08.18.15
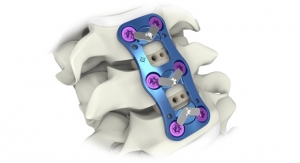
Carlsbad, Calif.-based Aurora Spine Corp. received CE Mark issuance for its full line of its TiNano sterile-packed titanium coated spinal fusion implants made of polyether ether ketone (PEEK) thermoplastic material.
"This CE Mark issuance is a major accomplishment for Aurora Spine. TiNano intervertebral implants are developed to support the entire spine from cervical to lumbar regions and to accommodate the company's Zip Minimally Invasive Interspinous Fusion System portfolio as part of Aurora's Screwless Procedure," said Trent J. Northcutt, president and CEO of Aurora Spine.
TiNano is Aurora Spine's unique Titanium Plasma Spray coating on PEEK Interbody implants. TiNano-coated implants provide the advantages of the titanium material as well as the modulus and post-op imaging benefits of PEEK fusion implants.
"We are very excited that after a successful product launch in the United States now we can offer our sterile packed, TiNano-coated fusion cage portfolio to all European markets," said Michael Thompson, director of regulatory affairs and quality assurance.
"The CE Mark approval includes several interbody fusion devices, including configurations for anterior cervical, anterior lumbar, posterior lumbar, transforaminal lumbar and the fastest growing segment, lateral Lumbar interbody spacers," said Laszlo Garamszegi, chief technology officer.
Aurora Spine is an early-stage company focused on screwless regenerative spinal implant technologies.
"This CE Mark issuance is a major accomplishment for Aurora Spine. TiNano intervertebral implants are developed to support the entire spine from cervical to lumbar regions and to accommodate the company's Zip Minimally Invasive Interspinous Fusion System portfolio as part of Aurora's Screwless Procedure," said Trent J. Northcutt, president and CEO of Aurora Spine.
TiNano is Aurora Spine's unique Titanium Plasma Spray coating on PEEK Interbody implants. TiNano-coated implants provide the advantages of the titanium material as well as the modulus and post-op imaging benefits of PEEK fusion implants.
"We are very excited that after a successful product launch in the United States now we can offer our sterile packed, TiNano-coated fusion cage portfolio to all European markets," said Michael Thompson, director of regulatory affairs and quality assurance.
"The CE Mark approval includes several interbody fusion devices, including configurations for anterior cervical, anterior lumbar, posterior lumbar, transforaminal lumbar and the fastest growing segment, lateral Lumbar interbody spacers," said Laszlo Garamszegi, chief technology officer.
Aurora Spine is an early-stage company focused on screwless regenerative spinal implant technologies.