Robert Joyce, President & Founder of FibreTuff04.03.23
The orthopedic implants used today were brought into the market over 20 years ago. These orthopedic implants continue to be the standard of care, however, they have also produced a variability of outcomes from a low grade infection to total body rejection. The removal of orthopedic implants in today’s health care market is the 5th largest expense for hospitals, generating poor return on investments in terms of patient outcomes and contributing to third party payor financial challenges.
The orthopedic implants referenced above are manufactured with biomaterials to include metals, ceramics and synthetic polymers. The synthetic polymers are known to have challenges including a poor interface to bone and show a binary radiographic image, either radiopaque or radiolucent. In either case, the synthetic polymer implants need to show evidence-based healing such as bone bridging, an extremely valuable tool for hospitals, physicians and insurance companies to work together for the best patient outcome.
Further, the synthetic polymers will not help gather radiographic image information to establish data on implant to bone interface, stabilization, or even infection. The most popular synthetic implantable polymer for orthopedics includes an extrudable radiolucent filament or sutures manufactured with Nylon or Polypropylene. Both Nylon and Polypropylene are biocompatible, not resorbable in the body and have long healing rates associated with implants. The Nylons have degradation in the body when exposed to hydrolysis while the Polypropylene is hydrophobic. The second most popular synthetic polymer for use as medical implants by orthopedic surgeons includes a radiolucent and non resorbable Poly ether ether ketone (PEEK) and High molecular weight Polyethylene (HMWPE). The PEEK and HMWPE biomaterials are biocompatible, hydrophobic, expensive and have been mostly used as bone substitutes that promote long healing rates. The healing rate for an implant with the above biomaterial is anywhere, from 12 weeks to 18 weeks, and occasionally 24 weeks.
By incorporating mineral additives with hydrophobic synthetic polymers, an improved implant to bone interface has been observed. However, the minerals have negative results that include loss of strength and elongation, degradation of the implant mechanical properties. Further, minerals have been used with synthetic polymers for identification of the orthopedic implant in radiographic images. The mineral additives or radio pacifiers will produce a binary or radiopaque image of an implant in the body. These minerals additives—barium sulfate, bismuth and tungsten with extremely high density—are very expensive and have cause for concern by lowering the biocompatibility level of an implant. These minerals can also cause processing challenges and a 50% loss in flexural and tensile strength in the orthopedic implant.
The synthetic polymers for implantables have a very poor implant to bone interface. This is largely due to the hydrophobicity or degradation when exposed to hydrolysis. To decrease hydrophobicity of today’s synthetic polymer implants, minerals are being utilized like hydroxyapatite, a calcium phosphate and or coating. The mineral additives compounded with synthetic polymer will compromise melt flow for processing sutures, filaments to 3D print.
In addition, the minerals can separate from the synthetic polymer implant in the body to be resorbed, a cause for infection. To reduce separation between mineral and synthetic polymers, additional processing aids such as couplings, stearate is required. These couplings normally have high amounts of toxicity, and if couplings aren’t used with synthetic polymers, the minerals may leach into the body. The coatings used with synthetic polymers require a coupling in producing a hydrogen bond. The hydrogen bond has not proven to be flawless and will tend to dissolve over time and flake between interfaces that cause infection.
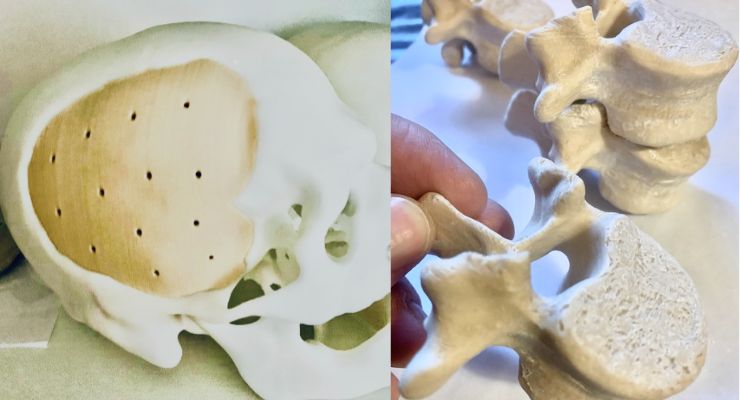
Robert Joyce, the President and Founder of Innovative Plastics and Molding believes cellulose fibers with polyamide and polyolefin has provided an attractive solution to address the orthopedic implant challenges with synthetic polymers. This biomaterial composition with cellulose fibers, trademarked FibreTuff, is a brand that has unexpectedly performed in both mechanical and biological testing. The medical grade composition has created novel monofilaments to 3D print bone like functional models for pre-clinical assessment.
Further, third party testing from the Implant Research Center (IRC) at Drexel University demonstrated the monofilaments produced 3D printed cellular structures to have a Young’s modulus similar to actual bone stress and strain curve. Dr. Edward Strong MD Professor and Vice-chairman Department of Otolaryngology University of California, Davis and Chairman of Computer Aided Maxillofacial Surgery (CAMLS) believes FibreTuff is a very realistic bone model and encourages orthopedic implant companies to investigate.
Innovative Plastics and Molding promotes the functionality of the 3D printed bone models will lead to superior prototypes and eventually permanent implants. The company has also produced 3D-printed phantoms and worked with orthopedic surgeons in producing 3D printed surgical guides. Dr. Hartman, a Cranio maxillofacial surgeon at Hartman Oral and Maxillofacial Surgery, PC. has printed surgical guides with FibreTuff. Dr. Hartman has confirmed 3D printing this biocompatible composition meets the standards set by Radiological Society of North America (RSNA) per the November 2018 published paper. Overall, the orthopedic surgeons, as well as University researchers have expressed interest in evaluating the bone like qualities for permanent implants to include but not limited to grafts, bone substitutes, and monofilaments that 3D print microporous scaffolds.
So far, the disruptive technology is claimed as a printed three dimensional non re sorbable cellular structure with hydrophilic ingredients to include cellulose fibers with a melted blend of polyamide and polyolefin. An unexpected behavior of the cellulose fiber in the thermoplastic matrix has shown radiopacity, a nonbinary image in Xray and CT scans at - 200 to 200 HU or Hounsfield units. These cellulose fibers “most importantly” replace traditional radio pacifiers that are radiopaque, heavy and expensive. The cellulose fibers mimic collagen fibers and promote anisotropy behavior with the melt blended polyamide and polyolefin. The fibers will help increase strength for bone like behavior and not reduce strength like minerals. The polyolefin helps with a lipid bilayer construct and the polyamide has repeating peptide units similar to proteins to provide food for cell growth.
Further investigation into characterization of the printed 3-dimensional article made with the advanced composition has identified unique features such as microporosity and bioactive performance. The microporous cellular structure design capabilities help reinforce tensile and flexural strength compared to other neat resins. Robert Joyce claims the advanced composition has fiber alignment with a high glass transition temperature of 70-90 tg. The high tg is due to the fibers because they do not conduct heat to close pore structure unlike PEEK.
Joyce further explains that the advanced composition can produce a microporous cellular structure at 60-80 micron, best for bone growth per research, while PEEK is printed at >120 a micropore structure. A 3D printed micropore cellular structure which includes additional mass and exhibits more strength is ”truly” additive manufacturing versus taking material out of the 3D printed “subtractive manufacturing” in a part like PEEK. Joyce further believes the 3D printed macro pore structure is necessary to create osteo integration to stabilize an implant.
The biological data generated from third parties show this advanced composition as biocompatible and it has passed in vivo testing to be certified as USP Class VI. Further, a Dr. Fayez Safadi from Northeast Ohio College of Medicine had shown osteoblast adhesion without minerals such as hydroxyapatite for a very probable and attractive bone interface. Dr. Prabaha Sikder at Cleveland State University has performed a bioactive investigation of the 3D printed microporous cellular structure made with the innovative composition. The data points generated have identified that the advanced composition is bioactive, showing apatite formation on the porous structure in simulated body fluids (SBF) with a negligible degradation rate. It’s unexpected the printed bone-like construction, having hydrophilicity will have negligible degradation and without pore closure in the SBF. This observation is leading to more research with 3D printing the innovative composition immersed in simulated body fluids (SBF) to possibly show regenerative properties.
Recently, two research papers have been published about the advanced biomaterial, cellulose fibers with polyamide and polyolefin for producing printed scaffolds. The research publications were under the direction of Dr. Ross Salary Director of Advanced Manufacturing at Marshall University in West Virginia. Dr. Salary has identified the biomaterial as novel, with disruptive features for 3D printing. He will be continuing the investigation of cellulose fiber with polyamide and polyolefin to generate more testing data for advancing the technology to replace less effective and costly synthetic polymers for orthopedic implants.
Innovative Plastics and Molding will collaborate with Dr. Salary to focus on the mechanical functionality of the monofilaments for 3D printing the novel based medical composition. The collaborative research will intend to show relevant data for helping patients, hospitals, medical centers with evidence-based healing. Innovative Plastics and Molding will not only require more research but General Laboratory Practice (GLP) testing data for achieving permanent implant status. To achieve permanent implant status, the company will need to submit 3D printed parts for ISO 10993 testing. By completing these ISO tests and submitting a 510K permanent implantable 3D printed part to FDA this disruptive technology can be validated.
Robert Joyce is President and Founder of FibreTuff.
The orthopedic implants referenced above are manufactured with biomaterials to include metals, ceramics and synthetic polymers. The synthetic polymers are known to have challenges including a poor interface to bone and show a binary radiographic image, either radiopaque or radiolucent. In either case, the synthetic polymer implants need to show evidence-based healing such as bone bridging, an extremely valuable tool for hospitals, physicians and insurance companies to work together for the best patient outcome.
Further, the synthetic polymers will not help gather radiographic image information to establish data on implant to bone interface, stabilization, or even infection. The most popular synthetic implantable polymer for orthopedics includes an extrudable radiolucent filament or sutures manufactured with Nylon or Polypropylene. Both Nylon and Polypropylene are biocompatible, not resorbable in the body and have long healing rates associated with implants. The Nylons have degradation in the body when exposed to hydrolysis while the Polypropylene is hydrophobic. The second most popular synthetic polymer for use as medical implants by orthopedic surgeons includes a radiolucent and non resorbable Poly ether ether ketone (PEEK) and High molecular weight Polyethylene (HMWPE). The PEEK and HMWPE biomaterials are biocompatible, hydrophobic, expensive and have been mostly used as bone substitutes that promote long healing rates. The healing rate for an implant with the above biomaterial is anywhere, from 12 weeks to 18 weeks, and occasionally 24 weeks.
By incorporating mineral additives with hydrophobic synthetic polymers, an improved implant to bone interface has been observed. However, the minerals have negative results that include loss of strength and elongation, degradation of the implant mechanical properties. Further, minerals have been used with synthetic polymers for identification of the orthopedic implant in radiographic images. The mineral additives or radio pacifiers will produce a binary or radiopaque image of an implant in the body. These minerals additives—barium sulfate, bismuth and tungsten with extremely high density—are very expensive and have cause for concern by lowering the biocompatibility level of an implant. These minerals can also cause processing challenges and a 50% loss in flexural and tensile strength in the orthopedic implant.
The synthetic polymers for implantables have a very poor implant to bone interface. This is largely due to the hydrophobicity or degradation when exposed to hydrolysis. To decrease hydrophobicity of today’s synthetic polymer implants, minerals are being utilized like hydroxyapatite, a calcium phosphate and or coating. The mineral additives compounded with synthetic polymer will compromise melt flow for processing sutures, filaments to 3D print.
In addition, the minerals can separate from the synthetic polymer implant in the body to be resorbed, a cause for infection. To reduce separation between mineral and synthetic polymers, additional processing aids such as couplings, stearate is required. These couplings normally have high amounts of toxicity, and if couplings aren’t used with synthetic polymers, the minerals may leach into the body. The coatings used with synthetic polymers require a coupling in producing a hydrogen bond. The hydrogen bond has not proven to be flawless and will tend to dissolve over time and flake between interfaces that cause infection.
Addressing Orthopedic Implant Challenges with Synthetic Polymers—FibreTuff
A six-year investigation by Innovative Plastics and Molding Inc has shown a promising and novel medical grade biomaterial technology for 3D printing.Robert Joyce, the President and Founder of Innovative Plastics and Molding believes cellulose fibers with polyamide and polyolefin has provided an attractive solution to address the orthopedic implant challenges with synthetic polymers. This biomaterial composition with cellulose fibers, trademarked FibreTuff, is a brand that has unexpectedly performed in both mechanical and biological testing. The medical grade composition has created novel monofilaments to 3D print bone like functional models for pre-clinical assessment.
Further, third party testing from the Implant Research Center (IRC) at Drexel University demonstrated the monofilaments produced 3D printed cellular structures to have a Young’s modulus similar to actual bone stress and strain curve. Dr. Edward Strong MD Professor and Vice-chairman Department of Otolaryngology University of California, Davis and Chairman of Computer Aided Maxillofacial Surgery (CAMLS) believes FibreTuff is a very realistic bone model and encourages orthopedic implant companies to investigate.
Innovative Plastics and Molding promotes the functionality of the 3D printed bone models will lead to superior prototypes and eventually permanent implants. The company has also produced 3D-printed phantoms and worked with orthopedic surgeons in producing 3D printed surgical guides. Dr. Hartman, a Cranio maxillofacial surgeon at Hartman Oral and Maxillofacial Surgery, PC. has printed surgical guides with FibreTuff. Dr. Hartman has confirmed 3D printing this biocompatible composition meets the standards set by Radiological Society of North America (RSNA) per the November 2018 published paper. Overall, the orthopedic surgeons, as well as University researchers have expressed interest in evaluating the bone like qualities for permanent implants to include but not limited to grafts, bone substitutes, and monofilaments that 3D print microporous scaffolds.
So far, the disruptive technology is claimed as a printed three dimensional non re sorbable cellular structure with hydrophilic ingredients to include cellulose fibers with a melted blend of polyamide and polyolefin. An unexpected behavior of the cellulose fiber in the thermoplastic matrix has shown radiopacity, a nonbinary image in Xray and CT scans at - 200 to 200 HU or Hounsfield units. These cellulose fibers “most importantly” replace traditional radio pacifiers that are radiopaque, heavy and expensive. The cellulose fibers mimic collagen fibers and promote anisotropy behavior with the melt blended polyamide and polyolefin. The fibers will help increase strength for bone like behavior and not reduce strength like minerals. The polyolefin helps with a lipid bilayer construct and the polyamide has repeating peptide units similar to proteins to provide food for cell growth.
Further investigation into characterization of the printed 3-dimensional article made with the advanced composition has identified unique features such as microporosity and bioactive performance. The microporous cellular structure design capabilities help reinforce tensile and flexural strength compared to other neat resins. Robert Joyce claims the advanced composition has fiber alignment with a high glass transition temperature of 70-90 tg. The high tg is due to the fibers because they do not conduct heat to close pore structure unlike PEEK.
Joyce further explains that the advanced composition can produce a microporous cellular structure at 60-80 micron, best for bone growth per research, while PEEK is printed at >120 a micropore structure. A 3D printed micropore cellular structure which includes additional mass and exhibits more strength is ”truly” additive manufacturing versus taking material out of the 3D printed “subtractive manufacturing” in a part like PEEK. Joyce further believes the 3D printed macro pore structure is necessary to create osteo integration to stabilize an implant.
The biological data generated from third parties show this advanced composition as biocompatible and it has passed in vivo testing to be certified as USP Class VI. Further, a Dr. Fayez Safadi from Northeast Ohio College of Medicine had shown osteoblast adhesion without minerals such as hydroxyapatite for a very probable and attractive bone interface. Dr. Prabaha Sikder at Cleveland State University has performed a bioactive investigation of the 3D printed microporous cellular structure made with the innovative composition. The data points generated have identified that the advanced composition is bioactive, showing apatite formation on the porous structure in simulated body fluids (SBF) with a negligible degradation rate. It’s unexpected the printed bone-like construction, having hydrophilicity will have negligible degradation and without pore closure in the SBF. This observation is leading to more research with 3D printing the innovative composition immersed in simulated body fluids (SBF) to possibly show regenerative properties.
Recently, two research papers have been published about the advanced biomaterial, cellulose fibers with polyamide and polyolefin for producing printed scaffolds. The research publications were under the direction of Dr. Ross Salary Director of Advanced Manufacturing at Marshall University in West Virginia. Dr. Salary has identified the biomaterial as novel, with disruptive features for 3D printing. He will be continuing the investigation of cellulose fiber with polyamide and polyolefin to generate more testing data for advancing the technology to replace less effective and costly synthetic polymers for orthopedic implants.
Innovative Plastics and Molding will collaborate with Dr. Salary to focus on the mechanical functionality of the monofilaments for 3D printing the novel based medical composition. The collaborative research will intend to show relevant data for helping patients, hospitals, medical centers with evidence-based healing. Innovative Plastics and Molding will not only require more research but General Laboratory Practice (GLP) testing data for achieving permanent implant status. To achieve permanent implant status, the company will need to submit 3D printed parts for ISO 10993 testing. By completing these ISO tests and submitting a 510K permanent implantable 3D printed part to FDA this disruptive technology can be validated.
Robert Joyce is President and Founder of FibreTuff.