Business Wire03.20.18
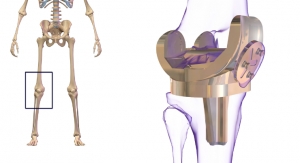
Exactech, a developer and producer of bone and joint restoration products and biologic solutions for extremities, knee and hip announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market the Novation XLE Vitamin E acetabular polyethylene liners for hip replacement surgery. These liners are designed to work in conjunction with the Novation Crown Cup Acetabular System including InteGrip printed, 3D porous material.
The advanced bearing material in the new XLE liners is designed for low wear and contains vitamin E. Oxidation in-vivo can contribute to polyethylene degradation in orthopedic implant polyethylene. Vitamin E, a natural antioxidant, is blended into the raw material of Novation XLE liners to mitigate oxidative effects both before and after implantation.
This product complements Exactech’s Hip product family, which also includes the Alteon HA Femoral Stem, Tapered Wedge Femoral Stem, Neck Preserving Stem, and Monobloc Revision Stem.
The advanced bearing material in the new XLE liners is designed for low wear and contains vitamin E. Oxidation in-vivo can contribute to polyethylene degradation in orthopedic implant polyethylene. Vitamin E, a natural antioxidant, is blended into the raw material of Novation XLE liners to mitigate oxidative effects both before and after implantation.
This product complements Exactech’s Hip product family, which also includes the Alteon HA Femoral Stem, Tapered Wedge Femoral Stem, Neck Preserving Stem, and Monobloc Revision Stem.