Michael Barbella, Managing Editor03.12.19
Christopher J. Centeno, M.D., could arguably be considered the “father” of regenerative medicine. He claims, after all, to be the first person on the planet to perform stem cell injections to treat joint arthritis and various other conditions.
There are several worthy contenders, including Arnie Caplan, Ph.D., pioneer of mesenchymal stem cell use (late 1980s); Philippe Henigou, developer of bone marrow concentrate treatment for bone disease (early 1990s); or Svante Gehring, M.D., the first physician to document use of platelet-rich plasma in ophthalmology (1999).
Centeno, however, believes the title should go to Ohio surgeon George S. Hackett, M.D., who clinically and scientifically demonstrated the efficacy of irritating tissue to promote healing back in 1963. Hackett’s studies showed that creating controlled inflammation could increase ligament size by up to 35-40 percent (more recent analyses have confirmed those initial findings).
While certainly revolutionary, Hackett’s work nevertheless was pre-dated by several thousand years: The concept of irritating tissue to promote healing actually originated with the ancient Greeks. Hippocrates reportedly treated Olympic javelin throwers with unstable shoulders by touching a “slender hot iron” to the joint’s ligaments (the heat would irritate the ligament, causing it to tighten).
Chronologically speaking, then, Hippocrates is deserving of the regenerative medicine “father” title. Yet Centeno maintains that Hackett warrants a place in history, if only for his contributions to the evolution of the field.
“So, is there one modern ‘father’?” Centeno, an international expert and regenerative medicine expert, asked in a blog last spring. “You could make arguments for Hernigou, Caplan, the early users of PRP, and so on. However, if you had to award the title to one person, that honor likely goes to George Hackett, who figured out that you could help ligaments heal with injections.”
Regardless of its origins, regenerative medicine has become a fast-growing subsegment of the multi billion-dollar orthobiologics industry, thanks to an aging world population and advanced stem cell/gene therapy technologies in developed countries. ODT’s January/February feature “Natural Selection” examines the innovation being developed in this burgeoning sector that eventually will contribute to its double-digit annual growth over the next half-decade.
Tyler Richardson, director of marketing, Surgical Biologics, at AMNIOX Medical Inc., a subsidiary of TissueTech Inc., shared his insights on the market for the story. His full input is provided in the following Q&A.
Michael Barbella: Please discuss the current trends you are see shaping the orthobiologics market.
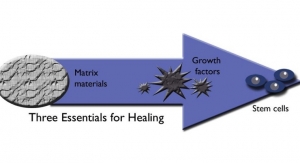
Tyler Richardson: Like many industries, there has been significant innovation in the orthobiologics market to optimize reconstructive materials and shift from a 'repair' to a 'regeneration' mindset. True regeneration is restorative and produces tissues with the same form and function as the native tissue.
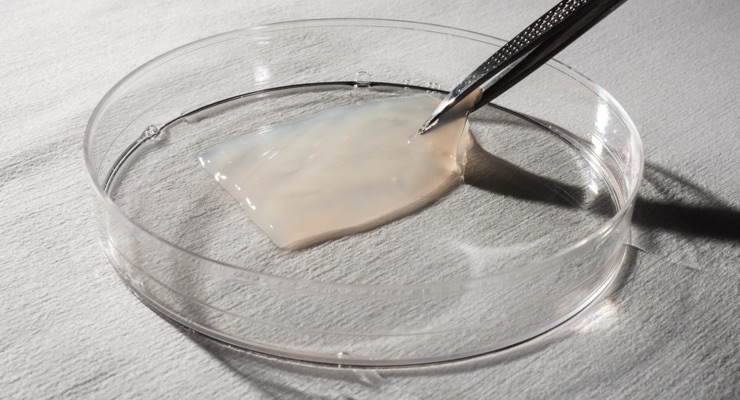
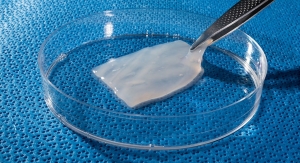
AMNIOX Medical products are human placental-based biological products. AMNIOX Medical (part of TissueTech Inc.) manufactures and markets products containing human umbilical cord (UC) and amniotic membrane (AM) tissues. The company's proprietary cryopreservation method to retain significantly greater amounts of key effector proteins and growth factors, cytokines, and the HC-HA/PTX3 complex, has the potential to facilitate such regenerative healing and help manage functional recovery. Heavy chain-Hyaluronan/Pentraxin 3 (HC-HA/PTX3) is a complex purified from human amniotic membrane and demonstrated to exert anti-inflammatory and anti-scarring actions.
Barbella: What factors are driving innovation in the orthobiologics market?
Richardson: The number one factor in clinicians/ and industry professionals' mind is not only patient outcomes but also a long-term safety record: allowing the patient's body to heal itself in an optimized environment, preventing recurrences, and restoring full function. Secondly, in today's healthcare environment, cost plays a major role. Utilizing a product that facilitates wound management and healing with superior outcomes, while having a long-term safety record can assist in reducing re-admissions and other costly expenses. The suite of products offered by AMNIOX Medical can help orchestrate healing using cryopreserved umbilical cord and amniotic membrame tissue allograft, facilitating regenerative healing of the soft tissues, bones, nerves, and cartilage while helping to manage functional recovery.
Barbella: What factors are impeding growth in the orthobiologics market? How can these challenges be overcome?
Richardson: Any time foreign material is placed in the body, there is a concern of what the patient's response to that material will be with respect to rejection. That's why using an immune-privileged biologic source like human placental tissue with devitalized cells is advantageous. AMNIOX Medical's impeccable safety record allows clinicians to confidently do what they do best—help their patients. The rising cost of healthcare can be a challenge as well. Promoting regenerative healing and incorporation may help reduce surgical complications and have positive downstream health economic effects.
Barbella: What new technologies are in the works?
Richardson: As the pioneering leader in cryopreserved umbilical cord and human placental tissue allografts, AMNIOX Medical has a number of products that are currently in clinical trials to gain new indications. Clarix cryopreserved umbilical cord allograft is currently available to help support the healing process across a range of surgical procedures. Clarix helps orchestrate a favorable healing environment. When incorporating Clarix into the treatment plan, physicians can help their patients heal by promoting a supportive healing environment for sports medicine injuries of the joints and soft tissues. Clarix is also suitable to assist with the restoration of the patient's range of motion. The Clarix family of umbilical cord allografts enhances a range of sports medicine procedures and is suitable to restore function and get patients back in the game. Clarix is also available in versatile and easy-to-use alternatives in various sizes and configurations to address the patient's specific treatment needs.
Barbella: Where does innovation come from in the orthobiologics market? How does AMNIOX Medical stay innovative amid all the competition?
Richardson: The foundation for innovation is basic science combined with continuous scientific research. With over 30 consecutive years of National Institutes of Health (NIH) grant-funded research and more than 300 peer-reviewed clinical and scientific papers, the co-founder of AMNIOX's corporate parent, TissueTech Inc., and Chief Scientific Officer Dr. Scheffer Tseng, is truly a pioneer in the field of regenerative biologics.
AMNIOX also has a dedicated team of scientists working on advanced research seeking to understand the core processes behind regenerative healing, as well as the impact. This has allowed us to identify how human placental tissue components, including the HC-HA/PTX3 complex, might have the potential to assist in managing pain and inflammation while facilitating cell migration, proliferation, and tissue regeneration. Our proprietary CryoTek cryopreservation method helps AMNIOX products maintain the structural and biochemical integrity of fresh tissue, facilitating the delivery of the HC-HA/PTX3 complext to the injury. Finally, AMNIOX has committed to setting the bar for proving enhanced safety and efficacy by working closely with the FDA and investing considerable resources into multiple clinical trials to gain new indications.
There are several worthy contenders, including Arnie Caplan, Ph.D., pioneer of mesenchymal stem cell use (late 1980s); Philippe Henigou, developer of bone marrow concentrate treatment for bone disease (early 1990s); or Svante Gehring, M.D., the first physician to document use of platelet-rich plasma in ophthalmology (1999).
Centeno, however, believes the title should go to Ohio surgeon George S. Hackett, M.D., who clinically and scientifically demonstrated the efficacy of irritating tissue to promote healing back in 1963. Hackett’s studies showed that creating controlled inflammation could increase ligament size by up to 35-40 percent (more recent analyses have confirmed those initial findings).
While certainly revolutionary, Hackett’s work nevertheless was pre-dated by several thousand years: The concept of irritating tissue to promote healing actually originated with the ancient Greeks. Hippocrates reportedly treated Olympic javelin throwers with unstable shoulders by touching a “slender hot iron” to the joint’s ligaments (the heat would irritate the ligament, causing it to tighten).
Chronologically speaking, then, Hippocrates is deserving of the regenerative medicine “father” title. Yet Centeno maintains that Hackett warrants a place in history, if only for his contributions to the evolution of the field.
“So, is there one modern ‘father’?” Centeno, an international expert and regenerative medicine expert, asked in a blog last spring. “You could make arguments for Hernigou, Caplan, the early users of PRP, and so on. However, if you had to award the title to one person, that honor likely goes to George Hackett, who figured out that you could help ligaments heal with injections.”
Regardless of its origins, regenerative medicine has become a fast-growing subsegment of the multi billion-dollar orthobiologics industry, thanks to an aging world population and advanced stem cell/gene therapy technologies in developed countries. ODT’s January/February feature “Natural Selection” examines the innovation being developed in this burgeoning sector that eventually will contribute to its double-digit annual growth over the next half-decade.
Tyler Richardson, director of marketing, Surgical Biologics, at AMNIOX Medical Inc., a subsidiary of TissueTech Inc., shared his insights on the market for the story. His full input is provided in the following Q&A.
Michael Barbella: Please discuss the current trends you are see shaping the orthobiologics market.
Tyler Richardson: Like many industries, there has been significant innovation in the orthobiologics market to optimize reconstructive materials and shift from a 'repair' to a 'regeneration' mindset. True regeneration is restorative and produces tissues with the same form and function as the native tissue.
AMNIOX Medical products are human placental-based biological products. AMNIOX Medical (part of TissueTech Inc.) manufactures and markets products containing human umbilical cord (UC) and amniotic membrane (AM) tissues. The company's proprietary cryopreservation method to retain significantly greater amounts of key effector proteins and growth factors, cytokines, and the HC-HA/PTX3 complex, has the potential to facilitate such regenerative healing and help manage functional recovery. Heavy chain-Hyaluronan/Pentraxin 3 (HC-HA/PTX3) is a complex purified from human amniotic membrane and demonstrated to exert anti-inflammatory and anti-scarring actions.
Barbella: What factors are driving innovation in the orthobiologics market?
Richardson: The number one factor in clinicians/ and industry professionals' mind is not only patient outcomes but also a long-term safety record: allowing the patient's body to heal itself in an optimized environment, preventing recurrences, and restoring full function. Secondly, in today's healthcare environment, cost plays a major role. Utilizing a product that facilitates wound management and healing with superior outcomes, while having a long-term safety record can assist in reducing re-admissions and other costly expenses. The suite of products offered by AMNIOX Medical can help orchestrate healing using cryopreserved umbilical cord and amniotic membrame tissue allograft, facilitating regenerative healing of the soft tissues, bones, nerves, and cartilage while helping to manage functional recovery.
Barbella: What factors are impeding growth in the orthobiologics market? How can these challenges be overcome?
Richardson: Any time foreign material is placed in the body, there is a concern of what the patient's response to that material will be with respect to rejection. That's why using an immune-privileged biologic source like human placental tissue with devitalized cells is advantageous. AMNIOX Medical's impeccable safety record allows clinicians to confidently do what they do best—help their patients. The rising cost of healthcare can be a challenge as well. Promoting regenerative healing and incorporation may help reduce surgical complications and have positive downstream health economic effects.
Barbella: What new technologies are in the works?
Richardson: As the pioneering leader in cryopreserved umbilical cord and human placental tissue allografts, AMNIOX Medical has a number of products that are currently in clinical trials to gain new indications. Clarix cryopreserved umbilical cord allograft is currently available to help support the healing process across a range of surgical procedures. Clarix helps orchestrate a favorable healing environment. When incorporating Clarix into the treatment plan, physicians can help their patients heal by promoting a supportive healing environment for sports medicine injuries of the joints and soft tissues. Clarix is also suitable to assist with the restoration of the patient's range of motion. The Clarix family of umbilical cord allografts enhances a range of sports medicine procedures and is suitable to restore function and get patients back in the game. Clarix is also available in versatile and easy-to-use alternatives in various sizes and configurations to address the patient's specific treatment needs.
Barbella: Where does innovation come from in the orthobiologics market? How does AMNIOX Medical stay innovative amid all the competition?
Richardson: The foundation for innovation is basic science combined with continuous scientific research. With over 30 consecutive years of National Institutes of Health (NIH) grant-funded research and more than 300 peer-reviewed clinical and scientific papers, the co-founder of AMNIOX's corporate parent, TissueTech Inc., and Chief Scientific Officer Dr. Scheffer Tseng, is truly a pioneer in the field of regenerative biologics.
AMNIOX also has a dedicated team of scientists working on advanced research seeking to understand the core processes behind regenerative healing, as well as the impact. This has allowed us to identify how human placental tissue components, including the HC-HA/PTX3 complex, might have the potential to assist in managing pain and inflammation while facilitating cell migration, proliferation, and tissue regeneration. Our proprietary CryoTek cryopreservation method helps AMNIOX products maintain the structural and biochemical integrity of fresh tissue, facilitating the delivery of the HC-HA/PTX3 complext to the injury. Finally, AMNIOX has committed to setting the bar for proving enhanced safety and efficacy by working closely with the FDA and investing considerable resources into multiple clinical trials to gain new indications.