PR Newswire06.02.16
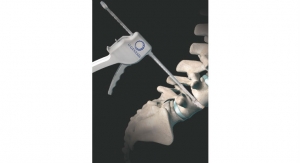
Pinnacle Spine Group LLC has launched its InFill Anterior Lumbar Interbody Fusion (ALIF) device. This product is designed to maximize wall thickness and graft chamber volume, ensuring optimal contact with the vertebral endplates by both the interbody device and bone graft material.
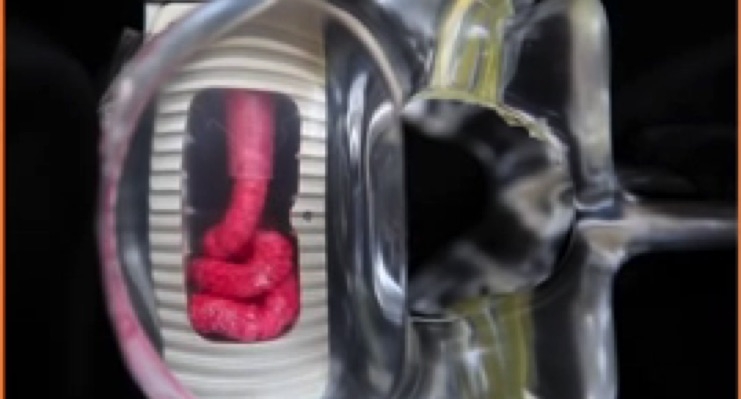
The InFill ALIF device is compatible with Pinnacle’s InFill Graft Delivery System, which allows surgeons to deliver the optimal volume of graft material to the implant in situ without disrupting the interbody device’s placement, providing an opportunity to achieve maximum bone graft-to-endplate contact.
Watch the video below to learn more about Pinnacle's InFill Graft Delivery System:
“Anterior lumbar spinal fusion is a common spine fusion technique, but often must be combined with an additional posterior approach to provide immediate stabilization. Science shows that in many unilateral posterior lumbar interbody fusion procedures, less than 50 percent of the disc area is actually grafted,” said Zach Sowell, president of Pinnacle Spine Group. “We are excited to add the InFill ALIF device to our growing line of patented fusion systems.”
Features of the ALIF device include:
Pinnacle Spine Group has pioneered the concept of placing bone graft material in situ into an implanted device to allow optimal bone graft material volume and maximized contact with vertebral endplates—a critical element for a successful spine fusion outcome.
Pinnacle Spine’s InFill Fusion Systems include a full line of interbody fusion devices engineered for easier insertion, reduced subsidence through maximum contact with the apophyseal ring, a generous bone grafting chamber, and compatibility with the InFill Graft Delivery System. The backbone of the technology is based on controlled and precise in situ placement of bone graft material.
Pinnacle recently announced its first international patent, (awarded in China), which followed receipt of three U.S. patents on the InFill Fusion System.
Pinnacle Spine Group develops medical devices conceived in the operating room for surgical spinal procedures. Backed by the private equity firm Sowell & Co., the company is based in Dallas, Texas.
The InFill ALIF device is compatible with Pinnacle’s InFill Graft Delivery System, which allows surgeons to deliver the optimal volume of graft material to the implant in situ without disrupting the interbody device’s placement, providing an opportunity to achieve maximum bone graft-to-endplate contact.
Watch the video below to learn more about Pinnacle's InFill Graft Delivery System:
“Anterior lumbar spinal fusion is a common spine fusion technique, but often must be combined with an additional posterior approach to provide immediate stabilization. Science shows that in many unilateral posterior lumbar interbody fusion procedures, less than 50 percent of the disc area is actually grafted,” said Zach Sowell, president of Pinnacle Spine Group. “We are excited to add the InFill ALIF device to our growing line of patented fusion systems.”
Features of the ALIF device include:
- Optimization for use in the L5/S1 disc space;
- A gently bulleted nose for ease of insertion;
- The option for in situ delivery of bone graft material to ensure a more complete fill;
- A large graft chamber which facilitates formation of a robust fusion column;
- An aggressive tooth design to prevent displacement of the device;
- A large surface area which provides maximum contact with the apophyseal ring, helping to restore and maintain disc space and height; and
- Tantalum rod markers which provide excellent radiographic visualization of interbody device orientation and placement.
Pinnacle Spine Group has pioneered the concept of placing bone graft material in situ into an implanted device to allow optimal bone graft material volume and maximized contact with vertebral endplates—a critical element for a successful spine fusion outcome.
Pinnacle Spine’s InFill Fusion Systems include a full line of interbody fusion devices engineered for easier insertion, reduced subsidence through maximum contact with the apophyseal ring, a generous bone grafting chamber, and compatibility with the InFill Graft Delivery System. The backbone of the technology is based on controlled and precise in situ placement of bone graft material.
Pinnacle recently announced its first international patent, (awarded in China), which followed receipt of three U.S. patents on the InFill Fusion System.
Pinnacle Spine Group develops medical devices conceived in the operating room for surgical spinal procedures. Backed by the private equity firm Sowell & Co., the company is based in Dallas, Texas.