Globe Newswire07.07.17
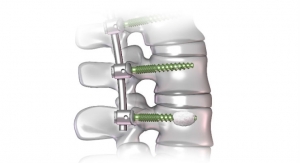
Since January 2015, Spine Wave Inc. has transformed its interbody fusion offering by launching the Leva PX Expandable Interbody Device, the Leva PF Interbody Device and the Velocity Expandable Interbody Device for posterior and transforaminal lumbar interbody fusions (PLIF & TLIF); the True Position Pivoting Spacer System for TLIF procedures using steerable crescent-style devices; and the Velocity L Expandable Interbody Device and the XLR II Access System for lateral lumbar interbody fusion (LLIF) procedures. Now, Spine Wave has successfully completed its limited market release of the Leva AF Interbody Device for anterior lumbar interbody fusion (ALIF) procedures and will fully launch the technology in September with the arrival of commercial-scale inventory quantities.
The launch of this new product marks Spine Wave’s entry into the large and growing ALIF implant market. The Leva AF Interbody Device features all-titanium construction, modular anterior plates and a selection of bone screws for integrated and standalone vertebral body fixation. As with all Leva brand products, the device also offers a large graft chamber that facilitates copious grafting with complete graft-to-endplate contact within the device. The Leva AF Interbody Device will also be complemented with a novel expandable option, the Leva AX Interbody Device, which is currently in limited release.
“The Leva AF Interbody Device is a very appealing new alternative for me in my practice,” said Matthew A. Hazzard, M.D., a neurological surgeon with The Longstreet Clinic P.C. in metropolitan Atlanta, Ga. “The device’s all titanium construction, very generous graft chambers, pre and post placement grafting capabilities and modular fixation components make it a unique and very versatile tool for my ALIF procedures, whether done with an anterior only or combined anterior and posterior approach.”
“As recently as two years ago, many surgeons and distributors associated Spine Wave exclusively with the StaXx XD Expandable Device. With the introduction of the Leva AF and AX Interbody Devices, Spine Wave now offers what is arguably the broadest range of novel and expandable interbody devices on the market,” said Mark LoGuidice, CEO of Spine Wave. “In addition to what is now a comprehensive lumbar portfolio, by the end of 2017 we expect to launch two important new cervical technologies that will complete our transformation to a full-line company.”
Spine Wave also offers the StaXx XD and XD Ti Expandable Devices, the Sniper Spine System complemented with Sniper XT extended tab screws, the Annex Adjacent Level System and the versatile and easy-to-use CapSure PS3 Spine System for traditional open thoracolumbar fixation. Spine Wave develops expandable fusion technologies. The company is based in Shelton, Conn.
The launch of this new product marks Spine Wave’s entry into the large and growing ALIF implant market. The Leva AF Interbody Device features all-titanium construction, modular anterior plates and a selection of bone screws for integrated and standalone vertebral body fixation. As with all Leva brand products, the device also offers a large graft chamber that facilitates copious grafting with complete graft-to-endplate contact within the device. The Leva AF Interbody Device will also be complemented with a novel expandable option, the Leva AX Interbody Device, which is currently in limited release.
“The Leva AF Interbody Device is a very appealing new alternative for me in my practice,” said Matthew A. Hazzard, M.D., a neurological surgeon with The Longstreet Clinic P.C. in metropolitan Atlanta, Ga. “The device’s all titanium construction, very generous graft chambers, pre and post placement grafting capabilities and modular fixation components make it a unique and very versatile tool for my ALIF procedures, whether done with an anterior only or combined anterior and posterior approach.”
“As recently as two years ago, many surgeons and distributors associated Spine Wave exclusively with the StaXx XD Expandable Device. With the introduction of the Leva AF and AX Interbody Devices, Spine Wave now offers what is arguably the broadest range of novel and expandable interbody devices on the market,” said Mark LoGuidice, CEO of Spine Wave. “In addition to what is now a comprehensive lumbar portfolio, by the end of 2017 we expect to launch two important new cervical technologies that will complete our transformation to a full-line company.”
Spine Wave also offers the StaXx XD and XD Ti Expandable Devices, the Sniper Spine System complemented with Sniper XT extended tab screws, the Annex Adjacent Level System and the versatile and easy-to-use CapSure PS3 Spine System for traditional open thoracolumbar fixation. Spine Wave develops expandable fusion technologies. The company is based in Shelton, Conn.