K2M Group Holdings Inc.11.30.17
K2M Group Holdings Inc., a global developer of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance, announced the completion of 300 surgical cases using the RHINE Cervical Disc System.* The announcement was made during the 12th Annual Meeting of the German Spine Society, occurring Nov. 30–Dec. 2, 2017, in Stuttgart, where the company is exhibiting its product portfolio and the Balance ACS platform.
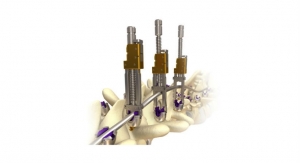
The RHINE Cervical Disc System* is an artificial disc replacement that features a one-piece compressible polymer core design with dome-shaped, plasma-coated endplates and a central-split keel. Proprietary molding technology is incorporated to minimize wear between the polymer core and metal endplates. The system’s instrumentation simplifies spinal surgery by integrating trialing and keel cutting into one instrument. A built-in adjustable stop allows for customized anterior or posterior positioning of the disc based on surgeon preference.
“The RHINE Cervical Disc was designed to mimic the characteristics of a normal healthy disc,” said Dr. Ardavan Ardeshiri, a neurosurgeon and spine surgeon at the Hessing Klinik in Augsburg, Germany, and an investigator of the RHINE Cervical Disc Prospective Observational Clinical Study. “The product design, combined with easy-to-use modular instrumentation and a simple surgical technique, provides surgeons with an advanced solution for cervical disc replacement.”
The RHINE Cervical Disc Prospective Observational Clinical Study is presently gathering additional operative data and feedback to confirm device performance, including radiographic data and clinical outcomes.
“K2M is proud to acknowledge the 300 surgeries that have been completed using our RHINE Cervical Disc System*,” said K2M Chairman, President, and CEO Eric Major. “RHINE, which received a CE Mark last year, is an advancement in cervical disc technology, and its continued commercialization and clinical validation in Europe is an important piece of our corporate strategy.”
In addition to the RHINE Cervical Disc System, K2M is showcasing differentiated spinal technologies from its comprehensive complex spine, minimally invasive, and degenerative portfolios. These products include the 3D-printed CASCADIA Interbody Systems featuring K2M’s Lamellar 3D Titanium Technology, the EVEREST Minimally Invasive XT Spinal System, the NILE Alternative Fixation and NILE Proximal Fixation Spinal Systems, the MESA Deformity Spinal System, the SAHARA AL Expandable Stabilization System, and more.
K2M is also featuring its Balance ACS platform, which provides solutions focused on achieving balance of the spine by addressing each anatomical vertebral segment with a 360-degree approach to the axial, coronal, and sagittal planes, emphasizing Total Body Balance as an important component of surgical success. K2M is demonstrating the BACS Surgical Planner, a surgical image measuring technology that assists in planning and preoperative implant selection.
*This product is intended for export and is not sold or offered for sale in the United States.
Since its inception, K2M has designed, developed, and commercialized complex spine and minimally invasive spine technologies and techniques used by spine surgeons to treat complicated spinal pathologies. K2M has leveraged these core competencies into Balance ACS, a platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes, with the goal of supporting the full continuum of care to facilitate quality patient outcomes. The Balance ACS platform, in combination with the company’s technologies, techniques and leadership in the 3D-printing of spinal devices, enable K2M to compete favorably in the global spinal surgery market.
The RHINE Cervical Disc System* is an artificial disc replacement that features a one-piece compressible polymer core design with dome-shaped, plasma-coated endplates and a central-split keel. Proprietary molding technology is incorporated to minimize wear between the polymer core and metal endplates. The system’s instrumentation simplifies spinal surgery by integrating trialing and keel cutting into one instrument. A built-in adjustable stop allows for customized anterior or posterior positioning of the disc based on surgeon preference.
“The RHINE Cervical Disc was designed to mimic the characteristics of a normal healthy disc,” said Dr. Ardavan Ardeshiri, a neurosurgeon and spine surgeon at the Hessing Klinik in Augsburg, Germany, and an investigator of the RHINE Cervical Disc Prospective Observational Clinical Study. “The product design, combined with easy-to-use modular instrumentation and a simple surgical technique, provides surgeons with an advanced solution for cervical disc replacement.”
The RHINE Cervical Disc Prospective Observational Clinical Study is presently gathering additional operative data and feedback to confirm device performance, including radiographic data and clinical outcomes.
“K2M is proud to acknowledge the 300 surgeries that have been completed using our RHINE Cervical Disc System*,” said K2M Chairman, President, and CEO Eric Major. “RHINE, which received a CE Mark last year, is an advancement in cervical disc technology, and its continued commercialization and clinical validation in Europe is an important piece of our corporate strategy.”
In addition to the RHINE Cervical Disc System, K2M is showcasing differentiated spinal technologies from its comprehensive complex spine, minimally invasive, and degenerative portfolios. These products include the 3D-printed CASCADIA Interbody Systems featuring K2M’s Lamellar 3D Titanium Technology, the EVEREST Minimally Invasive XT Spinal System, the NILE Alternative Fixation and NILE Proximal Fixation Spinal Systems, the MESA Deformity Spinal System, the SAHARA AL Expandable Stabilization System, and more.
K2M is also featuring its Balance ACS platform, which provides solutions focused on achieving balance of the spine by addressing each anatomical vertebral segment with a 360-degree approach to the axial, coronal, and sagittal planes, emphasizing Total Body Balance as an important component of surgical success. K2M is demonstrating the BACS Surgical Planner, a surgical image measuring technology that assists in planning and preoperative implant selection.
*This product is intended for export and is not sold or offered for sale in the United States.
Since its inception, K2M has designed, developed, and commercialized complex spine and minimally invasive spine technologies and techniques used by spine surgeons to treat complicated spinal pathologies. K2M has leveraged these core competencies into Balance ACS, a platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes, with the goal of supporting the full continuum of care to facilitate quality patient outcomes. The Balance ACS platform, in combination with the company’s technologies, techniques and leadership in the 3D-printing of spinal devices, enable K2M to compete favorably in the global spinal surgery market.