Certifications
- All News
- Breaking News
- Certifications
- Clinical Trials
- Device Approvals & Patents
- Financial & Deals
- International
- Market Data & Trends
- ODT Best Bets
- OEM News
- Orthopedic Innovators
- People
- Product & Service Releases
- Products
- Quality, Recalls & Risk
- Regulatory & Legal
- Supplier & Contract Mfg.
- Videobites
-
3-D Printing/Additive Mfg.

ISO 13485 Certification Granted to Sculpteo
The company's certification covers class I and class IIa devices.Michael Barbella, Managing Editor 07.18.24
-

Zimmer Biomet ROSA Shoulder System Receives FDA Clearance
The system becomes the first-to-market robotic surgical assistant for shoulder arthroplasty.Rachel Klemovitch, Assistant Editor 02.22.24
-

Mediliant Acquires CTE Solutions
Makes further progress in forming an orthopedic contract manufacturing group.Charles Sternberg, Assistant Editor 03.09.21
-

Fundamental Surgery Achieves CME Accreditation from AAOS
The company's haptic VR platform qualifies for CME credits.Fundamental Surgery 12.18.19
-
Spine/Neurology

Intralink-Spine Achieves ISO 13485 Certification
Company has also completed ISO 10993 biocompatibility testing.Business Wire 11.20.19
-
3-D Printing/Additive Mfg.

PrinterPrezz Announces ISO 13485:2016 Certification for 3D Printing of Medical Devices
PrinterPrezz announced that the company has achieved ISO 13485:2016 certification for Quality Management Systems for medical devices.Business Wire 06.19.19
-
Instruments | Instruments, Cases & Trays

gSource Achieves ISO 13485:2016 Recertification
Certificate was awarded to the company by British Standards Institute (BSI).gSource LLC 12.13.18
-

Five Star Companies Achieves ISO 13485:2016
Latest version of the quality management system standard addresses risk-based approaches to product development.Five Star Companies 10.24.18
-
Machining & Tooling | Software & Quality

LaVezzi Precision Achieves ISO 13485:2016 Certification
The company has demonstrated its quality management system consistently provides products meeting requirements.LaVezzi Precision 08.31.18
-

UL Achieves Transition Accreditation for ISO 13485:2016
Medical device customers can now receive audits and certificates to standard to align with global regulatory requirements.Underwriters Laboratories 03.30.17
-

OrthoPro Services Earns Highly Coveted BOC Facility Accreditation
Facility accreditation shows a supplier's focus and commitment on providing quality patient care.PR Newswire 11.02.16
-
Spine/Neurology

Phygen LLC Receives Notice of Allowance on AutoLok Fixation Screw Assembly
Notice was received under the company's U.S. Patent Application 13/274,243.PR Newswire 09.30.16
-
MIS/Robotics

Bioness Vector Gait and Safety System Receives Expanded UL Certification
The Vector Gait and Safety System helps increase patient mobility while preventing falls and injury.PR Newswire 08.25.16
-
BioCut Systems Introduces 'Cancellous C5' Bone Cutting Press
Company also achieves ISO 9001 certification.BioCut Systems 06.06.16
-
Large Joint
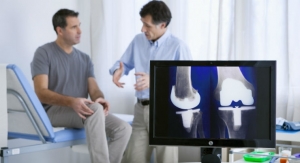
First Advanced Certification for Total Hip and Total Knee Replacement Awarded
Thomas Jefferson University Hospital in Philadelphia is the first in the United States to achieve certification.The Joint Commission 04.27.16
-
Spine/Neurology
Spine Pain Management Expands Board
Company also meets IEC standard for medical electrical equipment.10.06.15
-
Imaging | Instruments | Spine/Neurology

Intellirod Spine Planning for Growth (includes video)
Recently achieved milestones position the orthopedic sensor technology company for continued expansion.Online Exclusives Christopher Delporte, Editor 06.30.15
-
Trauma/Sports
Magnesium Elektron U.K. Facility Earns Quality ISO
Facility makes bioabsorbable magnesium alloys.05.19.15
-
Spine/Neurology

SpineGuard Earns Certification in Brazil
Company will begin Brazilian registration process for PediGuard device.11.28.14
-
Spine/Neurology
Custom Spine Gets ISO Certification and CE Mark
European OK opens international markets for the firm.01.30.12













