Stellenbosch University06.19.18
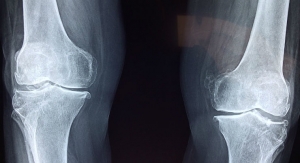
People whose knees have been affected by osteoarthritis, also known as the “wear and tear” arthritis, often have to get knee implants to repair the damage. But if a knee implant does not fit the specific individual, it could lead to soft tissue damage, collapsing of the underlying bone, loosening of the implant and an increased likelihood of joint pain after an operation.
"One possible solution is to design implants that are patient-specific, anatomically accurate, reproduce normal knee motion and are ultimately repeatable," said Dr. Johan van der Merwe, a lecturer in the Department of Mechanical and Mechatronic Engineering at Stellenbosch University (SU). Van der Merwe, who is also a member of the Biomedical Engineering Research Group in the same department, recently obtained his doctorate in Mechatronic Engineering at SU. He focused on developing a repeatable, semi-automated method of generating patient-specific implant components that reproduces an individual's healthy anatomy to facilitate natural joint movement after an operation.
Van der Merwe pointed out that the knee is one of the joints most susceptible to osteoarthritis, a form of arthritis that occurs when the protective cartilage in joints breaks down.
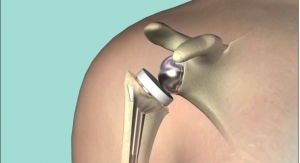
Van der Merwe said for localized osteoarthritis, surgical reconstruction or replacement of only the damaged part of the knee (Unicompartmental Knee Arthroplasty) may be preferred over the replacement of the entire joint (Total Knee Arthroplasty). Projections are that by 2030 three million knee replacement surgeries will be done globally.
"Replacing or repairing only the damaged part may result in faster recovery, improved post-operative joint movement, retains as much of the joint's natural anatomy as possible and is done at a lower cost," he said. "But designing patient-specific Unicompartmental Knee Replacements ultimately rely on a technician's interpretation and skill in order to consistently reproduce healthy knee shapes. This is a difficult problem—what is considered healthy for one patient might not be the same for another."
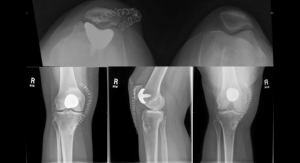
Van der Merwe pointed out that there is often a mismatch between current standardized sizes of commercially available implants and the shape and form of an individual patient's knee. This generally requires the surgeon to make the patient fit the implant, and sometimes even deviate from the ideal intended implant placement based on experience and current best practices to achieve good outcomes.
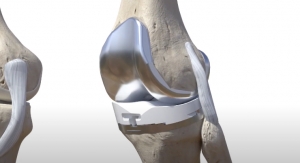
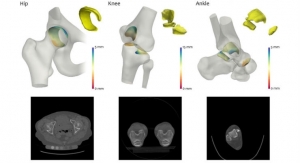
In an attempt to address these shortcomings, Van der Merwe developed a model based on the various shapes of healthy knees, which could then be used to estimate the healthy shape of damaged parts on a patient's knee.
Van der Merwe used this information to write a semi-automated design program that fits smooth surfaces onto the estimated healthy shape to create implants ready for computer-assisted manufacturing.
"To validate the shapes created by our implant design method, we performed the semi-automated design process on a set of test knees,” he said. “Our semi-automated approach to designing patient-specific Unicompartmental Knee Replacements resulted in implants that compare favorably with the original, normal knees. These implant components matched the normal anatomy of the knee. The method proved able to repeatedly generate anatomically compatible implant shapes. We hope that the design could therefore facilitate the restoration of more normal joint function after an operation."
Van der Merwe said by tailoring each implant to the individual, the mismatch between the shape and form of the patient's knee and that of the implant could be reduced.
"This could potentially lead to reduced recovery times and shorter operating times because the patient's bone surfaces don't have to be modified a lot. Implants with a better fit and, in our case, more natural shape, could potentially also improve movement of the joint after an operation."
Van der Merwe said people in their 50s and early 60s who already suffer from osteoarthritis and need a knee replacement, and more active patients that would like to preserve their quality of life for a period before getting a total knee replacement as the disease progresses would benefit from his research.
He added that the implant designed as part of his study is ready for preliminary clinical testing.
"One possible solution is to design implants that are patient-specific, anatomically accurate, reproduce normal knee motion and are ultimately repeatable," said Dr. Johan van der Merwe, a lecturer in the Department of Mechanical and Mechatronic Engineering at Stellenbosch University (SU). Van der Merwe, who is also a member of the Biomedical Engineering Research Group in the same department, recently obtained his doctorate in Mechatronic Engineering at SU. He focused on developing a repeatable, semi-automated method of generating patient-specific implant components that reproduces an individual's healthy anatomy to facilitate natural joint movement after an operation.
Van der Merwe pointed out that the knee is one of the joints most susceptible to osteoarthritis, a form of arthritis that occurs when the protective cartilage in joints breaks down.
Van der Merwe said for localized osteoarthritis, surgical reconstruction or replacement of only the damaged part of the knee (Unicompartmental Knee Arthroplasty) may be preferred over the replacement of the entire joint (Total Knee Arthroplasty). Projections are that by 2030 three million knee replacement surgeries will be done globally.
"Replacing or repairing only the damaged part may result in faster recovery, improved post-operative joint movement, retains as much of the joint's natural anatomy as possible and is done at a lower cost," he said. "But designing patient-specific Unicompartmental Knee Replacements ultimately rely on a technician's interpretation and skill in order to consistently reproduce healthy knee shapes. This is a difficult problem—what is considered healthy for one patient might not be the same for another."
Van der Merwe pointed out that there is often a mismatch between current standardized sizes of commercially available implants and the shape and form of an individual patient's knee. This generally requires the surgeon to make the patient fit the implant, and sometimes even deviate from the ideal intended implant placement based on experience and current best practices to achieve good outcomes.
In an attempt to address these shortcomings, Van der Merwe developed a model based on the various shapes of healthy knees, which could then be used to estimate the healthy shape of damaged parts on a patient's knee.
Van der Merwe used this information to write a semi-automated design program that fits smooth surfaces onto the estimated healthy shape to create implants ready for computer-assisted manufacturing.
"To validate the shapes created by our implant design method, we performed the semi-automated design process on a set of test knees,” he said. “Our semi-automated approach to designing patient-specific Unicompartmental Knee Replacements resulted in implants that compare favorably with the original, normal knees. These implant components matched the normal anatomy of the knee. The method proved able to repeatedly generate anatomically compatible implant shapes. We hope that the design could therefore facilitate the restoration of more normal joint function after an operation."
Van der Merwe said by tailoring each implant to the individual, the mismatch between the shape and form of the patient's knee and that of the implant could be reduced.
"This could potentially lead to reduced recovery times and shorter operating times because the patient's bone surfaces don't have to be modified a lot. Implants with a better fit and, in our case, more natural shape, could potentially also improve movement of the joint after an operation."
Van der Merwe said people in their 50s and early 60s who already suffer from osteoarthritis and need a knee replacement, and more active patients that would like to preserve their quality of life for a period before getting a total knee replacement as the disease progresses would benefit from his research.
He added that the implant designed as part of his study is ready for preliminary clinical testing.