Business Wire10.26.17
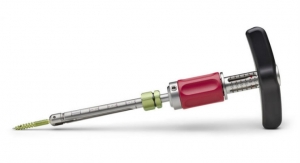
AMNIOX Medical Inc., a TissueTech, Inc. company, highlighted the introduction of RESPINA its cryopreserved umbilical cord/amniotic membrane matrix at the North American Spine Society’s (NASS) annual meeting in Orlando, Fla., October 25 - 27.
AMNIOX Medical’s proprietary amniotic membrane technology has been clinically proven in a randomized controlled trial to enhance surgical outcomes in microdiscectomy, improving function and reducing both pain and the incidence of recurrent herniation1. The umbilical cord/amniotic membrane tissue, processed through CRYOTEK technology, offers a simple solution for physicians to support restorative healing and improve procedural benefits.
RESPINA will also be recognized as the 2017 Orthopedics This Week Spine Technology Award recipient in the biomaterials and biologics category at NASS.
“Spine Tech Awards recognize excellence, innovation, and the advancement of spinal care,” said Tom Dugan, AMNIOX Medical CEO. “This recognition underscores the clinical evidence and economic benefits that continue to accumulate in support of the use of our cryopreserved placental tissue with its decades long track record in ocular surgery. The restorative healing proven in microdiscectomy has encouraged us to explore other promising applications in spine and orthopedics. This is a simple, natural, safe, and proven platform technology that we are excited to launch into spine.”
Microdiscectomy is a minimally invasive procedure with a small incision required to remove the herniated lumbar disc material. This removal relieves pressure on the nearby spinal cord and nerves. The biological components of RESPINA have been demonstrated to modulate local inflammation and minimize the potential for the formation of scar tissue and adhesions.
To learn more about AMNIOX Medical and RESPINA, NASS 2017 attendees may visit booth 237.
Reference
1Anderson, D. Greg; Popov, Victor; Raines, Andrew L., Ph.D., Connell, Julie PhD. “Cryopreserved Amniotic Membrane Improves Clinical Outcomes Following Microdiscectomy.” Clinical Spine Surgery, 26 May 2017, http://journals.lww.com/jspinaldisorders/Abstract/publishahead/Cryopreserved_Amniotic_Membrane_Improves_Clinical.9962
AMNIOX Medical’s proprietary amniotic membrane technology has been clinically proven in a randomized controlled trial to enhance surgical outcomes in microdiscectomy, improving function and reducing both pain and the incidence of recurrent herniation1. The umbilical cord/amniotic membrane tissue, processed through CRYOTEK technology, offers a simple solution for physicians to support restorative healing and improve procedural benefits.
RESPINA will also be recognized as the 2017 Orthopedics This Week Spine Technology Award recipient in the biomaterials and biologics category at NASS.
“Spine Tech Awards recognize excellence, innovation, and the advancement of spinal care,” said Tom Dugan, AMNIOX Medical CEO. “This recognition underscores the clinical evidence and economic benefits that continue to accumulate in support of the use of our cryopreserved placental tissue with its decades long track record in ocular surgery. The restorative healing proven in microdiscectomy has encouraged us to explore other promising applications in spine and orthopedics. This is a simple, natural, safe, and proven platform technology that we are excited to launch into spine.”
Microdiscectomy is a minimally invasive procedure with a small incision required to remove the herniated lumbar disc material. This removal relieves pressure on the nearby spinal cord and nerves. The biological components of RESPINA have been demonstrated to modulate local inflammation and minimize the potential for the formation of scar tissue and adhesions.
To learn more about AMNIOX Medical and RESPINA, NASS 2017 attendees may visit booth 237.
Reference
1Anderson, D. Greg; Popov, Victor; Raines, Andrew L., Ph.D., Connell, Julie PhD. “Cryopreserved Amniotic Membrane Improves Clinical Outcomes Following Microdiscectomy.” Clinical Spine Surgery, 26 May 2017, http://journals.lww.com/jspinaldisorders/Abstract/publishahead/Cryopreserved_Amniotic_Membrane_Improves_Clinical.9962