DePuy Synthes06.01.17
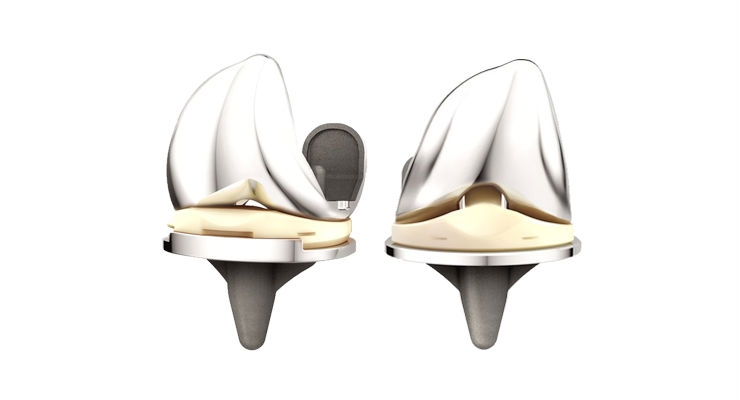
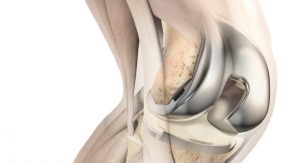
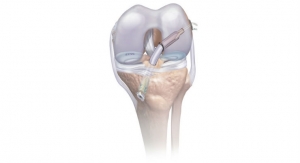
DePuy Synthes, part of the Johnson & Johnson Family of Companies, today announced new ATTUNE Knee implant survivorship data based on an independent analysis of 10,605 ATTUNE Knee implantations, showing that the cumulative revision rate for the ATTUNE Knee is 1.3 percent at four years (98.7 percent implant survivorship at four years), comparing favorably to the 1.9 percent cumulative revision rate (98.1 percent implant survivorship at four years) for the overall class of total knee replacement.1
Of the 10,605 ATTUNE Knees included in the analysis from the National Joint Registry for England, Wales, Northern Ireland and the Isle of Man (NJR), 46 required revision. Based on a separate analysis, the report concluded that 63 ATTUNE Knee revisions would be expected at four years—a difference that was statistically significant (p<0.05).1 The expected number of ATTUNE Knee revisions was calculated by the NJR based on duration of implantation, age group, gender and indications.
DePuy Synthes announced the four-year data in Vienna, coinciding with the 18th European Federation of the National Associations of Orthopaedics and Traumatology (EFORT) Congress. The full analysis, including a breakdown of the reasons for revision, is available on ATTUNEevidence.com.
“This most recent analysis adds to the body of evidence on the positive performance of the ATTUNE Knee,” said Joshua Bridgens, DePuy Synthes franchise medical director. “The information and data in this analysis, such as reasons for revision and preliminary findings on patient-reported outcomes, continue to validate the design principles and our expectations for the clinical performance of the ATTUNE Knee.”
NJR publishes an annual report including 1-, 3-, and 5-year outcome data; however, additional survivorship data is now also available more frequently to manufacturers who subscribe to the NJR’s Implant Summary Report service.
DePuy Synthes is committed to the continued evaluation of outcomes through its comprehensive ATTUNE Knee Evidence Generation Program that, in addition to national joint registries, gathers information from investigator-initiated studies, independent studies and company initiated studies.
References
1 National Joint Registry for England, Wales, Northern Ireland and the Isle of Man. Implant Summary Report for DePuy ATTUNE CR and ATTUNE PS. NJR Database extract April 7, 2017, pages 1-17. Licensed for use until April 19, 2018. Available at www.attuneevidence.com.
Of the 10,605 ATTUNE Knees included in the analysis from the National Joint Registry for England, Wales, Northern Ireland and the Isle of Man (NJR), 46 required revision. Based on a separate analysis, the report concluded that 63 ATTUNE Knee revisions would be expected at four years—a difference that was statistically significant (p<0.05).1 The expected number of ATTUNE Knee revisions was calculated by the NJR based on duration of implantation, age group, gender and indications.
DePuy Synthes announced the four-year data in Vienna, coinciding with the 18th European Federation of the National Associations of Orthopaedics and Traumatology (EFORT) Congress. The full analysis, including a breakdown of the reasons for revision, is available on ATTUNEevidence.com.
“This most recent analysis adds to the body of evidence on the positive performance of the ATTUNE Knee,” said Joshua Bridgens, DePuy Synthes franchise medical director. “The information and data in this analysis, such as reasons for revision and preliminary findings on patient-reported outcomes, continue to validate the design principles and our expectations for the clinical performance of the ATTUNE Knee.”
NJR publishes an annual report including 1-, 3-, and 5-year outcome data; however, additional survivorship data is now also available more frequently to manufacturers who subscribe to the NJR’s Implant Summary Report service.
DePuy Synthes is committed to the continued evaluation of outcomes through its comprehensive ATTUNE Knee Evidence Generation Program that, in addition to national joint registries, gathers information from investigator-initiated studies, independent studies and company initiated studies.
References
1 National Joint Registry for England, Wales, Northern Ireland and the Isle of Man. Implant Summary Report for DePuy ATTUNE CR and ATTUNE PS. NJR Database extract April 7, 2017, pages 1-17. Licensed for use until April 19, 2018. Available at www.attuneevidence.com.