slugs and snails for motivation, eventually developing an adhesive that infiltrates tissue upon contact and cures when exposed to light (an instant bond). The new glue reportedly works throughout the body, including inside a beating heart—one of the roughest environments.
Karp’s work in developing the adhesive (both models) served as an initiation of sorts into bioinspiration, a small but growing scientific discipline that uses biological systems/processes to solve technological and medical challenges. Not to be confused with biomimicry, which aims to precisely replicate organic functions, bioinspiration merely uses Mother Nature as a muse.
“Bioinspiration is not imitation. The basic goal is to take an idea in nature and improve upon it for your own purposes,” Karp, Ph.D., a professor of Medicine at Brigham and Women’s Hospital, Harvard Medical School, said in an American Museum of Natural History online post. Karp is also a principal faculty member at the Harvard Stem Cell Institute, and an affiliate faculty member at the Broad Institute and at the Harvard-MIT Division of Health Sciences and Technology. “...every living thing—every plant, every animal, every creature that exists today—is here because it has solved an incredible number of challenges and evolved to tell the tale. Evolution is the best problem-solver.”
Better than humans, actually. Homo sapiens have spent several millennia trying to improve upon nature, with only limited success. One of the more challenging areas for bioinspirationalists has been bone regeneration due to the tissue’s complex heirarchical structure, dense vascularization, and the considerable shortcomings, limitations, and complications of current bone repair treatments.
Nevertheless, scientists and orthopedic companies alike continue to work at decrypting the natural code of bone regeneration. They’ve turned to natural polymers like collagen, silk, fibrin gels, and soy; palm plants (rattan); and turmeric to make new bone. And that’s just the tip of the proverbial iceberg.
Coral also is an ideal bone regenerative tool, as it does not evoke an inflammatory response and—like real bone—does not form scar tissue. It also boasts excellent osteoconduction and resorption properties.
Israeli firm CoreBone, founded 11 years ago by CEO Ohad Schwartz and Prof. Itzhak Binederman, manufactures grafts made from actual coral grown in closed, controlled aquarium systems that prevent the tiny, soft-bodied organisms from succumbing to ocean pollution. CoreBone’s corals are bred from selected species suitable for particular clinical indications for optimal strength and porosity.
The company’s proprietary technology embeds specific nutrients such as silicium and strontium into the pure coral mineral (largely calcium carbonate) during its growth process, enabling it to become bioactive and osteoconductive. The interconnected porosity of CoreBone coral allows 3D bone generation that has been shown to lead to higher fusion rates, without loss of strength. In fact, CoreBone’s coral-derived grafts are up to five times stronger than cancellous bone or synthetics, according to data on the firm’s website. Moreover, the coral’s bioactive surface, interconnected canals, and pores present for optimal net and dimensions for developing blood vessels and depositing bone.
A portfolio company of The Trendlines Group, CoreBone markets four products for dental and maxillofacial procedures: one for filling narrow bone gaps and between implants; another for filling wide bone gaps and voids, mostly in sinus lift procedures; a third for large bone defects and sinus lift procedures; and CoreBlock, for augmenting facial bone defects and reconstructive surgery.
The CE-marked products are used mainly in Europe and Israel.
Such was the case with GreenBone Ortho SpA, an 8-year-old Italian company developing rattan-based bone replacement. Rattan checks all the boxes for a “miracle” bone replacement material: It’s rigid, yet flexible enough to move with the body, and comprises an internal structure that nurtures nerve and blood vessel growth. It also fuses naturally with surrounding bone when implanted and eventually transforms into real living tissue.
Rattan’s promising bone-like properties are supported by a decade of research by scientists at the Institute for Science and Technology for Ceramics of the Italian National Research Council (ISTEC-CNR). ISTEC-CNR Director Dr. Anna Tamperi is credited with creating the chemical process for turning rattan wood into biomimetic and regenerative implants.
To make the transformation, GreenBone first removes all plant materials from the rattan. Subsequent steps—named biomorphic transformation—transform the wood into a mixture of hydroxyapatite (HA) and beta-tricalcium phosphate (TCP), preserving its porous architecture and producing a structure similar to that found in normal, healthy bone. This porosity acts like Haversian canals throughout the whole graft, allowing vascularization and the necessary exchange of biological fluids to support bone regeneration.
The biomorphic transformation process to create b.Bone achieves its final chemical composition of HA and PCP without the need for high temperature sintering as is required for other ceramic implants. As a result, b.Bone possesses a nanocrystalline structure that supports increased mineralization of the regenerated new bone. The process has been extensively patented around the world.
b.Bone is available in various shapes and sizes—cylinder, block, wedge, and granules to match clinical applications including traumatology, bone augmentation, joint augmentation, deformity correction, lengthening, non-union, bone loss, osteotomy, and joint revision.
To support expected demand for its products, GreenBone opened a manufacturing facility in Northern Italy last year and closed a €10 million funding round. Earlier this year, the firm signed an Italian distribution agreement with Abiogen Pharma SpA for its b.Bone scaffold.
“We are very excited about this agreement with Abiogen,” said Diogo C. Moreira-Rato, non-executive board member and former J&J senior executive. “We are confident this agreement will lead not only to successful commercial results for b.Bone in the Italian market but will also be a driving force for the development of other European markets where GreenBone is focusing its efforts.”
Sean Fenske • Editor-in-Chief
While the orthopedic industry has traditionally been one of the slowest in terms of embracing new technologies, recent years have seen a rapid emergence of new products that run counter to the established methods to which many practitioners may be accustomed. New offerings have sparked a movement toward an interest in more natural healing options, safer and less invasive surgical procedures, and an acceptance of alternative materials. As such, orthopedic device manufacturers are putting forth robotic surgical systems, innovations powered by artificial intelligence and data, and biologic substitutes that leverage the body’s natural healing capabilities to achieve results. The following products reflect this trend as they present a different type of option to meet the aforementioned goals.
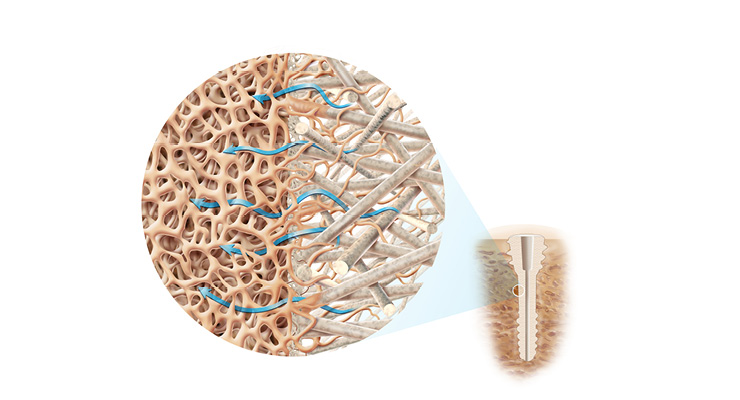
According to the company, its OSSIOfiber is an “intelligent bone regeneration technology” that eliminates the need for metal. The material is stronger than cortical bone and also avoids the challenges associated with permanent metal implants such as foreign body reactions and potential patient discomfort. It is also stronger and more predictable than polymers when they are used alone.
The make-up of OSSIOfiber involves minerals much like those in bone (silica, magnesium, calcium, and others), which are used to form a unique matrix of fibers. Poly-L/D-lactic acid is used to bind the fiber matrix together.The technology enables “early bone attachment, gradual bone and tissue ingrowth, and complete integration, all without local or systemic adverse response,” according to the firm’s website.
The innovation has been made available in a number of form factors. As a Trimmable Fixation Nail, it can be used in a variety of applications within the foot, ankle, hand, and wrist. The Hammertoe Fixation Implant is available in three sizes and two orientations. When the Compression Screws integrate completely into the patient’s bone, they leave behind natural bone in its place versus a permanent “alien” artifact.
More recently, the company has gained additional FDA clearances for applications since December 2021. It can be used for suture anchors for procedures involving the shoulder, foot, ankle, knee, hand, wrist, and elbow. It is also now available as compression staples for use in “fixation of arthrodesis, osteotomies, and fractures in hand or foot surgery in the presence of appropriate brace and/or immobilization.”
“With more than 6,000 implantations of OSSIOfiber products performed to date, we have already achieved considerable momentum and market penetration in the United States, demonstrating meaningful adoption of our novel, innovative, and proprietary orthopedic fixation technology,” said OSSIO’s CEO, Brian Verrier.
Unfortunately, it isn’t a perfect system and does come with a few downsides. One is the lack of feeling of natural anatomy when performing a procedure. Getting a visualization of the human body does not present the same experience as cutting into a cadaver or actual patient, feeling the tissue and bone beneath an instrument.
FundamentalVR sought to change that with its unique solution. Coupling a headset with a pair of hand controls makes this company’s offering look very similar to other firms marketing a VR technology. What makes Fundamental’s offering novel, however, is the haptic feedback that’s provided to the operator through the handpieces. The “sense of touch” that’s enabled is a remarkable differentiator between this product and others. Further, bone feels different from muscle, which feels different from other tissue.
“By combining immersive VR with cutting-edge haptics and AI at scale, we can now create an incredibly life-like training experience with accurate physical sensations. This is a game-changer. We are laser focused on pre-human competence, which is about training surgeons in the most realistic way possible before they enter any operating theater,” explained Richard Vincent, CEO of FundamentalVR, in a Future of Healthcare report published in the UK’s The Times. “Our Fundamental Surgery platform is the first in the world to combine VR, haptics, deep data, AI, and multi-modal learning. We are also helping life science companies accelerate the safe and compliant introduction of medical innovations.”
While the technology can be used for a variety of healthcare segments, within orthopedics it already has capabilities for knee, hip, and spine surgeries. The company also states its Fundamental Surgery product is the only fully accredited (AAOS and Royal College of Surgeons, England) VR education platform. The system is available in several form factors—HapticVR, which provides the immersive virtual experience including the sense of touch, and the @HomeVR, which enables the technology to be used “on the go.”
Sam Brusco • Associate Editor
ODT is headquartered in New Jersey, which readers may not know is a surprisingly rich medical technology hub.
A large manufacturing facility for Top 10 leader Stryker in Mahwah is a mere 15-minute drive from the ODT office. Drive another ten minutes and Stryker’s Spine division will enter the view in Allendale. Half an hour away is the Franklin Lakes headquarters for medtech giant Becton, Dickinson and Company (BD). (CR Bard, which BD acquired a few years ago, had its home in Murray Hill.)
And that’s not all. Former Top 10 denizen Integra LifeSciences is situated in Princeton. Neurostimulation firm electroCore resides in Rockaway. Perhaps most noteworthy, the headquarters of multi-billion-dollar conglomerate and MPO Top 30 bronze medalist Johnson & Johnson towers over New Brunswick. Several other medtech firms have facilities in the Garden State, but there are far too many to list given this article’s space constraints.
The two unique orthopedic firms highlighted in my portion of this feature happen to live in the same town, less than an hour away from the ODT office. Who’s up for a couple facility tours?
“Onkos Surgical is 100% focused on bringing new technology and solutions to surgical oncologists,” he said when announcing the company’s establishment. “These specialists serve a population where a large portion of patients are children and the broader population may require personalized solutions. Surgeons, patients, and families deserve a focused company dedicated to this important cause.”
Its first FDA clearance came in 2016 with the ELEOS modular limb salvage system to treat significant bone loss due to cancer, trauma, or previous surgical procedures. The system leverages 3D printing capabilities to be a long-term reconstruction option for radical resection and replacement of the distal femur, proximal femur, proximal tibia, or total femur.
In addition to ELEOS, the company’s portfolio includes uDesign for surgical planning; MyTray personalized instrument trays; the GenVie biological adjuvant; and My3D personalized implant, instrument, and anatomic model designs.
The company made a couple large strides this year. In June, Onkos began the acquisition of Stryker’s Juvenile Tumor System (JTS)—an extendible prosthesis that promotes limb stabilization and growth for children affected by bone cancers, severe arthropathy, and severe trauma, revisions, oncology, and malignant diseases. The JTS helps non-invasively lengthen the implant post-surgery in a clinical setting. The company also received 510(k) clearance for its My3D personalized pelvic reconstruction system in July. The system includes 3D-printed implants, instruments, models, and an advanced planning tool for deformity, trauma, disease, and revisions where other treatments or revisions have failed.
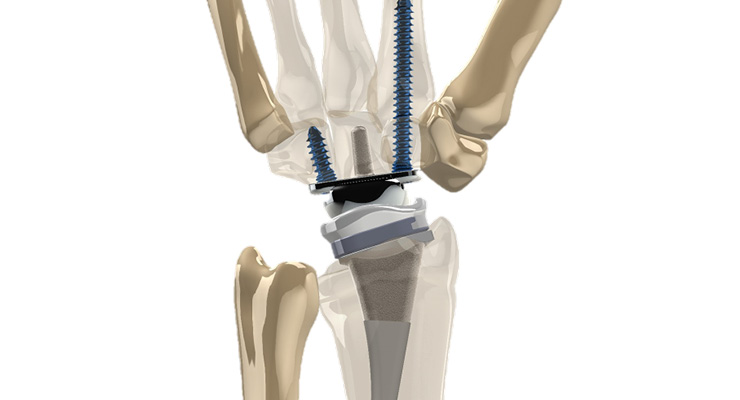
The company’s first product, the IO FiX MCP intraosseous fixation system, gained FDA clearance and CE mark approval in 2010. It enabled arthrodesis of the metacarpal phalangeal joints in the hand, the first such technology to do so at the time. Fusion can be achieved with a stronger construct and greater compression than traditional methods, using a fixed angle and avoiding irritation from plates, screws, and wires.
The company’s portfolio also includes UpperFiX and KinematX for upper extremities. For lower extremities, the franchise is comprised of the AlignX ankle fusion system, Axis Charcot fixation system, CompressX2 compression screws, EXPRESS staple system, HammerFiX, Jones union system, OMNI plating system, and OMNI ankle fracture system.
In April and May, the company initiated patent lawsuits against Zimmer Biomet, Nextremity Solutions, and Fusion Orthopedics. Extremity Medical alleges all three companies infringed on patents for its intraosseous fixation platform, for which the company has 49 in the U.S.
“Extremity Medical invests a great deal into building and defending its intellectual property portfolio,” said CEO Matthew Lyons when commenting on the Fusion Orthopedics lawsuit. “We will not tolerate competitors who blatantly copy our products while infringing on our patent rights. We intend to seek all remedies allowed by law.”
In happier news, the firm rolled out its OMNI stable ankle fracture system in May as well, expanding its purview into the trauma market. The system’s variety of low-profile anatomic plates closely match patient anatomy and reduce impingement of soft tissue. A proprietary Stability Post optimizes stability for patients with poor bone quality and/or segmental loss of bone.
Michael Barbella • Managing Editor
Jeff Karp owes his research career to Spider-Man.
Yes, that Spider-Man—the web-slinging, comic book superhero who assumes inhuman, arachnid-like abilities after being bitten by a radioactive spider.
Stan Lee would be proud.
Spidey caught Karp’s eye from the pages of a medical journal, where he accompanied an article describing a waterproof dry adhesive inspired by the Tokay gecko’s sticky feet (and Spider-Man’s own sticky limbs). Reading the article inspired Karp to develop an adhesive surgical patch to replace the sutures and staples used in wound closure. He pitched the idea to the National Science Foundation, and along with a global research team, created a waterproof adhesive bandage made of “biorubber.”
For the bandage’s second-generation version, Karp and his team turned to slugs and snails for motivation, eventually developing an adhesive that infiltrates tissue upon contact and cures when exposed to light (an instant bond). The new glue reportedly works throughout the body, including inside a beating heart—one of the roughest environments.
Karp’s work in developing the adhesive (both models) served as an initiation of sorts into bioinspiration, a small but growing scientific discipline that uses biological systems/processes to solve technological and medical challenges. Not to be confused with biomimicry, which aims to precisely replicate organic functions, bioinspiration merely uses Mother Nature as a muse.
“Bioinspiration is not imitation. The basic goal is to take an idea in nature and improve upon it for your own purposes,” Karp, Ph.D., a professor of Medicine at Brigham and Women’s Hospital, Harvard Medical School, said in an American Museum of Natural History online post. Karp is also a principal faculty member at the Harvard Stem Cell Institute, and an affiliate faculty member at the Broad Institute and at the Harvard-MIT Division of Health Sciences and Technology. “...every living thing—every plant, every animal, every creature that exists today—is here because it has solved an incredible number of challenges and evolved to tell the tale. Evolution is the best problem-solver.”
Better than humans, actually. Homo sapiens have spent several millennia trying to improve upon nature, with only limited success. One of the more challenging areas for bioinspirationalists has been bone regeneration due to the tissue’s complex heirarchical structure, dense vascularization, and the considerable shortcomings, limitations, and complications of current bone repair treatments.
Nevertheless, scientists and orthopedic companies alike continue to work at decrypting the natural code of bone regeneration. They’ve turned to natural polymers like collagen, silk, fibrin gels, and soy; palm plants (rattan); and turmeric to make new bone. And that’s just the tip of the proverbial iceberg.
Secrets from the Sea
Coral reefs are the most diverse of all marine ecosystems. They teem with life, providing food and shelter to perhaps one-quarter of all ocean species.Coral also is an ideal bone regenerative tool, as it does not evoke an inflammatory response and—like real bone—does not form scar tissue. It also boasts excellent osteoconduction and resorption properties.
Israeli firm CoreBone, founded 11 years ago by CEO Ohad Schwartz and Prof. Itzhak Binederman, manufactures grafts made from actual coral grown in closed, controlled aquarium systems that prevent the tiny, soft-bodied organisms from succumbing to ocean pollution. CoreBone’s corals are bred from selected species suitable for particular clinical indications for optimal strength and porosity.
The company’s proprietary technology embeds specific nutrients such as silicium and strontium into the pure coral mineral (largely calcium carbonate) during its growth process, enabling it to become bioactive and osteoconductive. The interconnected porosity of CoreBone coral allows 3D bone generation that has been shown to lead to higher fusion rates, without loss of strength. In fact, CoreBone’s coral-derived grafts are up to five times stronger than cancellous bone or synthetics, according to data on the firm’s website. Moreover, the coral’s bioactive surface, interconnected canals, and pores present for optimal net and dimensions for developing blood vessels and depositing bone.
A portfolio company of The Trendlines Group, CoreBone markets four products for dental and maxillofacial procedures: one for filling narrow bone gaps and between implants; another for filling wide bone gaps and voids, mostly in sinus lift procedures; a third for large bone defects and sinus lift procedures; and CoreBlock, for augmenting facial bone defects and reconstructive surgery.
The CE-marked products are used mainly in Europe and Israel.
Into the Woods
Sometimes, the easiest answers are the most difficult to find. Especially when they hide in plain sight.Such was the case with GreenBone Ortho SpA, an 8-year-old Italian company developing rattan-based bone replacement. Rattan checks all the boxes for a “miracle” bone replacement material: It’s rigid, yet flexible enough to move with the body, and comprises an internal structure that nurtures nerve and blood vessel growth. It also fuses naturally with surrounding bone when implanted and eventually transforms into real living tissue.
Rattan’s promising bone-like properties are supported by a decade of research by scientists at the Institute for Science and Technology for Ceramics of the Italian National Research Council (ISTEC-CNR). ISTEC-CNR Director Dr. Anna Tamperi is credited with creating the chemical process for turning rattan wood into biomimetic and regenerative implants.
To make the transformation, GreenBone first removes all plant materials from the rattan. Subsequent steps—named biomorphic transformation—transform the wood into a mixture of hydroxyapatite (HA) and beta-tricalcium phosphate (TCP), preserving its porous architecture and producing a structure similar to that found in normal, healthy bone. This porosity acts like Haversian canals throughout the whole graft, allowing vascularization and the necessary exchange of biological fluids to support bone regeneration.
The biomorphic transformation process to create b.Bone achieves its final chemical composition of HA and PCP without the need for high temperature sintering as is required for other ceramic implants. As a result, b.Bone possesses a nanocrystalline structure that supports increased mineralization of the regenerated new bone. The process has been extensively patented around the world.
b.Bone is available in various shapes and sizes—cylinder, block, wedge, and granules to match clinical applications including traumatology, bone augmentation, joint augmentation, deformity correction, lengthening, non-union, bone loss, osteotomy, and joint revision.
To support expected demand for its products, GreenBone opened a manufacturing facility in Northern Italy last year and closed a €10 million funding round. Earlier this year, the firm signed an Italian distribution agreement with Abiogen Pharma SpA for its b.Bone scaffold.
“We are very excited about this agreement with Abiogen,” said Diogo C. Moreira-Rato, non-executive board member and former J&J senior executive. “We are confident this agreement will lead not only to successful commercial results for b.Bone in the Italian market but will also be a driving force for the development of other European markets where GreenBone is focusing its efforts.”
Sean Fenske • Editor-in-Chief
While the orthopedic industry has traditionally been one of the slowest in terms of embracing new technologies, recent years have seen a rapid emergence of new products that run counter to the established methods to which many practitioners may be accustomed. New offerings have sparked a movement toward an interest in more natural healing options, safer and less invasive surgical procedures, and an acceptance of alternative materials. As such, orthopedic device manufacturers are putting forth robotic surgical systems, innovations powered by artificial intelligence and data, and biologic substitutes that leverage the body’s natural healing capabilities to achieve results. The following products reflect this trend as they present a different type of option to meet the aforementioned goals.
Now You See It, Now You Don’t
Metal implants have been the treatment modality of choice within orthopedics for quite some time. Many, however, are embracing solutions that offer a more natural healing alternative than the permanent devices left in the body to support or repair bone and tissue. Those at OSSIO are among those embracing the quest to achieve this alternative to using more traditional options.According to the company, its OSSIOfiber is an “intelligent bone regeneration technology” that eliminates the need for metal. The material is stronger than cortical bone and also avoids the challenges associated with permanent metal implants such as foreign body reactions and potential patient discomfort. It is also stronger and more predictable than polymers when they are used alone.
The make-up of OSSIOfiber involves minerals much like those in bone (silica, magnesium, calcium, and others), which are used to form a unique matrix of fibers. Poly-L/D-lactic acid is used to bind the fiber matrix together.The technology enables “early bone attachment, gradual bone and tissue ingrowth, and complete integration, all without local or systemic adverse response,” according to the firm’s website.
The innovation has been made available in a number of form factors. As a Trimmable Fixation Nail, it can be used in a variety of applications within the foot, ankle, hand, and wrist. The Hammertoe Fixation Implant is available in three sizes and two orientations. When the Compression Screws integrate completely into the patient’s bone, they leave behind natural bone in its place versus a permanent “alien” artifact.
More recently, the company has gained additional FDA clearances for applications since December 2021. It can be used for suture anchors for procedures involving the shoulder, foot, ankle, knee, hand, wrist, and elbow. It is also now available as compression staples for use in “fixation of arthrodesis, osteotomies, and fractures in hand or foot surgery in the presence of appropriate brace and/or immobilization.”
“With more than 6,000 implantations of OSSIOfiber products performed to date, we have already achieved considerable momentum and market penetration in the United States, demonstrating meaningful adoption of our novel, innovative, and proprietary orthopedic fixation technology,” said OSSIO’s CEO, Brian Verrier.
A Sense of Touch
Virtual reality (VR) offers a broad array of advantages within the orthopedic space. It enables clinicians to practice a procedure on a simulated patient. It allows a device representative to demonstrate a new surgical instrument with a doctor even if they can’t be together in the same location. It permits an educator to share expertise with a larger number of students, giving everyone a front-row view. It also provides a platform for a surgeon to interface with a product development team to share feedback on a design.Unfortunately, it isn’t a perfect system and does come with a few downsides. One is the lack of feeling of natural anatomy when performing a procedure. Getting a visualization of the human body does not present the same experience as cutting into a cadaver or actual patient, feeling the tissue and bone beneath an instrument.
FundamentalVR sought to change that with its unique solution. Coupling a headset with a pair of hand controls makes this company’s offering look very similar to other firms marketing a VR technology. What makes Fundamental’s offering novel, however, is the haptic feedback that’s provided to the operator through the handpieces. The “sense of touch” that’s enabled is a remarkable differentiator between this product and others. Further, bone feels different from muscle, which feels different from other tissue.
“By combining immersive VR with cutting-edge haptics and AI at scale, we can now create an incredibly life-like training experience with accurate physical sensations. This is a game-changer. We are laser focused on pre-human competence, which is about training surgeons in the most realistic way possible before they enter any operating theater,” explained Richard Vincent, CEO of FundamentalVR, in a Future of Healthcare report published in the UK’s The Times. “Our Fundamental Surgery platform is the first in the world to combine VR, haptics, deep data, AI, and multi-modal learning. We are also helping life science companies accelerate the safe and compliant introduction of medical innovations.”
While the technology can be used for a variety of healthcare segments, within orthopedics it already has capabilities for knee, hip, and spine surgeries. The company also states its Fundamental Surgery product is the only fully accredited (AAOS and Royal College of Surgeons, England) VR education platform. The system is available in several form factors—HapticVR, which provides the immersive virtual experience including the sense of touch, and the @HomeVR, which enables the technology to be used “on the go.”
Sam Brusco • Associate Editor
ODT is headquartered in New Jersey, which readers may not know is a surprisingly rich medical technology hub.
A large manufacturing facility for Top 10 leader Stryker in Mahwah is a mere 15-minute drive from the ODT office. Drive another ten minutes and Stryker’s Spine division will enter the view in Allendale. Half an hour away is the Franklin Lakes headquarters for medtech giant Becton, Dickinson and Company (BD). (CR Bard, which BD acquired a few years ago, had its home in Murray Hill.)
And that’s not all. Former Top 10 denizen Integra LifeSciences is situated in Princeton. Neurostimulation firm electroCore resides in Rockaway. Perhaps most noteworthy, the headquarters of multi-billion-dollar conglomerate and MPO Top 30 bronze medalist Johnson & Johnson towers over New Brunswick. Several other medtech firms have facilities in the Garden State, but there are far too many to list given this article’s space constraints.
The two unique orthopedic firms highlighted in my portion of this feature happen to live in the same town, less than an hour away from the ODT office. Who’s up for a couple facility tours?
Musculoskeletal Tumor Surgery
Located in Parsippany, N.J., Onkos Surgical was founded in 2015 with the aim of focusing exclusively on the pediatric and adult surgical oncology market, particularly for musculoskeletal tumor patients. According to founder and CEO Patrick Treacy, surgical oncology had been an underserved market for far too long, and the complex disease state requires innovation and diverse solutions.“Onkos Surgical is 100% focused on bringing new technology and solutions to surgical oncologists,” he said when announcing the company’s establishment. “These specialists serve a population where a large portion of patients are children and the broader population may require personalized solutions. Surgeons, patients, and families deserve a focused company dedicated to this important cause.”
Its first FDA clearance came in 2016 with the ELEOS modular limb salvage system to treat significant bone loss due to cancer, trauma, or previous surgical procedures. The system leverages 3D printing capabilities to be a long-term reconstruction option for radical resection and replacement of the distal femur, proximal femur, proximal tibia, or total femur.
In addition to ELEOS, the company’s portfolio includes uDesign for surgical planning; MyTray personalized instrument trays; the GenVie biological adjuvant; and My3D personalized implant, instrument, and anatomic model designs.
The company made a couple large strides this year. In June, Onkos began the acquisition of Stryker’s Juvenile Tumor System (JTS)—an extendible prosthesis that promotes limb stabilization and growth for children affected by bone cancers, severe arthropathy, and severe trauma, revisions, oncology, and malignant diseases. The JTS helps non-invasively lengthen the implant post-surgery in a clinical setting. The company also received 510(k) clearance for its My3D personalized pelvic reconstruction system in July. The system includes 3D-printed implants, instruments, models, and an advanced planning tool for deformity, trauma, disease, and revisions where other treatments or revisions have failed.
Extremity Engineering
Also headquartered in Parsippany, Extremity Medical launched in 2008 to address the challenges extremity orthopedic surgeons face. The company develops products for fusion, fixation, and motion preservation for both upper and lower extremities.The company’s first product, the IO FiX MCP intraosseous fixation system, gained FDA clearance and CE mark approval in 2010. It enabled arthrodesis of the metacarpal phalangeal joints in the hand, the first such technology to do so at the time. Fusion can be achieved with a stronger construct and greater compression than traditional methods, using a fixed angle and avoiding irritation from plates, screws, and wires.
The company’s portfolio also includes UpperFiX and KinematX for upper extremities. For lower extremities, the franchise is comprised of the AlignX ankle fusion system, Axis Charcot fixation system, CompressX2 compression screws, EXPRESS staple system, HammerFiX, Jones union system, OMNI plating system, and OMNI ankle fracture system.
In April and May, the company initiated patent lawsuits against Zimmer Biomet, Nextremity Solutions, and Fusion Orthopedics. Extremity Medical alleges all three companies infringed on patents for its intraosseous fixation platform, for which the company has 49 in the U.S.
“Extremity Medical invests a great deal into building and defending its intellectual property portfolio,” said CEO Matthew Lyons when commenting on the Fusion Orthopedics lawsuit. “We will not tolerate competitors who blatantly copy our products while infringing on our patent rights. We intend to seek all remedies allowed by law.”
In happier news, the firm rolled out its OMNI stable ankle fracture system in May as well, expanding its purview into the trauma market. The system’s variety of low-profile anatomic plates closely match patient anatomy and reduce impingement of soft tissue. A proprietary Stability Post optimizes stability for patients with poor bone quality and/or segmental loss of bone.