Business Wire08.02.18
RTI Surgical Inc., a global surgical implant company, and Aziyo Biologics Inc., a fully integrated regenerative medicine company, announced the signing of an agreement under which Aziyo will provide ViBone to RTI Surgical for exclusive distribution in the U.S.
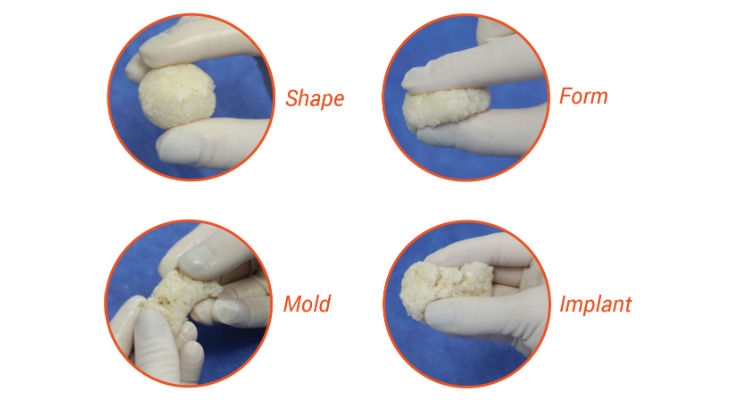
ViBone is a bone repair product designed to perform and handle more closely to autograft in a variety of orthopedic procedures. ViBone is processed using a proprietary method optimized to protect and preserve the health of native bone cells to potentially enhance new bone formation.
“ViBone is a next generation bone graft product that meets the diverse needs of surgeons with high potential for improved outcomes for patients,” said Camille Farhat, president and CEO, RTI Surgical. “ViBone will join RTI’s existing biologic portfolio as another important option for surgeons.”
RTI’s company strategy centers on organic and acquisitive growth focused on differentiated products supported by clinical data. RTI’s commercial team will integrate ViBone into its sales and growth strategy in the U.S., effective immediately. Together, RTI and Aziyo will initiate new research on ViBone in the coming months to build on the characterization and other data currently available.
“We are excited to partner with RTI Surgical, a recognized leader in spine and tissue-based implants, to bring this innovative option to more surgeons, and to further investigate the value it provides to patients,” said Ron Lloyd, president and CEO, Aziyo Biologics. “This agreement provides a platform for RTI and Aziyo to mutually grow and expand in the spinal fusion and orthopedic markets in service to more patients.”
ViBone is a bone repair product designed to perform and handle more closely to autograft in a variety of orthopedic procedures. ViBone is processed using a proprietary method optimized to protect and preserve the health of native bone cells to potentially enhance new bone formation.
“ViBone is a next generation bone graft product that meets the diverse needs of surgeons with high potential for improved outcomes for patients,” said Camille Farhat, president and CEO, RTI Surgical. “ViBone will join RTI’s existing biologic portfolio as another important option for surgeons.”
RTI’s company strategy centers on organic and acquisitive growth focused on differentiated products supported by clinical data. RTI’s commercial team will integrate ViBone into its sales and growth strategy in the U.S., effective immediately. Together, RTI and Aziyo will initiate new research on ViBone in the coming months to build on the characterization and other data currently available.
“We are excited to partner with RTI Surgical, a recognized leader in spine and tissue-based implants, to bring this innovative option to more surgeons, and to further investigate the value it provides to patients,” said Ron Lloyd, president and CEO, Aziyo Biologics. “This agreement provides a platform for RTI and Aziyo to mutually grow and expand in the spinal fusion and orthopedic markets in service to more patients.”