PR Newswire07.03.18
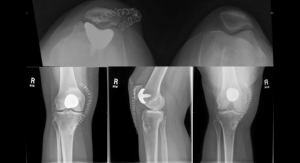
Arthrosurface Inc., a provider of minimally invasive, motion preserving joint restoration systems, has received U.S. Food and Drug Administration (FDA) 510(k) clearance to market the Patellofemoral (PF) WaveKahuna Arthroplasty System. The PF WaveKahuna Arthroplasty System is a line extension to the company’s previously cleared and commercially marketed family of HemiCAP Patellofemoral and PF Wave Systems.
Shane Shankle, vice president of U.S. sales, saidd, “As a market leader in Patellofemoral Arthroplasty, we are very excited to round out our line with the PF WaveKahuna System. The larger implants will allow a surgeon to choose from a wide range of size offerings to better serve the patient’s needs and allow them to maintain their quality of life.”
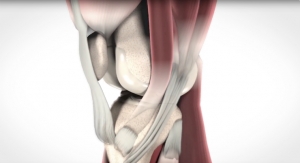
The PF WaveKahuna restores the unique articular surface geometry of the patella and femoral trochlea while maintaining the patient’s native anatomy. It’s extended lateral aspect and trochlear geometry is designed to address challenging patellofemoral tracking and stability conditions as well as providing increased coverage of the trochlear groove. Furthermore, the patella design combines the clinical benefits of both the anatomic and dome patella implants from the PF Wave System. Additionally, the PF WaveKahuna femoral component is designed to mate with the currently marketed Arthrosurface PF Wave Fixation Component. The existing surgical technique is unchanged, with the addition of a superior ream step.
“The FDA clearance comes shortly after data recently presented at ESSKA confirmed significantly improved postoperative outcomes with high patient satisfaction and no progression of knee arthritis at five years follow-up. The timing could not be better,” said Dawn Wilson, vice president of quality and regulatory at Arthrosurface.
Arthrosurface Inc. is a global orthopedic medical technology business providing a portfolio of products and instrumentation used to treat upper and lower extremity orthopedic conditions caused by trauma, injury, and arthritic disease. Arthrosurface provides joint preservation solutions with inlay arthroplasty. Its product offerings include joint preservation implants, instruments, and orthobiologics. Founded in 2002, Arthrosurface markets and distributes its products in the United States and around the world.
Shane Shankle, vice president of U.S. sales, saidd, “As a market leader in Patellofemoral Arthroplasty, we are very excited to round out our line with the PF WaveKahuna System. The larger implants will allow a surgeon to choose from a wide range of size offerings to better serve the patient’s needs and allow them to maintain their quality of life.”
The PF WaveKahuna restores the unique articular surface geometry of the patella and femoral trochlea while maintaining the patient’s native anatomy. It’s extended lateral aspect and trochlear geometry is designed to address challenging patellofemoral tracking and stability conditions as well as providing increased coverage of the trochlear groove. Furthermore, the patella design combines the clinical benefits of both the anatomic and dome patella implants from the PF Wave System. Additionally, the PF WaveKahuna femoral component is designed to mate with the currently marketed Arthrosurface PF Wave Fixation Component. The existing surgical technique is unchanged, with the addition of a superior ream step.
“The FDA clearance comes shortly after data recently presented at ESSKA confirmed significantly improved postoperative outcomes with high patient satisfaction and no progression of knee arthritis at five years follow-up. The timing could not be better,” said Dawn Wilson, vice president of quality and regulatory at Arthrosurface.
Arthrosurface Inc. is a global orthopedic medical technology business providing a portfolio of products and instrumentation used to treat upper and lower extremity orthopedic conditions caused by trauma, injury, and arthritic disease. Arthrosurface provides joint preservation solutions with inlay arthroplasty. Its product offerings include joint preservation implants, instruments, and orthobiologics. Founded in 2002, Arthrosurface markets and distributes its products in the United States and around the world.